Abstract
Background. Cystinuria is a rare autosomal recessive disorder leading to recurrent cystine stone formation, often necessitating repeated surgical interventions. In Türkiye, tiopronin—a proven medical therapy—has become inaccessible since 2019, raising concerns about its economic and clinical consequences. This study aimed to compare the annual hospital costs of surgical management with the theoretical cost of tiopronin therapy in pediatric cystinuria patients.
Methods. This single-center, retrospective study included 10 consecutive pediatric patients (median age: 12 years; range: 3-17 years) with genetically or biochemically confirmed cystinuria who underwent surgery for cystine stones in 2023. The annual cost of surgical management was calculated by summing all direct medical costs (operating room, anesthesia, devices, hospitalization, and related diagnostics) obtained from the hospital billing database. The theoretical annual cost of tiopronin therapy was calculated based on the last accessible market price of the drug in Türkiye, converted to Turkish Lira at the 2023 average exchange rate, and adjusted per patient using a standard dosing regimen of 15 mg/kg/day. Costs were compared using the Mann-Whitney U test.
Results. The median annual surgical cost per patient was 49,936 TL (range: 14,791–84,576 TL), compared to a theoretical tiopronin cost of 27,923 TL (range: 9,307–27,925 TL). Surgical management was significantly more expensive than tiopronin therapy (p=0.001) in 9 of 10 cases. Six patients had a history of tiopronin use, and five of them experienced a surgery-free interval during medication (median duration: 3 years; range: 2-6 years). Preliminary follow-up data for 2024 revealed that 70% of patients required further surgical interventions (median: 2 surgeries per patient), demonstrating the persistent and recurrent nature of the disease when managed solely with surgery.
Conclusion. Surgical treatment of cystinuria poses a substantially higher economic burden compared to tiopronin therapy. Our findings support the reintroduction of tiopronin into the Turkish healthcare system, particularly for pediatric patients with frequent stone recurrence.
Keywords: cystinuria, tiopronin, pediatric urolithiasis, healthcare costs, cost-effectiveness, Türkiye
Introduction
Cystinuria is a rare autosomal recessive disorder leading to impaired renal reabsorption of cystine, resulting in recurrent stone formation.1,2 Cystinuria has a reported incidence of 1 in 2,155 to 1 in 7,000 in children, and constitutes 6–8% of pediatric and 1–2% of adult urolithiasis.1-3 The disease often manifests in childhood and necessitates repeated surgical interventions, placing a significant burden on patients, families, and healthcare systems.4,5
The cornerstone of cystinuria management involves high fluid intake and urinary alkalization. For patients who continue to form stones despite these conservative measures, the 2025 European Association of Urology (EAU) Guidelines recommend the use of cystine-binding thiol agents, primarily tiopronin and D-penicillamine, and in selected cases captopril, to reduce cystine excretion and prevent recurrence.4-6 Tiopronin has demonstrated efficacy in reducing urinary cystine levels and preventing new stone formation. In pediatric cystinuria, long-term treatment with cystine-binding thiols, including tiopronin, has been associated with longer stone-free intervals and a reduced need for repeated endoscopic or percutaneous stone surgery.4-8
In Türkiye, tiopronin was available and reimbursed until 2019, but was subsequently withdrawn from the market due to cost considerations. We noticed an increase in surgical frequency among our pediatric cystinuria patients following this withdrawal. This study aimed to objectively compare the annual hospital costs associated with surgical management with the theoretical cost of tiopronin therapy in pediatric patients treated at our center in 2023.
Materials and Methods
Study design and ethical approval
This single-center, retrospective cost-comparison study was conducted at a tertiary care university hospital. The study was approved by the local ethics committee (Approval number: 25-4T/110).
Patient characteristics
The study included the first ten consecutive pediatric patients (aged 0-18 years) with a confirmed diagnosis of cystinuria (based on stone analysis and/or genetic testing for SLC3A1 or SLC7A9 mutations) who underwent at least one surgical intervention for cystine stones at our institution during the 2023 calendar year. Patients with non-cystine stones, those with incomplete medical or financial records, and patients who were primarily managed at another center were excluded from the analysis.
