Graphical Abstract
Abstract
Background. In unresectable hepatoblastoma (HB), particularly “pre-treatment extent of tumor” (PRETEXT) IV tumors or those with positive annotation factors, standard management consists of intensive chemotherapy followed by surgical resection or orthotopic liver transplantation (OLT). Radiotherapy has traditionally been avoided because of the liver’s radiosensitivity and the risk of radiation-induced liver disease. Proton beam therapy (PBT), owing to its dosimetric advantage and ability to spare uninvolved liver parenchyma, may represent a potential local control strategy in selected pediatric patients for whom curative surgery or OLT is not feasible.
Case Presentation. We describe five pediatric patients with advanced hepatoblastoma treated with proton beam therapy at our institution between February 2022 and January 2024. The cohort included three girls and two boys, with a median age of 3.0 years (interquartile range [IQR], 1.6–4.0) and a median alpha-fetoprotein level of 435,453 ng/mL (IQR: 7,668–1,276,681) at diagnosis. All patients were initially considered inoperable because of extensive hepatic involvement, inadequate future liver remnant, or multifocal disease, and OLT was not feasible owing to donor limitations or medical comorbidities. All received neoadjuvant chemotherapy using SIOPEL-based regimens, achieving partial tumor response. Tumors ranged from 5 to 12 cm and involved central hepatic segments, the portal region, or both lobes. PBT was delivered at a total dose of 50 GyE in 10–25 fractions as definitive or consolidative therapy, followed by surgical resection in three patients. Two patients additionally received targeted therapy and immunotherapy. At last follow-up, four patients were alive with no evidence of disease, while one patient died from tumor progression.
Conclusions. These cases suggest that proton beam therapy may serve as a feasible liver-sparing local treatment option for selected pediatric patients with unresectable or residual hepatoblastoma when surgery or OLT is not possible. While limited by availability and cost, PBT may facilitate multimodal therapy and preserve future treatment options.
Keywords: hepatoblastoma, proton beam therapy, pediatric liver tumor, unresectable cancer, radiotherapy, PRETEXT
Introduction
Hepatoblastoma (HB) is the most common pediatric liver malignancy, with surgery and chemotherapy as the main treatment pillars. Complete resection, via partial hepatectomy or orthotopic liver transplantation (OLT), remains the only curative approach. Tumors are considered unresectable when there is extensive vascular involvement, multifocal bilobar disease, or an inadequate residual liver volume post-resection. OLT is indicated for unresectable disease without extrahepatic spread.
Radiotherapy has traditionally been avoided in HB due to liver radiosensitivity, but modern techniques, particularly proton beam therapy (PBT), allow for conformal dose delivery with reduced low-dose exposure to the normal liver, lungs, heart, and kidneys. This makes PBT attractive for selected pediatric liver tumors. In adults, PBT has shown encouraging results in hepatocellular carcinoma,1,2 supporting its potential pediatric application. Selection criteria for pediatric PBT may include a tumor size < 12 cm, location near critical structures, the absence of widespread metastases, and preserved liver function.3
Case Presentations
Ethical approval
This study was reviewed and approved by the Institutional Review Board of Chang Gung Medical Foundation (IRB No. 202500060B0, date September 1, 2025). The requirement for informed consent was waived by the IRB due to the retrospective nature of the study and the use of anonymized clinical data. The study was conducted in accordance with the principles of the Declaration of Helsinki and Good Clinical Practice guidelines.
Patient-1
A 6-year-old girl with recurrent HB involving segments 7 and 8 and an associated portal vein thrombus was deemed inoperable due to an inadequate future liver remnant. She received four cycles of salvage chemotherapy with carboplatin plus etoposide, followed by PBT (50 GyE in 10 fractions), which resulted in significant tumor shrinkage. She has remained with no evidence of disease (NED) at 55 months of follow-up.
Patient-2
A 3-year-old girl with pre-treatment extent of tumor (PRETEXT) IV HB, complicated by lung metastases and celiac lymph node involvement, achieved a partial response following neoadjuvant chemotherapy. She subsequently underwent hepatectomy and pulmonary metastasectomy, followed by PBT to the residual hepatic lesion (50 GyE in 25 fractions). She has remained with NED at 108 months of follow-up.
