Graphical Abstract
Abstract
Introduction. Musculoskeletal (MSK) complaints are common in children, with many cases ranging from benign to serious conditions. While these patients are often referred to pediatric rheumatologists, only about half are diagnosed with rheumatic diseases. The objective of this study is to identify key clues that assist in the differential diagnosis of patients presenting with joint complaints at pediatric rheumatology outpatient clinic.
Material Methods. In this one-year study, patients with joint pain were assessed using a standardized form. Demographic, clinical, and examination data were collected and anonymized. Initial assessments were done by pediatric rheumatology fellows or residents, with final diagnoses confirmed by a pediatric rheumatologist.
Results. This study included 414 patients aged 0-18 years with joint pain, of whom 273 were diagnosed with rheumatologic conditions and 141 with non-rheumatologic conditions. Patients with rheumatologic conditions had significantly higher laboratory values, including leukocyte counts, neutrophils, platelets, C-reactive protein, and erythrocyte sedimentation rate, all with p < 0.001. Multivariable analysis revealed that arthritis (adjusted odds ratio [aOR] 2.63, 95% confidence interval [CI] 1.09-6.33) and rash (aOR 4.37, 95% CI 1.38-13.86) predicted rheumatic disease in acute presentations, while in chronic complaints arthritis (aOR 2.61, 95% CI 1.30-5.21), morning stiffness (aOR 3.47, 95% CI 1.69-7.11), migratory pain (aOR 3.45, 95% CI 1.01-11.80), and fever (aOR 12.89, 95% CI 4.41-37.68) were independent predictors, whereas myalgia was associated with non-rheumatic conditions (aOR 0.35, 95% CI 0.15-0.83).
Conclusion. In conclusion, this study emphasized the importance of clinical clues in diagnosing rheumatic diseases in children with joint pain. An accurate diagnosis depends on a thorough history and physical examination. Improving the differential diagnosis of joint pain is essential to reduce unnecessary referrals and enhance the efficiency of healthcare services.
Keywords: arthralgia, arthritis, joint complaints, pediatric rheumatology, rheumatic disease
Introduction
Musculoskeletal (MSK) complaints constitute approximately 10-20% of pediatric presentations in primary care clinics.1 Studies indicate that around 50% of children and adolescents experience MSK complaints at least once in their lifetime.1 The underlying causes of these complaints are highly diverse, spanning from benign muscular issues to rheumatic diseases or even malignancies. Key clinical features, including constitutional symptoms such as fever and fatigue, the nature and anatomical location of pain, the duration of morning stiffness, and abnormal findings on physical examination, can aid in identifying the underlying etiology. Certain “red flags” also assist clinicians in the differential diagnosis. Many patients presenting with joint pain, fever of unknown origin, elevated acute phase reactants (APRs), or positive anti-nuclear antibody (ANA) are often referred to pediatric rheumatologists. However, the underlying causes frequently include infectious, orthopedic, or traumatic conditions, which can be managed by primary care pediatricians.2 Studies have shown that only about half of patients referred to rheumatology clinics ultimately receive a rheumatic disease diagnosis.3-5
Previous studies have demonstrated that isolated musculoskeletal pain has poor predictive value for rheumatic disease, as most children with chronic arthritis present instead with joint swelling or gait abnormalities.6 Additionally, multiple cohorts have reported that only 40%-55% of children referred to pediatric rheumatology ultimately receive a rheumatic diagnosis, contributing to significant over-referral and unnecessary specialist burden.4,5 Although several studies have attempted to identify predictors of juvenile idiopathic arthritis (JIA) or chronic arthritis, these models were developed in selected populations and have not been validated in heterogeneous groups of children presenting with any type of MSK complaint.7,8 Despite the large number of referrals, there is limited evidence identifying which specific clinical and laboratory features most reliably distinguish rheumatic from non-rheumatic causes of joint pain in children. Existing decision-support tools, such as SimulConsult, show potential but remain underutilized in daily clinical practice, emphasizing the need for real-life data to guide diagnostic frameworks.2
Therefore, the primary objective of this study was to identify clinical and laboratory predictors that distinguish rheumatic from non-rheumatic conditions in children presenting with joint complaints. We hypothesized that specific features—particularly arthritis, rash, morning stiffness, and fever—would strongly predict rheumatic disease in both acute and chronic presentations. This study aimed to analyze data from children with joint complaints presenting to a pediatric rheumatology outpatient clinic over a one-year period, in order to assist clinicians in evaluating these patients. This approach will provide a practical method for pediatricians working in emergency or outpatient settings to use in the differential diagnosis of patients presenting with joint pain.
Materials and Methods
Study population
In this one-year observational study, all patients presenting with joint pain to the pediatric rheumatology outpatient clinic were systematically evaluated using a standardized assessment form.
Demographic data, including age, gender, consanguinity, and family history of rheumatic diseases, were meticulously collected as part of the patient evaluation. Clinical features, such as the nature, duration, and severity of joint pain, the presence of systemic symptoms (e.g., fever, fatigue), and findings from the physical examination (e.g., joint swelling, range of motion), were also systematically documented. All data were anonymized to ensure confidentiality, with each patient assigned a unique identification code by the attending physician. This approach ensured that personal identifiers were not linked to clinical information during data analysis. In addition, the specialty of the referring physician, whether from pediatrics, general practice, or another medical field, was carefully recorded to assess potential patterns or referral biases in the patient cohort.
All patients aged 0-18 years who presented with joint complaints to the pediatric rheumatology outpatient clinic between May 2023 and May 2024 were included in the study. Patients were followed until a final diagnosis could be confirmed; the duration of follow-up ranged from a single visit to several months, depending on the clinical context. Patients with incomplete records or lacking a confirmed diagnosis at the end of follow-up were excluded from the comparative analyses. Children were initially assessed by a fellow of pediatric rheumatology fellow or a pediatric resident and subsequently underwent a comprehensive evaluation by a pediatric rheumatologist prior to the establishment of the final diagnosis. Final diagnoses were independently verified and documented by a pediatric rheumatology specialist. The diagnosis of the patients was made based on the classification criteria for each rheumatologic condition. The classification criteria are described in the following sentences.