A total of 10 patients who met the inclusion criteria were identified from the hospital’s electronic database. The collected data included age, sex, body weight, the number and types of surgical procedures performed in 2023, surgical interventions in 2024 (where available), history of previous tiopronin use, and current medications.
Treatment protocols and cost calculation
The total annual cost for surgical management per patient were obtained directly from the hospital’s billing database and are presented in Turkish Lira (TL) at 2023 prices. These billings include costs for the operating room, anesthesia, surgical devices and equipment, inpatient hospital stay, medications, and laboratory and radiological investigations related to the procedure. The total cost was calculated by summing all direct medical costs associated with each stone-related procedure in 2023.
The theoretical annual cost of tiopronin therapy was calculated individually for each patient using a standardized dosing and pricing approach. Tiopronin was assumed to be administered as 100-mg tablets, and pricing was based on the last accessible market price of the commercially available tiopronin formulation in Türkiye (€34.11 per 100-tablet package). This price was converted to Turkish Lira using the average exchange rate for 2023. The daily tiopronin dose was calculated as 15 mg/kg/day, divided into three equal doses. For each patient, the total daily dose (mg/day) was divided by the tablet strength to determine the number of tablets required per day. The annual cost was calculated by multiplying the daily tablet requirement by 365 days and the unit cost per tablet.
Statistical analysis