Patient-3
A 6-month-old boy presented with a centrally located PRETEXT IV HB. He underwent eight cycles of neoadjuvant chemotherapy, followed by right hepatectomy and adjuvant chemotherapy. PBT was subsequently administered for a residual lesion. The patient has remained with NED at 55 months of follow-up.
Patient-4
An 18-month-old boy with PRETEXT IV HB and pulmonary metastases, with a history of extremely low birth weight, underwent a multi-step hepatectomy and pulmonary metastasectomy. Salvage targeted therapy with bevacizumab and sorafenib, followed by PBT and nivolumab, was administered. The patient died of disease at 28 months of age.
Patient-5
A 4-year-old girl with PRETEXT III HB involving the portal region received six cycles of neoadjuvant chemotherapy, followed by targeted therapy with bevacizumab and sorafenib, PBT, and nivolumab immunotherapy. Tumor resection was performed 16 months after diagnosis. She has remained with NED at 21 months of follow-up.
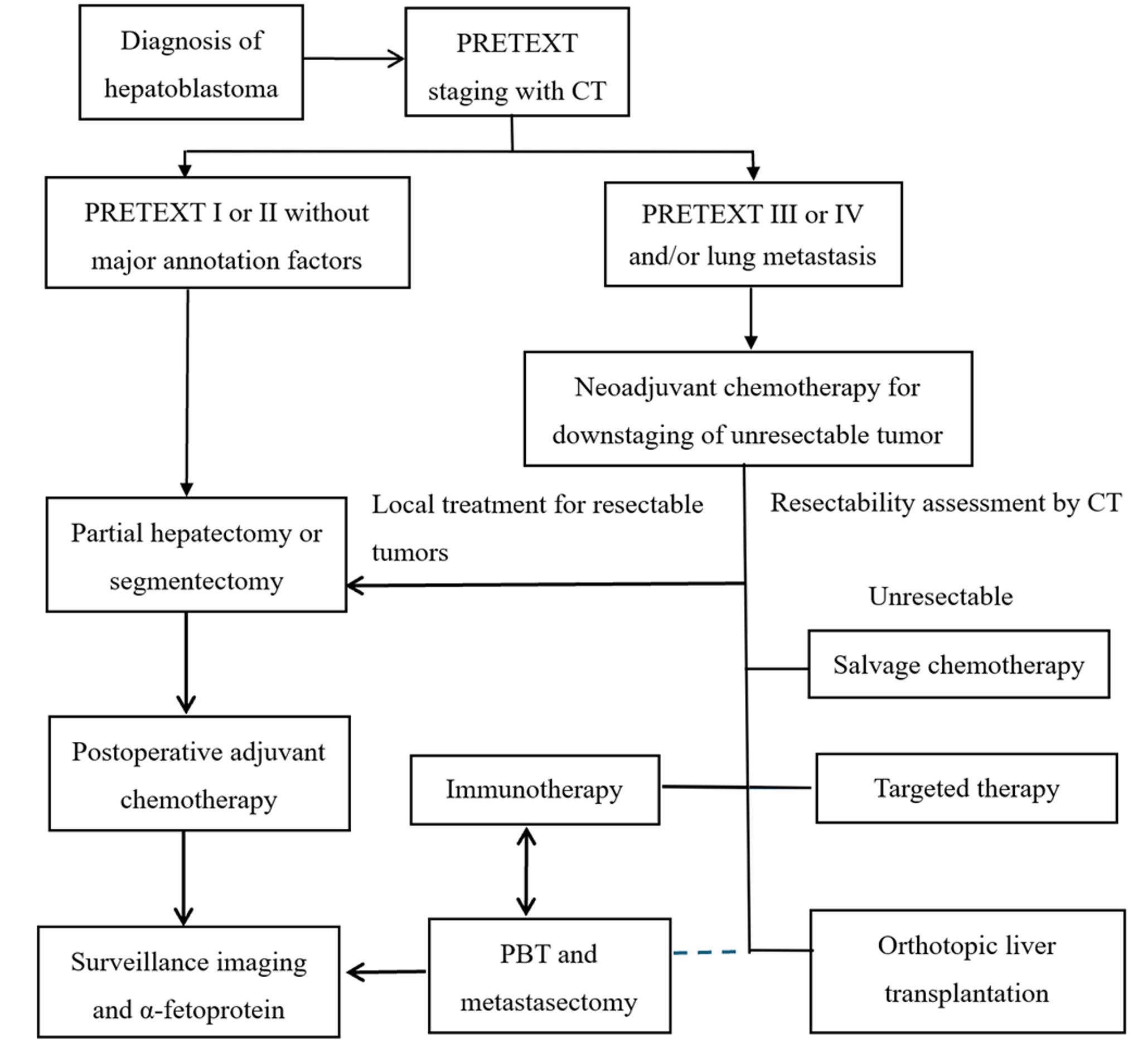
Table I outlines the PRETEXT stage, chemotherapy regimen, tumor location, surgical details, and PBT indication for these five patients. Fig. 1. presents a treatment algorithm for HB that is based on PRETEXT staging. It illustrates the pathways from initial diagnosis through surgical resection, chemotherapy, targeted therapy and immunotherapy, PBT, and OLT.
| DOD, died of disease; Fr, fractions; HB, hepatoblastoma; NED, no evidence of disease; PRETEXT, pre-treatment extent of tumor. | |||||||||
| Table I. Patient characteristics and treatment outcomes. | |||||||||
| Case | Age / Sex | PRETEXT stage | Tumor location | Metastasis / Other involvement | Chemotherapy regimen | Surgical details | PBT indication | PBT dose / Fractions | Outcome / Follow-up |
| 1 | 6 y / F | Recurrent HB | Segments 7–8 | Portal vein thrombus | 4 cycles carboplatin + etoposide (salvage) | None (inoperable) | Primary inoperable tumor with vascular involvement | 60 GyE / 10 Fr | NED at 55 mo |
| 2 | 3 y / F | IV | Right lobe | Lung metastases, celiac LN involvement | Neoadjuvant chemotherapy | Hepatectomy + pulmonary metastasectomy | Residual hepatic lesion post-surgery | 50 GyE / 25 Fr | NED at 108 mo |