Acute joint complaints were defined as symptoms that lasted less than 6 weeks, while chronic joint pain is defined was symptoms persisting for more than 6 weeks.
International classification criteria were used for patient classification. The diagnosis of JIA was based on the International League of Associations for Rheumatology (ILAR) classification criteria9; autoinflammatory recurrent fevers were classified according to the Eurofever/Pediatric Rheumatology International Trials Organisation (PRINTO) clinical criteria.10,11 Diagnoses of vasculitis followed the European League Against Rheumatism (EULAR)/PRINTO/Pediatric Rheumatology European Society (PRES) criteria12; Behçet’s disease (BD) was classified according to the Pediatric BD Consensus criteria.13 Systemic lupus erythematosus (SLE) diagnoses followed the Systemic Lupus International Collaborating Clinics (SLICC) criteria14; juvenile dermatomyositis was classified using the Bohan and Peter criteria.15,16 Juvenile systemic sclerosis was diagnosed according to the PRES/American College of Rheumatology (ACR)/EULAR criteria17; juvenile localized scleroderma followed widely used recommended classification criteria18, fibromyalgia diagnosis was based on the Budapest Pain Amplification Syndrome criteria19, Raynaud’s phenomenon diagnosis used the International Consensus Criteria 20, and acute rheumatic fever (ARF) was diagnosed based on the revised Jones Criteria.21 Psoriasis, inflammatory bowel disease, and isolated uveitis were categorized under “rheumatologic disease” only because these patients were referred to pediatric rheumatology and assessed within the same clinical pathway.
Patients lacking clinical or laboratory indicators consistent with a rheumatic disease were classified under non-rheumatic conditions. For patients identified with non-rheumatologic conditions, diagnoses were corroborated and recorded by specialists from the relevant medical departments.
Statistical methods
All statistical analyses were performed using SPSS version 29.0 (IBM Corp., Armonk, NY, USA) and R 4.3.0 programs. The Kolmogorov-Smirnov test was applied to assess the assumption of normality. Continuous variables were reported as medians and interquartile ranges (IQRs) since the normality assumption did not hold. Categorical variables were reported as counts and percentages. Comparisons of continuous variables between rheumatic and non-rheumatic groups were performed using Mann-Whitney U test. Associations between categorical variables were examined using Chi-square test.
Multiple logistic regression analysis was performed to identify risk factors for rheumatic disease. Variables found to be statistically significant in the univariate analyses were first selected as candidate variables, then entered into the least absolute shrinkage and selection operator (LASSO) logistic regression analysis for feature selection. With the parameters selected by LASSO regression, a multiple logistic regression analysis was performed to calculate adjusted odds ratios (aORs) and 95% confidence intervals (CIs). A p-value <0.05 was considered statistically significant. The Sankey diagram was constructed to visualize how patients transition through these key clinical and laboratory features and how different combinations of findings contribute to the final classification.
Results
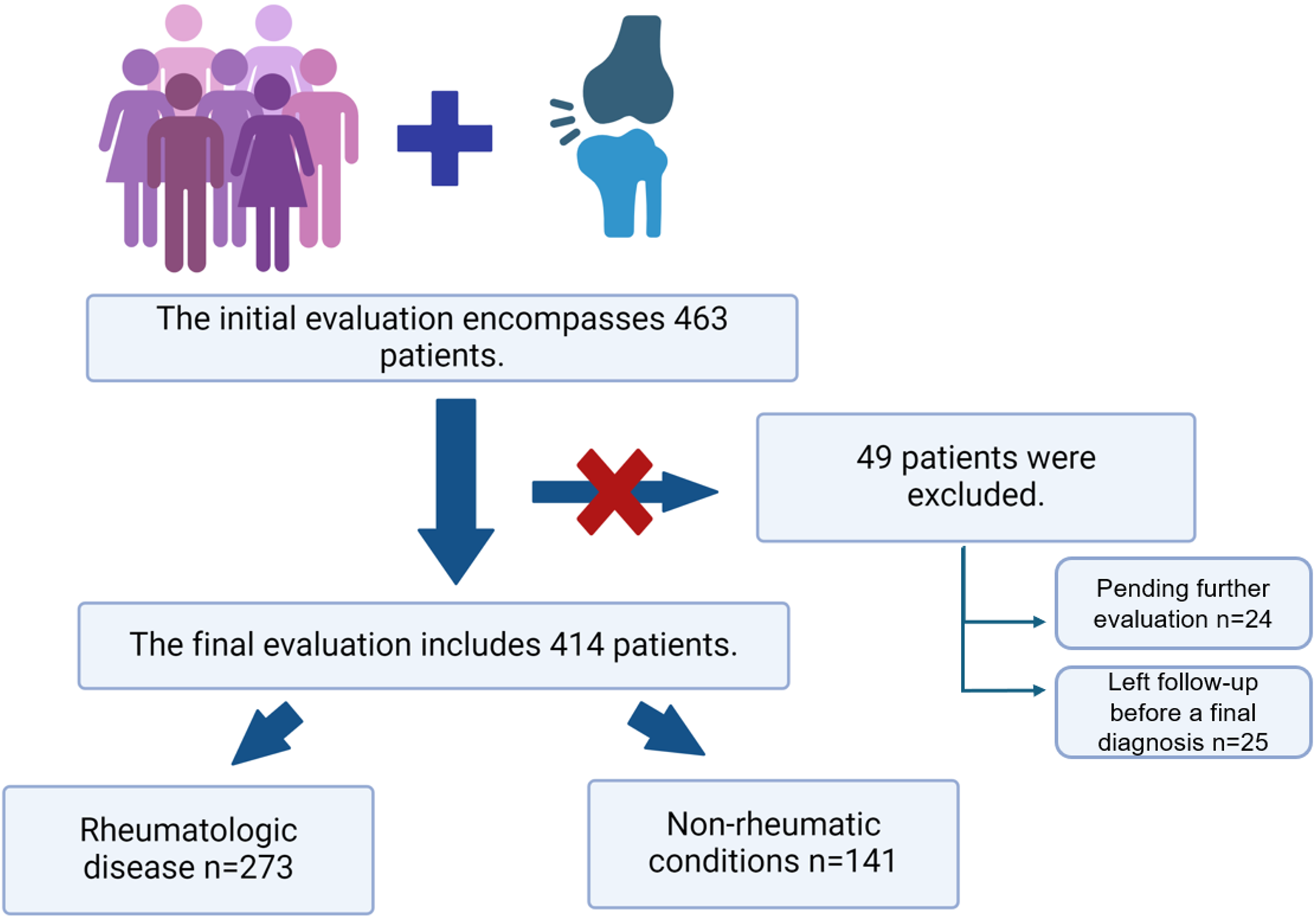
A total of 463 patients aged 0-18 years were included in this prospective study. These patients presented to the pediatric rheumatology clinic for the first time with joint pain between May 2023 and May 2024. Of these patients, 59% (n=273) received a rheumatologic diagnosis, while 30.5% (n=141) were diagnosed with non-rheumatologic conditions. The final diagnoses of patients are depicted in Supplementary Table S1. The diagnoses for 5.2% (n=24) of patients were pending further evaluation, and 5.3% (n=25) were lost to follow-up before a final diagnosis was made (Fig. 1). Final analyses were conducted exclusively in the 414 patients with definitive diagnoses.