Statistical analyses were performed using SPSS Statistics for Windows, Version 11.5 (SPSS Inc., Chicago, IL, USA). Normality of continuous data distribution was assessed using the Shapiro-Wilk test, histograms, and Q-Q plots. Since the cost data were not normally distributed, non-parametric tests were used. The Mann-Whitney U test was applied to compare the annual costs of surgical management versus theoretical tiopronin therapy. A p-value of < 0.05 was considered statistically significant. Continuous variables are presented as median and range.
Results
The median patient age was 12 years (range: 3-17 years). The median patient weight was 33 kg (range: 12-87 kg). Seven children were boys and three were girls. The diagnosis of cystinuria was genetically confirmed in three patients (SLC3A1 mutations in two patients and an SLC7A9 mutation in one patient). For the remaining seven patients, the diagnosis was established based on consistent biochemical parameters (urinary cystine excretion >400 mg/L) and stone analysis. All patients received standard conservative management consisting of potassium citrate at 1–3 mEq/kg/day in 2–3 divided doses, titrated to maintain a urinary pH target of 7.0–7.5. In addition, patients were instructed to maintain a high fluid intake (>2–3 L/m²/day) and dietary sodium restriction (<2 mEq/kg/day) to optimize urine dilution and cystine solubility.
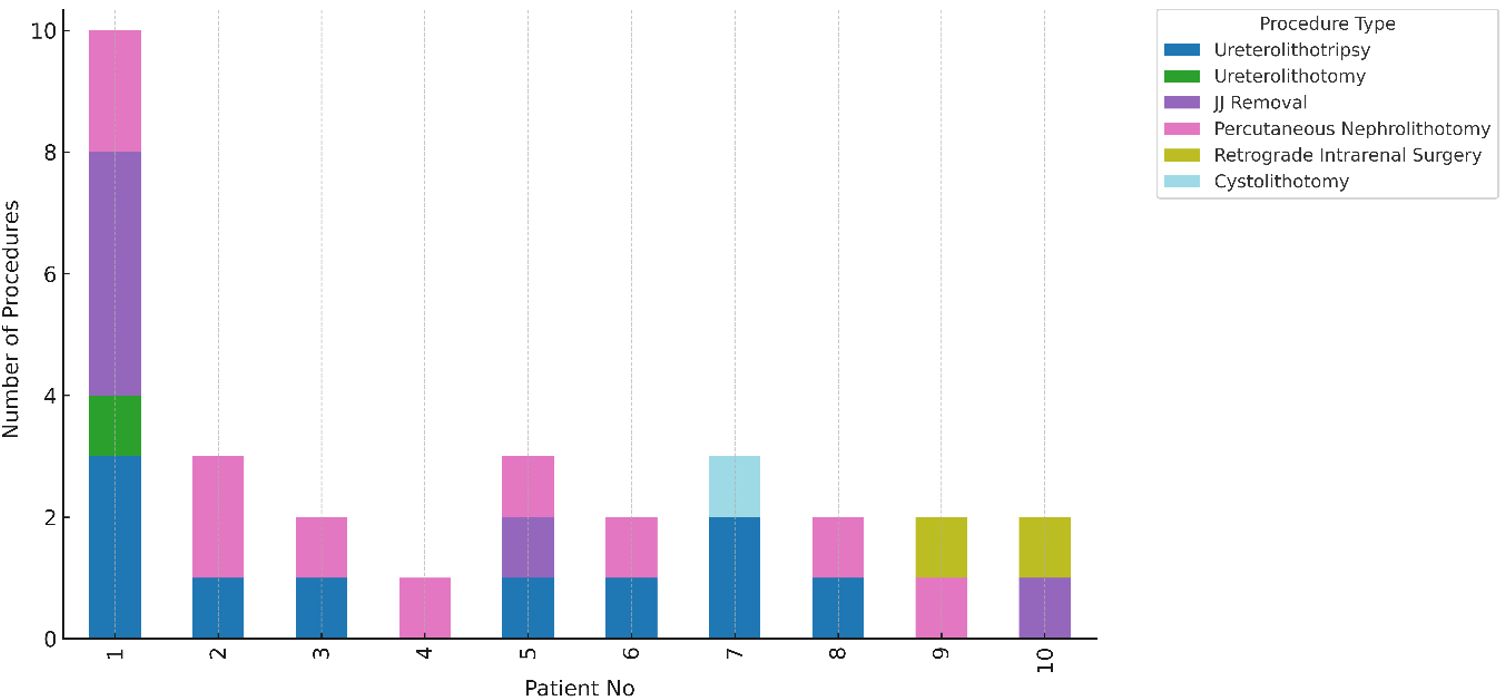
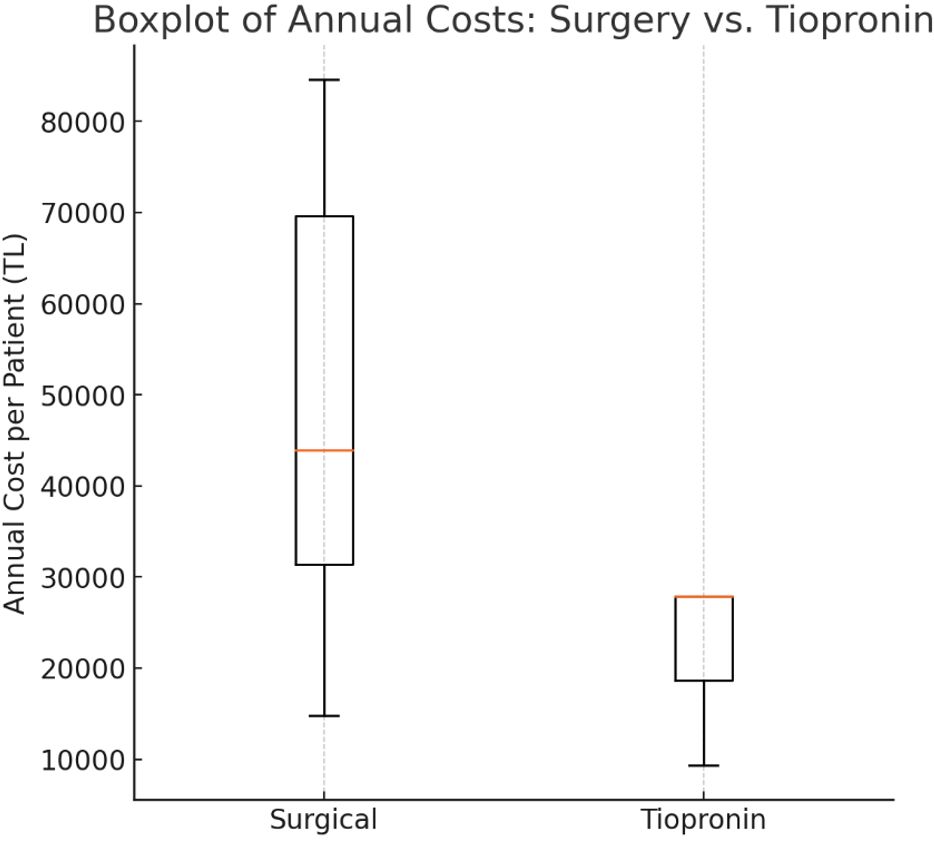
These ten patients each underwent a median of 2 surgeries in 2023, with individual surgical counts ranging from one to ten. The surgeries performed included ureterolithotripsy, retrograde intrarenal surgery, cystolithotomy, and percutaneous nephrolithotomy (PNL) (Fig. 1). The median annual cost for surgical management per patient was 49,936 TL, with costs ranging between 14,791 TL and 84,576 TL.