| 3 | 6 mo / M | IV | Central | None | 8 cycles neoadjuvant (cisplatin-based) + adjuvant chemotherapy | Right hepatectomy | Residual hepatic lesion post-hepatectomy | 50 GyE / 10 Fr | NED at 55 mo |
| 4 | 18 mo / M | IV | Multifocal | Pulmonary metastases; history of extremely low birth weight | Multi-step surgery + salvage targeted therapy (bevacizumab, sorafenib) | Multi-step hepatectomy + pulmonary metastasectomy | Residual/refractory disease | 50 GyE / 10 Fr | DOD at 28 mo |
| 5 | 4 y / F | III | Portal region | None | 6 cycles neoadjuvant chemotherapy + targeted therapy (bevacizumab, sorafenib) | Tumor resection (16 mo post-diagnosis) | Preoperative downstaging | 50 GyE / 10 Fr | NED at 21 mo |
Discussion
Surgery remains the definitive therapy for HB, with OLT as the standard for unresectable disease without extrahepatic spread.4 However, in cases where OLT is not feasible, PBT can provide local control with reduced normal tissue toxicity.5 In our series, PBT was delivered with curative or consolidative intent, enabling surgical resection in some patients and durable disease control in others.
PBT advantages include dosimetric precision, a reduction in high-grade toxicities, and preservation of liver function, which is critical for pediatric patients with high long-term survival expectations.6-9 Drawbacks include the high cost, limited global availability, and the logistical complexity of daily anesthesia.10
Our findings, supported by existing literature, indicate that PBT is best suited for patients with localized unresectable tumors (such as central or hilar lesions with an inadequate future liver remnant), those who have failed or are unsuitable for orthotopic liver transplantation, candidates for potential resection following downstaging, or individuals requiring re-irradiation or salvage therapy.11
Future studies should prioritize multicenter registries and prospective trials to evaluate the role of PBT in HB,12 its integration with systemic and immunotherapies, and long-term outcomes including growth, development, and the risk of secondary malignancies.