The initial presentation location differed significantly between the groups: 30% of patients (n=124) initially presented to the emergency department, while 70% (n=290) attended an outpatient clinic. Emergency department visits were more frequent among patients with rheumatologic conditions (n=98, 35.9%) compared to those with non-rheumatologic conditions (n=26, 18.4%; p < 0.001).
Detailed demographic, clinical, and laboratory data were analyzed for the 414 patients with a definitive diagnosis. These patients were classified as having either rheumatologic or non-rheumatologic conditions. Clinical and laboratory characteristics were compared between these groups (Table I). To further analyze acute and chronic joint pain, clinical features were compared between groups based on the duration of joint pain (Table II).
| All variables except for age were categorical and presented as n (%). Age was presented as median (IQR). For comparisons in rheumatic vs. non-rheumatic diseases, age was compared using the Mann-Whitney U test, and categorical variables using the chi-square test. The "All patients" column provides descriptive data and was not included in the statistical analysis. IQR, interquartile range. | ||||
| Table I. Comparison of demographic and clinical features between children diagnosed with rheumatic diseases and those with non-rheumatic musculoskeletal conditions | ||||
| Demographic and clinical features |
(n=414) |
|
|
|
| Age, median (IQR), years |
|
|
|
|
| Sex (female/male) |
|
|
|
|
| Family history |
|
|
|
|
| Consanguinity |
|
|
|
|
| Arthralgia |
|
|
|
|
| Monoarthralgia |
|
|
|
|
| Oligoarthralgia |
|
|
|
|
| Polyarthralgia |
|
|
|
|
| Arthritis |
|
|
|
|
| Monoarthritis |
|
|
|
|
| Oligoarthritis |
|
|
|
|
| Polyarthritis |
|
|
|
|
| Redness of the joint |
|
|
|
|
| Warmth |
|
|
|
|
| Morning stiffness |
|
|
|
|
|
Duration of morning stiffness <30 min / >30 min |
|
|
|
|
| Migratory pain |
|
|
|
|
| Acute joint complaints |
|
|
|
|
| Chronic joint complaints |
|
|
|
|
| Large joint |
|
|
|
|
| Shoulder |
|
|
|
|
| Elbow |
|
|
|
|
| Wrist |
|
|
|
|
| Hip |
|
|
|
|
| Knee |
|
|
|
|
| Ankle |
|
|
|
|
| Axial involvement |
|
|
|
|
| Neck |
|
|
|
|
| Lower back |
|
|
|
|
| Back |
|
|
|
|
| Small joint |
|
|
|
|
| Heel pain |
|
|
|
|
| Sacroiliitis |
|
|
|
|
| Fever |
|
|
|
|
| Weight loss |
|
|
|
|
| Night sweats |
|
|
|
|
| Abdominal pain |
|
|
|
|
| Myalgia |
|
|
|
|
| Rash |
|
|
|
|
| All variables except for age were categorical and presented as n (%). Age was presented as median (IQR). For comparison in acute rheumatic vs. acute non-rheumatic and chronic rheumatic vs. chronic non-rheumatic diseases, age was compared using the Mann-Whitney U test, and categorical variables using the chi-square test . IQR, interquartile range. | ||||||
| Table II. Comparison of demographic and clinical features between patients with rheumatic and non-rheumatic joint complaints, stratified by symptom duration | ||||||
|
|
|
|||||
|
|
|
|
|
|
|
|
| Age (years), median (IQR) |
|
|
|
|
|
|
| Female sex |
|
|
|
|
|
|
| Family history |
|
|
|
|
|
|
| Consanguinity |
|
|
|
|
|
|
| Arthralgia |
|
|
|
|
|
|
| Monoarthralgia |
|
|
|
|
|
|
| Oligoarthralgia |
|
|
|
|
||
| Polyarthralgia |
|
|
|
|
||
| Arthritis |
|
|
|
|
|
|
| Monoarthritis |
|
|
|
|
|
|
| Oligoarthritis |
|
|
|
|
||
| Polyarthritis |
|
|
|
|
||
| Redness in the joint |
|
|
|
|
|
|
| Warmth |
|
|
|
|
|
|
| Morning stiffness |
|
|
|
|
|
|
| Duration of morning stiffness >30 min |
|
|
|
|
|
|
| Migratory pain |
|
|
|
|
|
|
| Large joint |
|
|
|
|
|
|
| Shoulder |
|
|
|
|
|
|
| Elbow |
|
|
|
|
|
|
| Wrist |
|
|
|
|
|
|
| Hip |
|
|
|
|
|
|
| Knee |
|
|
|
|
|
|
| Ankle |
|
|
|