Based on the dosing regimen and the price of tiopronin (€34.11 per 100 tablets), the median annual cost of tiopronin per patient was 27,923 TL (range: 9,307 TL–27,925 TL). Statistical analysis demonstrated that the cost of tiopronin therapy was significantly lower than the hospital costs associated with surgical management (p = 0.001). In all but one patient, the cost of surgical management exceeded the estimated cost of tiopronin therapy (Table I). A cost analysis demonstrated a statistically significant difference favoring tiopronin over surgical treatment (p = 0.001) (Fig. 2).
| The table includes the number of surgeries per patient, total surgical costs, estimated annual tiopronin therapy costs (based on weight-adjusted dosing), and the cost ratio of surgical to medical management. In all but one case, surgical treatment was more expensive than tiopronin therapy. | ||||
| Table I. Comparison of annual hospital costs for surgical management versus theoretical tiopronin therapy in 10 patients treated for cystinuria in 2023. | ||||
| Patient no |
|
|
|
|
| 1 |
|
|
|
|
| 2 |
|
|
|
|
| 3 |
|
|
|
|
| 4 |
|
|
|
|
| 5 |
|
|
|
|
| 6 |
|
|
|
|
| 7 |
|
|
|
|
| 8 |
|
|
|
|
| 9 |
|
|
|
|
| 10 |
|
|
|
|
We also evaluated whether prior use of tiopronin influenced the frequency of surgical interventions. Among these ten patients, six had a history of tiopronin use. All six patients with a history of tiopronin use initiated therapy with potassium citrate and tiopronin following their initial surgical diagnosis. Despite overall requiring multiple surgeries in a year, only one of these six patients underwent surgical intervention during the period in which tiopronin was actively administered. The remaining five patients experienced a surgery-free interval during tiopronin therapy, with a median duration of three years (range:2-6 years). One patient (16.6%) required a percutaneous nephrolithotomy and JJ stent removal during the second year of treatment. This same patient experienced disease recurrence and required a repeat PNL one year later, suggesting a partial or inadequate response to medical therapy in this specific case.
Despite undergoing surgical procedures in 2023, 70% of the cohort (7 out of 10 patients) required at least one additional surgical intervention in 2024. The median number of surgeries per patient in this period was 2 (range: 0-3).
Discussion
Cystinuria management entails a dual challenge encompassing both clinical complexity and economic burden. Our findings clearly show that the average annual cost of surgical intervention in cystinuric patients far exceeds the projected cost of tiopronin therapy. Importantly, tiopronin therapy not only reduces the frequency of stone formation but may also prevent the need for surgical treatment altogether, as evidenced by the low surgical intervention rate during prior tiopronin availability in our patient cohort. This clinical benefit is supported by a detailed series from Türkiye by Asi et al., who evaluated 36 pediatric patients with cystine stone disease over a median follow-up of 6.4 years. The median age at diagnosis was 42 months, and most children presented with recurrent bilateral stones.9 Pharmacological management, including potassium citrate and cystine-binding thiols, was associated with a reduction in stone recurrence from 100% to 60% over time. Predictors of successful stone clearance included early initiation of medical therapy and strict adherence to alkalization. During follow-up, patients required various surgical procedures such as ureterorenoscopy, percutaneous nephrolithotomy, and shock-wave lithotripsy, but the frequency of repeat interventions decreased substantially among those maintained on medical therapy.9 This is in alignment with previous studies supporting long-term medical therapy for cystine stone prevention.4,5,7,9
Our follow-up data from 2024 clearly demonstrate that surgical intervention does not break the cycle of recurrence. In contrast, historical data from our cohort show that tiopronin therapy provided a median surgery-free interval of 3 years. This stark difference emphasizes the urgent need for accessible medical therapy to reduce the cumulative economic and clinical burden of repeated surgeries.
