Abstract
Background. Actinomycotic osteomyelitis is a rare, chronic infection caused by Actinomyces species, anaerobic bacteria normally found in the oral flora. Mandibular involvement is uncommon and may mimic malignancies, complicating diagnosis.
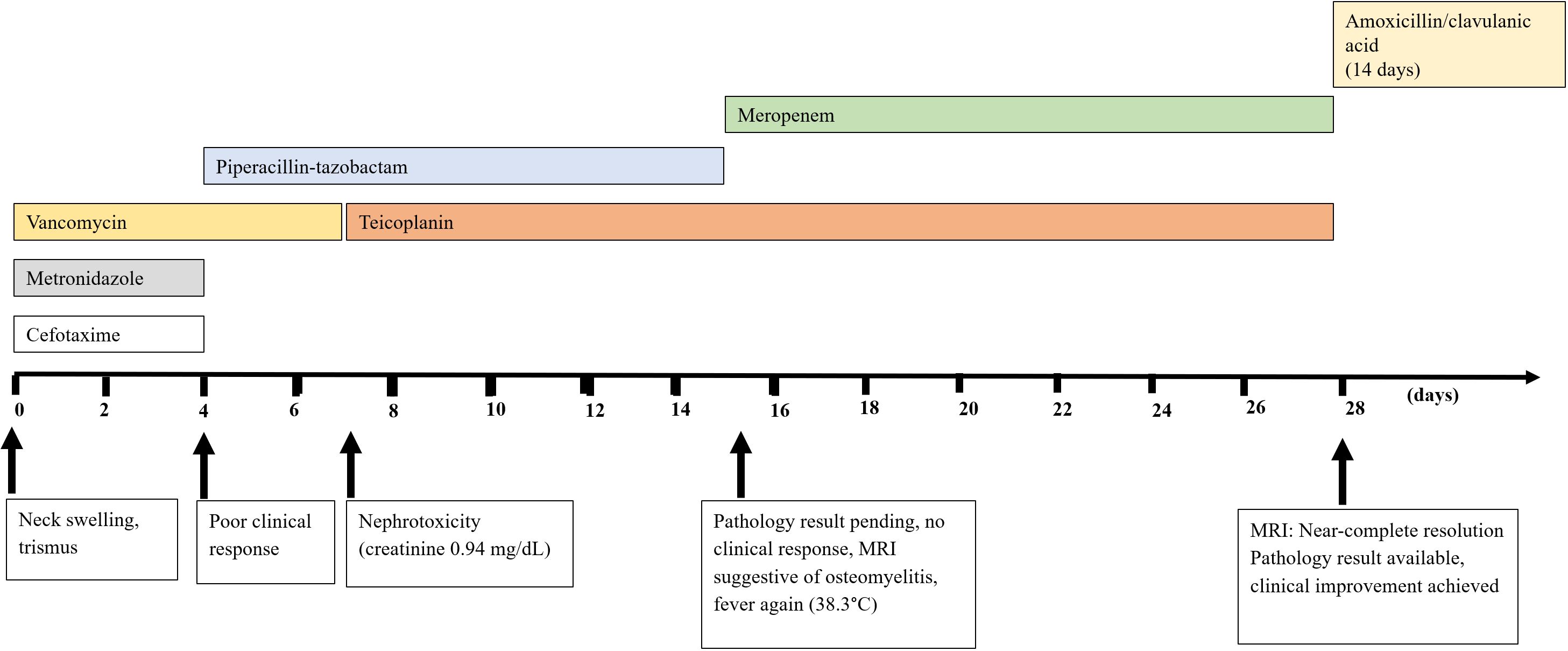
Case Presentation. A 15-year-old previously healthy male presented with painful neck swelling and trismus. Examination revealed a firm mass at the angle of the mandible. Imaging suggested osteomyelitis but raised concern for malignancy. Surgical drainage was performed, and histopathology confirmed actinomycotic infection. The patient initially received empirical intravenous vancomycin, cefotaxime, and metronidazole. Due to persistent fever and elevated inflammatory markers, the regimen was switched to teicoplanin plus piperacillin/tazobactam. Therapy was subsequently escalated to meropenem due to recurrent fever and based on magnetic resonance imaging findings suggestive of osteomyelitis, then vancomycin was replaced with teicoplanin because of vancomycin-associated nephrotoxicity. Following clinical improvement, the patient was discharged on oral amoxicillin/clavulanate to complete a total of six weeks of therapy.