|
|
|
| Axial involvement |
|
|
|
|
|
|
| Neck |
|
|
|
|
|
|
| Lower back |
|
|
|
|
|
|
| Back |
|
|
|
|
|
|
| Small joint |
|
|
|
|
|
|
| Heel pain |
|
|
|
|
|
|
| Sacroiliitis |
|
|
|
|
|
|
| Fever |
|
|
|
|
|
|
| Weight loss |
|
|
|
|
|
|
| Night sweats |
|
|
|
|
|
|
| Abdominal pain |
|
|
|
|
|
|
| Myalgia |
|
|
|
|
|
|
| Rash |
|
|
|
|
|
|
Among all patients, 334 (80.7%) had no rash. Rash was present in 80 (19.3%) of all patients, with a significantly higher frequency in the rheumatic group (70/273, 25.6%) compared to the non-rheumatic group (10/141, 7.1%; p <0.001). Vasculitic rashes such as purpura (n=29, 7%) and palpable purpura (n=22, 5.3%) were observed predominantly in the rheumatic group (purpura: 9.5%, palpable purpura: 8.1%). Maculopapular rashes were seen in 18 patients (4.3%), and urticaria in 8 patients (1.9%). Erysipelas-like erythema (ELE) was observed in 3 patients (0.7%), all within the rheumatic group. Notably, palpable purpura and ELE were absent in the non-rheumatic group, whereas urticaria, purpura, and maculopapular rashes were present in only a few non-rheumatic patients (each n≤4).
Swollen joints were observed in 73.3% (n=113) of patients, with swelling, warmth (25.9%, n=40), and redness (24.6%, n=38) occurring more frequently in the rheumatologic group (p=0.001). Morning stiffness lasting over 30 minutes was reported by 30 patients with a diagnosis of rheumatic disease, which was higher compared to the non-rheumatologic cases, but did not reach statistical significance. Large joint involvement was significantly higher in the rheumatologic disease group (96.3% vs. 85.1%, p<0.001), and ankle involvement was also significantly more frequent in this group (56% vs. 45.4%, p=0.048). Axial joint pain was more common in the non-rheumatologic disease group (19.1% vs. 10.6%, p=0.024), as was lower back pain (15.6% vs. 6.2%, p=0.004). Heel pain was significantly more prevalent in patients with non-rheumatologic diseases (11.3% vs. 2.9%, p=0.001). Sacroiliitis was significantly more common in the rheumatologic disease group (5.1% vs. 0%, p=0.003). No significant association was found between disease groups and small joint involvement. Fever was significantly more common in patients with rheumatologic diseases compared to those with non-rheumatologic diseases (28.6% vs. 10.6%, p<0.001). Abdominal pain was also significantly more frequent in patients with rheumatologic diseases compared to those with non-rheumatologic diseases (29.7% vs. 8.5%, p<0.001).
Analysis of laboratory values revealed significant differences between groups. The median leukocyte count was higher in rheumatologic patients than in non-rheumatologic patients (8970/mm³, IQR: 6850-28650 vs. 7740/mm³, IQR: 6580-9860, p<0.001). Similarly, median neutrophil counts (4700/mm³, IQR: 3540-5240 vs. 3780/mm³, IQR: 1560-6430, p<0.001) and platelet counts (350,000/mm³, IQR: 228,000-442,000 vs. 311,000/mm³, IQR: 154,000-480,000, p<0.001) were significantly higher in the rheumatologic group. Inflammatory markers were also elevated, with higher median CRP levels (5 mg/dL, IQR: 3.5-7.5 vs. 1.15 mg/dL, IQR: 0.6-2, p<0.001) and ESR values (17 mm/h, IQR: 10-26 vs. 10 mm/h, IQR: 6-20, p<0.001) compared with non-rheumatologic patients.
When clinical features were compared based on the duration of joint complaints, oligoarthritis (42.1% vs. 6.9%, p=0.001), myalgia (24% vs. 3.4%, p=0.026), and rash (49.6% vs. 13.8%, p=0.001) were more common in patients with a diagnosis of rheumatic disease in the presence of acute joint pain. Arthritis (28.9% vs. 17%, p=0.028), morning stiffness (27% vs. 14.3%, p=0.02), migratory pain (15.1% vs. 3.6%, p=0.004), large joint involvement (94.1% vs. 85.7%, p=0.037), and sacroiliitis (36.8% vs. 9.8%, p<0.001) were more common in patients with a diagnosis of rheumatic disease in the presence of chronic joint pain.