Conclusion
PBT offers a safe, precise, and potentially effective local treatment option for selected pediatric HB cases in which surgical resection or OLT is not feasible. Optimal utilization requires multidisciplinary patient selection, careful integration with systemic therapies, and consideration of resource availability. Further prospective research is warranted to clarify its long-term efficacy and outcomes.
Ethical approval
The study was approved by Institutional Review Board of Chang Gung Medical Foundation (date: September 1, 2025, number: 202500060B0).
Source of funding
The authors declare the study received no funding.
Conflict of interest
The authors declare that there is no conflict of interest.
References
- Mizumoto M, Ogino H, Okumura T, et al. Proton beam therapy for hepatocellular carcinoma: multicenter prospective registry study in Japan. Int J Radiat Oncol Biol Phys 2024; 118: 725-733. https://doi.org/10.1016/j.ijrobp.2023.09.047
- Kobeissi JM, Hilal L, Simone CB, Lin H, Crane CH, Hajj C. Proton therapy in the management of hepatocellular carcinoma. Cancers (Basel) 2022; 14: 2900. https://doi.org/10.3390/cancers14122900
- Singal AG, Llovet JM, Yarchoan M, et al. AASLD practice guidance on prevention, diagnosis, and treatment of hepatocellular carcinoma. Hepatology 2023; 78: 1922-1965. https://doi.org/10.1097/HEP.0000000000000466
- Al-Qabandi W, Jenkinson HC, Buckels JA, et al. Orthotopic liver transplantation for unresectable hepatoblastoma: a single center’s experience. J Pediatr Surg 1999; 34: 1261-1264. https://doi.org/10.1016/s0022-3468(99)90164-1
- Yoo GS, Yu JI, Park HC. Proton therapy for hepatocellular carcinoma: current knowledges and future perspectives. World J Gastroenterol 2018; 24: 3090-3100. https://doi.org/10.3748/wjg.v24.i28.3090
- Dionisi F, Scartoni D, Fracchiolla F, et al. Proton therapy in the treatment of hepatocellular carcinoma. Front Oncol 2022; 12: 959552. https://doi.org/10.3389/fonc.2022.959552
- Dionisi F, Brolese A, Siniscalchi B, et al. Clinical results of active scanning proton therapy for primary liver tumors. Tumori 2021; 107: 71-79. https://doi.org/10.1177/0300891620937809
- Dionisi F, Guarneri A, Dell’Acqua V, et al. Radiotherapy in the multidisciplinary treatment of liver cancer: a survey on behalf of the Italian Association of Radiation Oncology. Radiol Med 2016; 121: 735-743. https://doi.org/10.1007/s11547-016-0650-5
- Lee SU, Kim TH. Current evidence and the potential role of proton beam therapy for hepatocellular carcinoma. Clin Mol Hepatol 2023; 29: 958-968. https://doi.org/10.3350/cmh.2023.0274
- Lee CH, Hung SP, Hong JH, et al. How small is TOO small? New liver constraint is needed- Proton therapy of hepatocellular carcinoma patients with small normal liver. PLoS One 2018; 13: e0203854. https://doi.org/10.1371/journal.pone.0203854
- Chen CL, Dungca LBP, Yong CC, et al. Proton beam therapy for downstaging hepatocellular carcinoma with lobar portal vein tumor thrombosis to living donor liver transplantation. Hepatobiliary Surg Nutr 2023; 12: 966-974. https://doi.org/10.21037/hbsn-23-410
- Sakurai H, Ishikawa H, Okumura T. Proton beam therapy in Japan: current and future status. Jpn J Clin Oncol 2016; 46: 885-892. https://doi.org/10.1093/jjco/hyw102
License
Copyright (c) 2026 The Author(s). This is an open access article distributed under the Creative Commons Attribution License (CC BY), which permits unrestricted use, distribution, and reproduction in any medium or format, provided the original work is properly cited.