Complementary non-pharmacological strategies have also been shown to support long-term disease control in hereditary renal disorders, including structured hydration routines, improved patient–family education, and adherence-support measures.10 The 2025 EAU Guidelines emphasize aggressive hydration, dietary sodium restriction, and urinary alkalization—with a target urine pH of 7.0–7.5—as the core of recurrence prevention.6 Recent systematic reviews highlight emerging therapeutic avenues such as molecular pathway-targeted treatments, genotype-guided approaches, and novel cystine-solubilizing compounds, although these remain investigational.1,7 Pediatric literature further underscores the importance of individualized metabolic evaluation, urine monitoring, and multidisciplinary follow-up to optimize outcomes in children with cystinuria.11
In addition to tiopronin, alternative pharmacological agents have been explored for the management of cystinuria; however, their overall clinical effectiveness remains limited. D-penicillamine is capable of forming soluble cystine complexes, but its long-term efficacy is compromised by poor tolerability and limited sustained use in pediatric populations.4,5 Captopril, despite containing a sulfhydryl group, has demonstrated only modest and inconsistent reductions in urinary cystine levels and has not reliably prevented stone recurrence in clinical practice.1,2 Alpha-lipoic acid has shown preliminary biochemical efficacy in experimental and early clinical studies; however, evidence supporting its clinical effectiveness, particularly in children, remains insufficient.7 Consequently, current international guidelines do not recommend these agents as first-line therapy for pediatric cystinuria.6
However, in patients who continue to form stones despite adequate hydration, urinary alkalization, and dietary sodium restriction, cystine-binding thiol drugs remain the cornerstone of second-line therapy. According to contemporary recommendations, including the 2025 EAU Guidelines, agents such as tiopronin and D-penicillamine are the primary pharmacological options for reducing urinary cystine levels, while captopril may be considered in selected cases when other thiol drugs are unavailable or not tolerated.6 By increasing cystine solubility, these agents help prevent recurrent stone formation and may reduce the need for repeated surgical interventions.2,9
Tiopronin is generally better tolerated than other thiol-binding agents; however, adverse effects such as gastrointestinal intolerance, rash, arthralgia, and fatigue have been reported.4,5 Clinically relevant proteinuria and rare cases of membranous nephropathy may occur, particularly with long-term use, necessitating regular monitoring of urine protein and renal function.2 A recent systematic review on pharmacological metaphylaxis in cystinuria confirmed that, while thiol agents effectively reduce stone recurrence, tiopronin demonstrates a more favorable tolerability profile than D-penicillamine when appropriately monitored.7,12
Tiopronin was withdrawn from the Turkish market due to its low demand and high procurement cost. However, the findings of this study highlight that surgical management, although more readily accessible, is associated with significantly higher annual costs. Importantly, the present analysis only accounts for hospital-billed expenses and does not capture the full economic burden of care. Additional unaccounted costs include the use of specialized and costly surgical instruments for stone procedures, which are not always reimbursed. Furthermore, the economic impact extends beyond direct medical expenses; families may experience substantial productivity loss due to time away from work during their child’s hospitalization and postoperative care. Recurrent stone formation and repeated surgeries also pose long-term risks to renal health, potentially resulting in chronic kidney damage and escalating healthcare costs over time.
These findings emphasize the need for a broader health-economic perspective in evaluating treatment strategies for cystinuria. The case for reintroducing tiopronin to the Turkish market is not only a clinical necessity but also an economic imperative. Future cost-effectiveness studies should include these broader direct and indirect costs to better estimate the long-term benefit of medical therapy in cystinuria.
Limitations
First, we included only direct hospital costs, as indirect costs (e.g., parental work absenteeism, travel expenses, and long-term renal complications) were not retrospectively accessible. Second, the small sample size may limit generalizability. However, the large effect size and consistent cost difference across patients strengthen our conclusions. Additionally, laboratory data regarding kidney function, including estimated glomerular filtration rate (eGFR), were not consistently available, and patient adherence to conservative measures such as dietary sodium restriction, high fluid intake, and potassium citrate use could not be objectively assessed, which may influence recurrence patterns and overall cost outcomes. Future multicenter studies incorporating detailed renal function monitoring and adherence metrics, as well as broader societal perspectives, may provide a more comprehensive economic assessment.