Discussion. Pediatric mandibular actinomycotic osteomyelitis is extremely rare. Its indolent course often mimics tumors or granulomatous disease. In this case, delayed diagnosis and nonspecific imaging findings led to initial misinterpretation. Surgical intervention played a key role in diagnosis and treatment. Early recognition and combined medical-surgical management are crucial to avoid complications. This case highlights the importance of considering infectious causes in mandibular masses and underscores the diagnostic challenges associated with them.
Keywords: actinomycosis, Actinomyces, mandibular osteomyelitis, pediatric
Introduction
Actinomycosis is a chronic infection caused by anaerobic, Gram-positive filamentous bacteria of the genus Actinomyces. Actinomyces are normally found in the oropharynx, gastrointestinal tract, and genitourinary tract. Under certain conditions, these bacteria can become pathogenic.1,2 Pediatric actinomycosis is rare and often presents with nonspecific symptoms that mimic other infections or tumors, complicating diagnosis. Actinomycotic infections typically involve primary soft tissues in chronic inflammatory conditions, rarely affecting the bones.3 Bacterial cultures and histopathological examination are fundamental to establishing an accurate diagnosis.4
This case report presents a rare pediatric cervicofacial actinomycosis involving the mandible, highlighting the importance of imaging, biopsy, and multidisciplinary treatment for successful outcomes.
Case Presentation
A 15-year-old male patient with no known comorbidities presented to the clinic with progressively enlarging, painful neck swelling, difficulty opening his mouth and restricted neck movement over the past 10 days. On physical examination, hypertrophic tonsils and dental caries in the right mandibular region were noted. A firm-to-hard, diffuse erythematous swelling was observed in the paratracheal region, with restricted mobility in all directions.
Laboratory tests revealed an elevated C-reactive protein (CRP) level of 99 mg/L, erythrocyte sedimentation rate (ESR) of 38 mm/h, white blood cell (WBC) count of 8.13 ×10³/µL, hemoglobin level of 12.7 g/dL, absolute neutrophil count (ANC) of 5.18 ×10³/µL, and platelet count of 210000/µL
Ultrasound (US) examination showed thickened, inflamed subcutaneous tissue in the midline of the neck, with a 19x11 mm hypoechoic collection suggestive of phlegmon or abscess. A computed tomography (CT) scan revealed increased density and inflammatory changes extending from the anterior neck to the left mandibular region, consistent with deep neck infection. Mild density changes in the bone marrow of the left mandibular ramus raised concerns for osteomyelitis.
Empirical intravenous therapy with vancomycin, cefotaxime, and metronidazole was initiated (Fig. 1). An otolaryngology consultation resulted in the percutaneous drainage of approximately 3 mL of purulent material. On the third day of hospitalization, as the swelling and restricted mobility persisted, interventional radiology performed ultrasound-guided percutaneous drainage of the abscess.
On the fourth day, due to an inadequate clinical response, cefotaxime was discontinued and replaced with piperacillin/tazobactam, while vancomycin therapy was continued. On the seventh day, the patient developed vomiting following vancomycin administration, and an elevation in serum creatinine to 0.94 mg/dL raised concerns for vancomycin-associated nephrotoxicity. Vancomycin was replaced with teicoplanin.