Development of algorithms to facilitate diagnosis
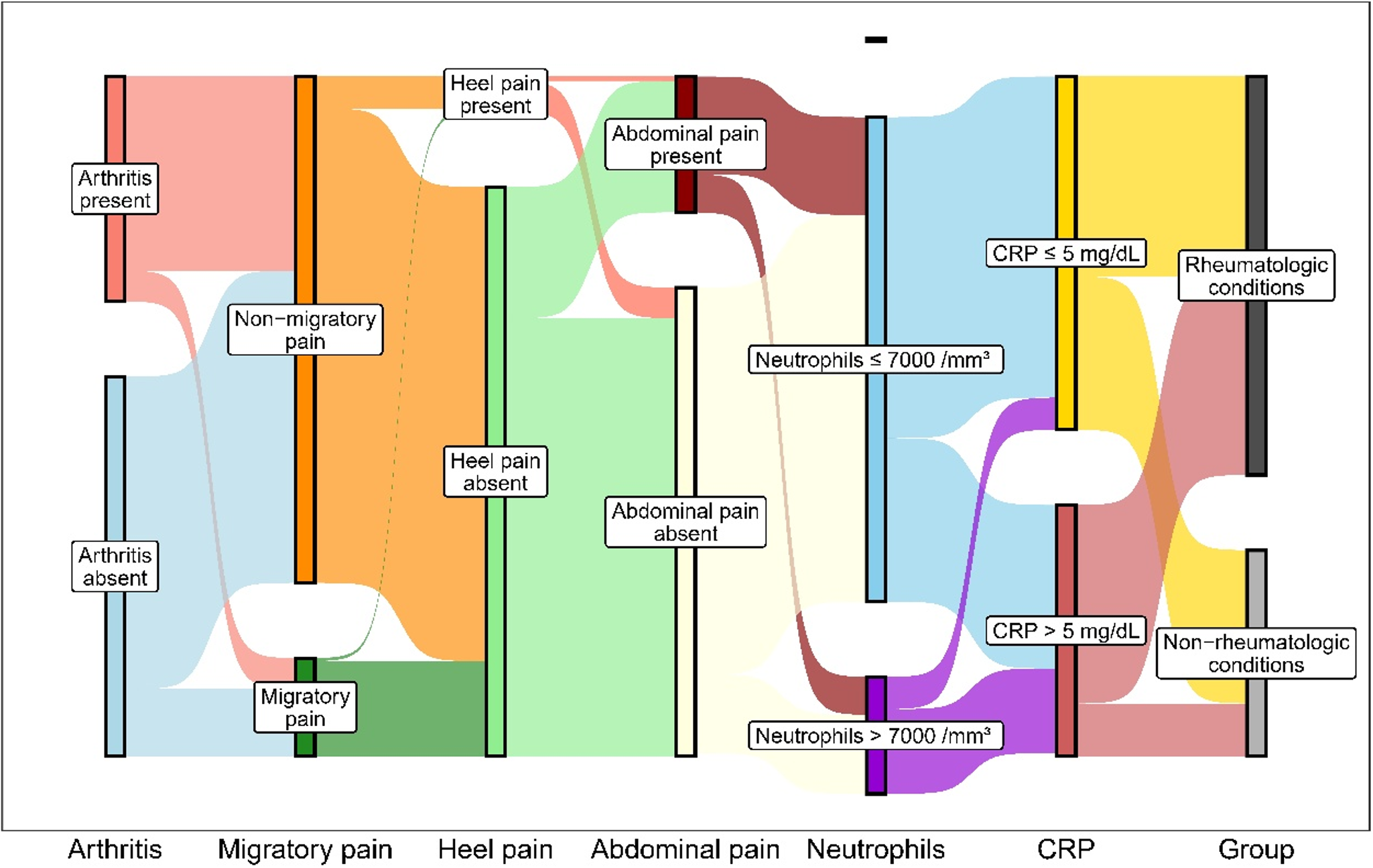
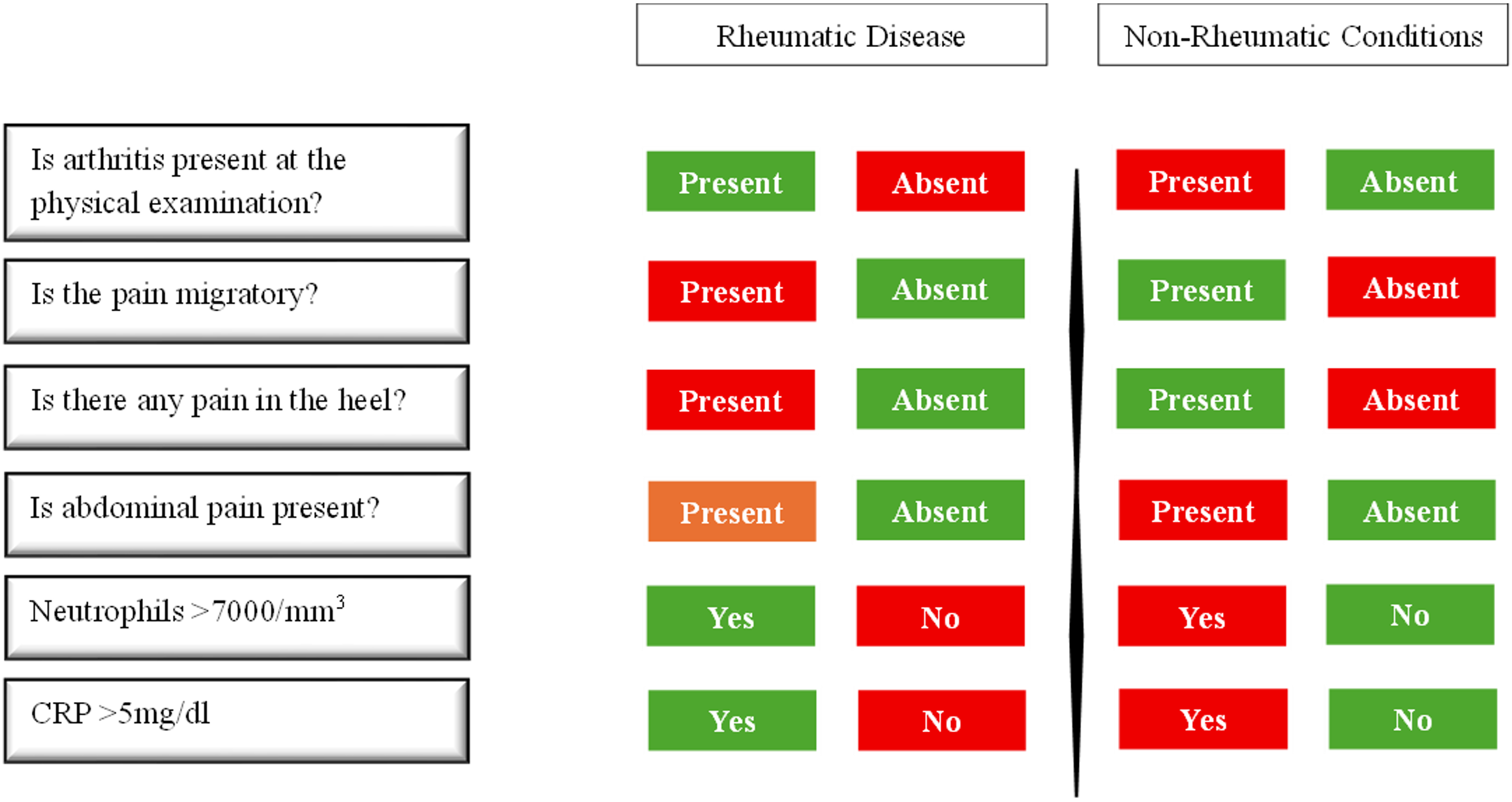
To determine the key factors involved in diagnosing rheumatic diseases, all clinical and laboratory data were initially subjected to univariate analysis. Parameters found to be statistically significant in the univariate analysis were subsequently evaluated using multivariate analysis to identify independent predictors. In LASSO logistic regression analysis, the presence of arthritis (aOR 2.63, 95% CI 1.09-6.33, p=0.031) and rash (aOR 4.37, 95% CI 1.38-13.86, p=0.012) were independently associated with the diagnosis of rheumatic disease in patients with acute joint complaints. In patients with chronic joint complaints, the presence of arthritis (aOR 2.61, 95% CI 1.30-5.21, p=0.007), morning stiffness (aOR 3.47, 95% CI 1.69-7.11, p=0.001), migratory pain (aOR 3.45, 95% CI 1.01-11.80, p=0.049), and fever (aOR 12.89, 95% CI 4.41-37.68, p<0.001) were independently associated with the diagnosis of rheumatic disease. Furthermore, the presence of myalgia was independently associated with the diagnosis of non-rheumatic conditions (aOR 0.35, 95% CI 0.15-0.83, p=0.017; Table III). Fig. 2 presents a Sankey diagram illustrating how key clinical and laboratory features are distributed across the final diagnostic categories. Each vertical axis represents a diagnostic predictor (arthritis, migratory pain, heel pain, abdominal pain, neutrophils, and CRP), and the width of each connecting band indicates the proportion of patients exhibiting that feature in the rheumatic and non-rheumatic groups. This visualization highlights the strongest discriminative features—such as the presence of arthritis, migratory pain, elevated neutrophils, and elevated CRP—and provides a graphical overview of how these parameters jointly guide diagnostic classification. Based on the results of the Sankey analysis, a diagnostic decision chart (Fig. 3) was constructed to show the relative frequency of each feature in rheumatic versus non-rheumatic conditions.
|
Multivariable analysis was performed using the least absolute shrinkage and selection operator (LASSO) method. Odds ratios (OR) and adjusted odds ratios (aOR) are presented with 95% confidence intervals (CI). Variables with statistically significant associations in univariable analysis were included in the LASSO model to identify the most predictive features for rheumatic diagnosis. Separate models were constructed for acute and chronic subgroups. |
||||
| Table III. Univariable and multivariable logistic regression analysis results for predictors of rheumatic disease among children with musculoskeletal complaints, stratified by acute and chronic symptom duration | ||||
|
|
|