Conclusion
This retrospective study demonstrates that the cost of surgical management in cystinuric patients who require operative intervention is significantly higher than the potential cost of tiopronin therapy. While not all patients may benefit equally from medical management, enhancing access to tiopronin—particularly for those with recurrent stones—could reduce the frequency of invasive procedures and alleviate the economic burden on both families and the healthcare system. These findings support policy initiatives aimed at reintroducing tiopronin into the Turkish market for the benefit of selected cystinuric patients who are most likely to require repeated surgical treatment.
Ethical approval
The study was approved by Ege University Ethical Board of Medical Research (date: 17.04.2025 number: 25-4T/110).
Source of funding
The authors declare the study received no funding.
Conflict of interest
The authors declare that there is no conflict of interest.
References
- Moussa M, Papatsoris AG, Abou Chakra M, Moussa Y. Update on cystine stones: current and future concepts in treatment. Intractable Rare Dis Res 2020; 9: 71-78. https://doi.org/10.5582/irdr.2020.03006
- Servais A, Thomas K, Dello Strologo L, et al. Cystinuria: clinical practice recommendation. Kidney Int 2021; 99: 48-58. https://doi.org/10.1016/j.kint.2020.06.035
- Hasanoglu A, Tumer L, Biberoglu G, Ezgu FS. Ten years experience of a metabolic investigation laboratory in Turkey. Gazi Medical Journal 2000; 11: 77-80.
- Joly D, Rieu P, Méjean A, Gagnadoux MF, Daudon M, Jungers P. Treatment of cystinuria. Pediatr Nephrol 1999; 13: 945-950. https://doi.org/10.1007/s004670050736
- Knoll T, Zöllner A, Wendt-Nordahl G, Michel MS, Alken P. Cystinuria in childhood and adolescence: recommendations for diagnosis, treatment, and follow-up. Pediatr Nephrol 2005; 20: 19-24. https://doi.org/10.1007/s00467-004-1663-1
- European Association of Urology (EAU). EAU guidelines on Urolithiasis. Arnhem, The Netherlands: European Association of Urology Guidelines Office; 2025. Available at: https://uroweb.org/guidelines/urolithiasis
- Bhatt NP, Deshpande AV, Starkey MR. Pharmacological interventions for the management of cystinuria: a systematic review. J Nephrol 2024; 37: 293-308. https://doi.org/10.1007/s40620-023-01795-6
- Elmacı AM, Ece A, Akın F. Pediatric urolithiasis: metabolic risk factors and follow-up results in a Turkish region with endemic stone disease. Urolithiasis 2014; 42: 421-426. https://doi.org/10.1007/s00240-014-0682-z
- Asi T, Dogan HS, Bozaci AC, Citamak B, Altan M, Tekgul S. A single center’s experience in pediatric cystine stone disease management: what changed over time? Urolithiasis 2020; 48: 493-499. https://doi.org/10.1007/s00240-020-01200-y
- Tokhmafshan F, Dickinson K, Akpa MM, Brasell E, Huertas P, Goodyer PR. A no-nonsense approach to hereditary kidney disease. Pediatr Nephrol 2020; 35: 2031-2042. https://doi.org/10.1007/s00467-019-04394-5
- Gökçe Mİ, Karaburun MC. Cystinuria in children: diagnosis and treatment. World J Urol 2025; 43: 226. https://doi.org/10.1007/s00345-025-05604-6
- Melessen IM, Henderickx MM, Merkx MM, van Etten-Jamaludin FS, Homan van der Heide JJ, Kamphuis GM. The effect of additional drug therapy as metaphylaxis in patients with cystinuria: a systematic review. Minerva Urol Nefrol 2020; 72: 427-440. https://doi.org/10.23736/S0393-2249.20.03704-2
License
Copyright (c) 2026 The Author(s). This is an open access article distributed under the Creative Commons Attribution License (CC BY), which permits unrestricted use, distribution, and reproduction in any medium or format, provided the original work is properly cited.