|||
|
|
|
|
|
|
| Acute joint complaints | ||||
| Arthritis |
|
|
|
|
| Myalgia |
|
|
|
|
| Rash |
|
|
|
|
| Chronic joint complaints | ||||
| Arthritis |
|
|
|
|
| Morning stiffness |
|
|
|
|
| Migratory pain |
|
|
|
|
| Large joint |
|
|
|
|
| Fever |
|
|
|
|
| Abdominal pain |
|
|
|
|
| Myalgia |
|
|
|
|
Discussion
This study evaluated 414 children aged 0-18 years who were referred for an initial pediatric rheumatology assessment due to joint pain, aiming to identify clinical features predictive of rheumatic diseases through LASSO logistic regression analysis. The model demonstrated that distinct clinical indicators were associated with rheumatic diagnoses in acute versus chronic presentations. The analysis revealed that arthritis and rash were the most informative features in acute presentations, whereas in chronic cases, a combination of arthritis, morning stiffness, migratory joint pain, and fever showed strong associations with rheumatic diagnoses.
The Sankey diagram in our study provides a clinically meaningful visualization of how individual clinical features are distributed across rheumatic diseases and non-rheumatic conditions, highlighting the relative diagnostic weight of key predictors such as arthritis, migratory pain, neutrophilia, and elevated CRP. By mapping these feature patterns, the diagram helps clinicians focus on the most informative clinical findings during the initial assessment of joint complaints. Complementing this, Fig. 3 provides a simplified visual comparison of the presence or absence of these predictors, enabling rapid estimation of rheumatic disease likelihood in routine clinical practice. The prevalence of pediatric rheumatic diseases varies across geographical regions. For example, familial Mediterranean fever (FMF) is more prevalent in countries within the Mediterranean region, BD is commonly observed along the historical Silk Road, and SLE occurs more frequently in African-American populations.5,22 A previous study from our country reported that 52% of all first referrals to the pediatric rheumatology department were diagnosed with a rheumatic disease, while 42% had non-rheumatic conditions. Among rheumatic diseases, FMF was the most common diagnosis, followed by JIA and vasculitis. In contrast, orthopedic or mechanical problems (24.7%) were the most frequent non-rheumatic conditions.5 Although our study differed from previous ones by including only children with joint pain, the most common diagnoses among rheumatologic diseases were still FMF and JIA, while orthopedic and mechanical causes were the most frequent non-rheumatologic conditions. Fever and abdominal pain, prominent features of FMF, were also significantly more common in patients with rheumatologic diseases in our cohort.
Pediatric rheumatology referrals are most often driven by joint pain or swelling, abnormal laboratory results such as elevated APRs and positive ANA, and unexplained fevers. However, these findings may suggest a wide range of underlying diseases, including infectious, genetic, hematologic, or orthopedic conditions.6 Despite substantial advancements in laboratory diagnostics, the cornerstone of the diagnostic process continues to be a comprehensive medical history and a detailed physical examination. Given the frequent occurrence of joint pain in pediatric populations, it is crucial to carefully consider the duration and temporal pattern of symptom onset in the diagnostic assessment. For instance, pain that exacerbates with activity and progresses throughout the day, coupled with worsening swelling, should raise suspicion for a mechanical etiology. Algorithms that assist clinicians in making differential diagnoses can help reduce unnecessary referrals. Pilot studies have been conducted on this topic. For instance, Segal et al.2 developed decision support software aimed at minimizing diagnostic errors in pediatric rheumatology practice (available at www.simulconsult.com). SimulConsult has expanded the diagnostic scope of complex cases, increased the success rate of early-stage diagnoses, and achieved results comparable to those of traditional assessments performed by experienced pediatricians.2 Len et al.23 proposed a questionnaire designed to facilitate the early identification of patients suitable for referral to pediatric rheumatology centers. Their 12-item score identified candidates for referral based on symptom patterns, with a cutoff score of 5. Structured tools such as SimulConsult or the 12-item referral questionnaire proposed by Len et al. offer algorithmic or score-based support for triage decisions.2,23 However, our study complements these tools by identifying diagnostic clues grounded in real-world observational data, such as the presence of morning stiffness or oligoarthritis in patients with chronic versus acute symptoms. Apart from such approaches, studies based on real-life patient data can assist clinicians in guiding the diagnostic process. There are a limited number of studies conducted with this objective. Cattalini et al.8 developed a predictive score for chronic arthritis in a cohort of children presenting with MSK complaints, achieving a sensitivity of 90.9% and a specificity of 95.3%. It incorporated clinical variables such as prolonged morning stiffness, non-mechanical pain, and limping. According to the study, recurrent pain occurring more than once a month was strongly associated with noninflammatory disorders, with evening/night pain more common in these patients. Pain precipitating factors differed by category: rest in 68% of chronic arthritis patients, prior infection in 79% of infection-related arthritis cases, and activity in 46% of noninflammatory disorder patients. Jeamsripong and Charuvanij7 investigated clinical findings that could predict the diagnosis of JIA in children presenting with MSK complaints. They found that morning stiffness lasting longer than 15 minutes, joint swelling on MSK examination, a duration of MSK complaints exceeding 6 weeks, and limping were significantly associated with the final diagnosis of JIA. In contrast, isolated joint pain—commonly reported—was not found to be predictive of JIA. Another study aimed at identifying specific complaints indicative of systemic inflammatory disease found that isolated pain without other symptoms rarely suggests chronic arthritis.6 Children with arthritis typically presented with joint swelling or gait issues. ANA and rheumatoid factor (RF) tests were not useful for evaluating musculoskeletal complaints.6 In our study, due to the heterogeneous nature of the referring group, patients were analyzed both as a whole and stratified by the duration of their symptoms. Although our primary objective was to differentiate rheumatic from non-rheumatic conditions, we additionally performed subgroup comparisons based on the chronicity of symptoms to explore whether certain diagnostic clues differ in acute versus chronic presentations. This approach may aid clinicians in early triage decisions. Specifically, in patients with acute joint complaints (<6 weeks), the presence of oligoarthritis and a rash were highly indicative of a rheumatic etiology. One of the most frequent diagnoses in this subgroup was IgA vasculitis, formerly known as Henoch-Schönlein purpura (HSP), which supports this hypothesis. Detailed analysis of rash subtypes showed that vasculitic rashes, particularly purpuric and palpable purpura, were almost exclusively observed in the rheumatic group. Conversely, non-specific rashes such as maculopapular exanthems or urticaria were uncommon and appeared in both groups. These results suggest that the predictive value of rash in our model mainly depends on vasculitic presentations, consistent with diagnoses like HSP. Conversely, in patients presenting with chronic joint complaints (>6 weeks), the presence of arthritis, morning stiffness, large joint involvement, migratory pain, and sacroiliitis were significant clinical features that supported the diagnosis of a rheumatic disease. JIA, the most common cause of chronic arthritis in children, is particularly associated with morning stiffness. Thus, morning stiffness in the context of chronic complaints may serve as a key diagnostic clue for JIA.
The main limitation of our study is its single-center design; however, its significant strength lies in the prospective evaluation of all patients using a standardized form and deriving clinical insights based on definitive diagnoses. Although all diagnoses were confirmed by an experienced pediatric rheumatologist based on the international classification criteria for each rheumatologic disease, we did not formally assess inter-rater variability, which represents a methodological limitation.
In conclusion, this study aimed to highlight clinical clues for diagnosing rheumatic diseases in children with joint pain. Accurate diagnosis relies heavily on a detailed history and physical examination. Enhancing the clues for the differential diagnosis of joint pain is crucial to reduce unnecessary referrals and help the healthcare system provide more effective services.
Supplementary materials
Supplementary materials for this article are available online at https://doi.org/10.24953/turkjpediatr.2026.6305.
Ethical approval
The study was approved by Kocaeli University Ethics Committee (date: 13.07.2023, number: 2023-170).
Source of funding
The authors declare the study received no funding.
Conflict of interest
The authors declare that there is no conflict of interest.
References
- Tallen G, Bielack S, Henze G, et al. Musculoskeletal pain: a new algorithm for differential diagnosis of a cardinal symptom in pediatrics. Klin Padiatr 2014; 226: 86-98. https://doi.org/10.1055/s-0034-1366989
- Segal MM, Athreya B, Son MB, et al. Evidence-based decision support for pediatric rheumatology reduces diagnostic errors. Pediatr Rheumatol Online J 2016; 14: 67. https://doi.org/10.1186/s12969-016-0127-z
- Hashkes PJ. Profile of a pediatric rheumatology practice in Israel. Clin Exp Rheumatol 2003; 21: 123-128.
- Rosenberg AM. Longitudinal analysis of a pediatric rheumatology clinic population. J Rheumatol 2005; 32: 1992-2001.
- Karadağ ŞG, Sönmez HE, Tanatar A, Çakmak F, Çakan M, Ayaz NA. Profile of new referrals to a single pediatric rheumatology center in Turkey. Rheumatol Int 2020; 40: 313-321. https://doi.org/10.1007/s00296-019-04421-6
- McGhee JL, Burks FN, Sheckels JL, Jarvis JN. Identifying children with chronic arthritis based on chief complaints: absence of predictive value for musculoskeletal pain as an indicator of rheumatic disease in children. Pediatrics 2002; 110: 354-359. https://doi.org/10.1542/peds.110.2.354
- Jeamsripong S, Charuvanij S. Features distinguishing juvenile idiopathic arthritis among children with musculoskeletal complaints. World J Pediatr 2020; 16: 74-81. https://doi.org/10.1007/s12519-018-0212-0
- Cattalini M, Parissenti I, Tononcelli E, Lancini F, Cantarini L, Meini A. Developing a predictive score for chronic arthritis among a cohort of children with musculoskeletal complaints--the chronic arthritis score study. J Pediatr 2016; 169: 188-193. https://doi.org/10.1016/j.jpeds.2015.10.081
- Petty RE, Southwood TR, Manners P, et al. International League of Associations for Rheumatology classification of juvenile idiopathic arthritis: second revision, Edmonton, 2001. J Rheumatol 2004; 31: 390-392.
- Gattorno M, Hofer M, Federici S, et al. Classification criteria for autoinflammatory recurrent fevers. Ann Rheum Dis 2019; 78: 1025-1032. https://doi.org/10.1136/annrheumdis-2019-215048
- Federici S, Sormani MP, Ozen S, et al. Evidence-based provisional clinical classification criteria for autoinflammatory periodic fevers. Ann Rheum Dis 2015; 74: 799-805. https://doi.org/10.1136/annrheumdis-2014-206580
- Ruperto N, Ozen S, Pistorio A, et al. EULAR/PRINTO/PRES criteria for Henoch-Schönlein purpura, childhood polyarteritis nodosa, childhood Wegener granulomatosis and childhood Takayasu arteritis: Ankara 2008. Part I: Overall methodology and clinical characterisation. Ann Rheum Dis 2010; 69: 790-797. https://doi.org/10.1136/ard.2009.116624
- Koné-Paut I, Shahram F, Darce-Bello M, et al. Consensus classification criteria for paediatric Behçet’s disease from a prospective observational cohort: PEDBD. Ann Rheum Dis 2016; 75: 958-964. https://doi.org/10.1136/annrheumdis-2015-208491
- Petri M, Orbai AM, Alarcón GS, et al. Derivation and validation of the Systemic Lupus International Collaborating Clinics classification criteria for systemic lupus erythematosus. Arthritis Rheum 2012; 64: 2677-2686. https://doi.org/10.1002/art.34473
- Bohan A, Peter JB. Polymyositis and dermatomyositis (first of two parts). N Engl J Med 1975; 292: 344-347. https://doi.org/10.1056/NEJM197502132920706
- Bohan A, Peter JB. Polymyositis and dermatomyositis (second of two parts). N Engl J Med 1975; 292: 403-407. https://doi.org/10.1056/NEJM197502202920807
- Zulian F, Woo P, Athreya BH, et al. The Pediatric Rheumatology European Society/American College of Rheumatology/European League against Rheumatism provisional classification criteria for juvenile systemic sclerosis. Arthritis Rheum 2007; 57: 203-212. https://doi.org/10.1002/art.22551
- Laxer RM, Zulian F. Localized scleroderma. Curr Opin Rheumatol 2006; 18: 606-613. https://doi.org/10.1097/01.bor.0000245727.40630.c3
- Harden NR, Bruehl S, Perez RSGM, et al. Validation of proposed diagnostic criteria (the “Budapest Criteria”) for Complex Regional Pain Syndrome. Pain 2010; 150: 268-274. https://doi.org/10.1016/j.pain.2010.04.030
- Maverakis E, Patel F, Kronenberg DG, et al. International consensus criteria for the diagnosis of Raynaud’s phenomenon. J Autoimmun 2014; 48-49: 60-65. https://doi.org/10.1016/j.jaut.2014.01.020
- Gewitz MH, Baltimore RS, Tani LY, et al. Revision of the Jones Criteria for the diagnosis of acute rheumatic fever in the era of Doppler echocardiography: a scientific statement from the American Heart Association. Circulation 2015; 131: 1806-1818. https://doi.org/10.1161/CIR.0000000000000205
- Harry O, Yasin S, Brunner H. Childhood-onset systemic lupus erythematosus: a review and update. J Pediatr 2018; 196: 22-30.e2. https://doi.org/10.1016/j.jpeds.2018.01.045
- Len CA, Terreri MT, Puccini RF, et al. Development of a tool for early referral of children and adolescents with signs and symptoms suggestive of chronic arthropathy to pediatric rheumatology centers. Arthritis Rheum 2006; 55: 373-377. https://doi.org/10.1002/art.21983
License
Copyright (c) 2026 The Author(s). This is an open access article distributed under the Creative Commons Attribution License (CC BY), which permits unrestricted use, distribution, and reproduction in any medium or format, provided the original work is properly cited.