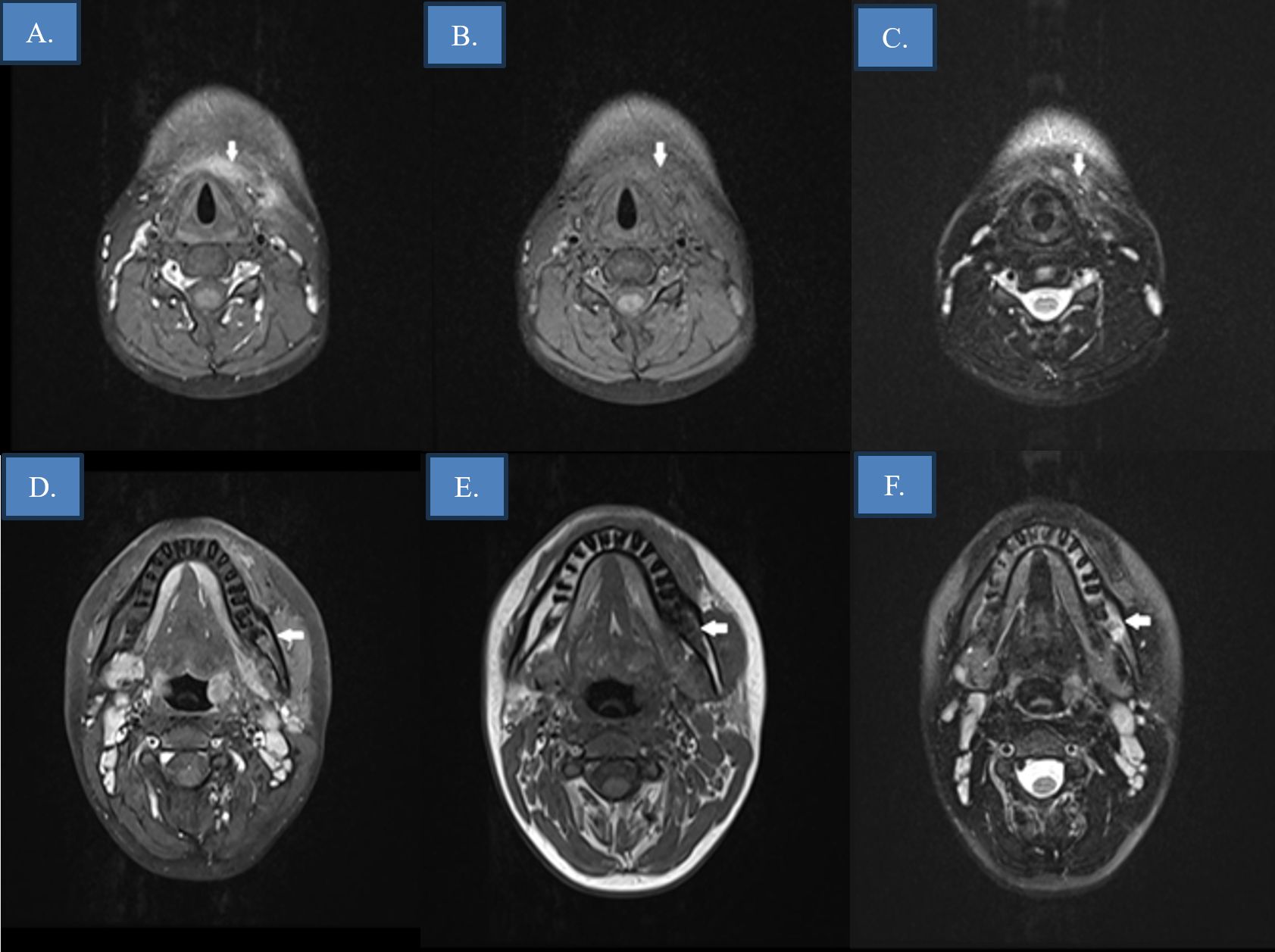
Magnetic resonance imaging (MRI) revealed extensive anterior cervical soft tissue inflammation extending from the submental region to the thyroid level. A deep-seated lesion measuring 13×9 mm adjacent to the hyoid bone was identified, consistent with a phlegmon. Multiple enlarged cervical lymph nodes were observed. Additionally, signal alterations around the roots of the fourth and fifth mandibular molars raised suspicion for mandibular osteomyelitis (Fig. 2).
On the seventh day, cervical mobility had returned to normal. US demonstrated residual cellulitis and a 10×4 mm hypoechoic area consistent with a resolving phlegmon. On day 15, the patient developed a fever of 38.3 °C, although no new infectious focus was identified. Laboratory results were as follows: creatinine 0.76 mg/dL, CRP 7.5 mg/L, ESR 38 mm/h, procalcitonin 0.21 ng/mL, WBC 13×10³/µL. Then, inflammatory markers worsened, with CRP increasing to 47 mg/L and WBC count rising to 15×10³/µL. Due to ongoing intermittent fever and the presence of suspected osteomyelitis, piperacillin/tazobactam was discontinued, and meropenem therapy was initiated.
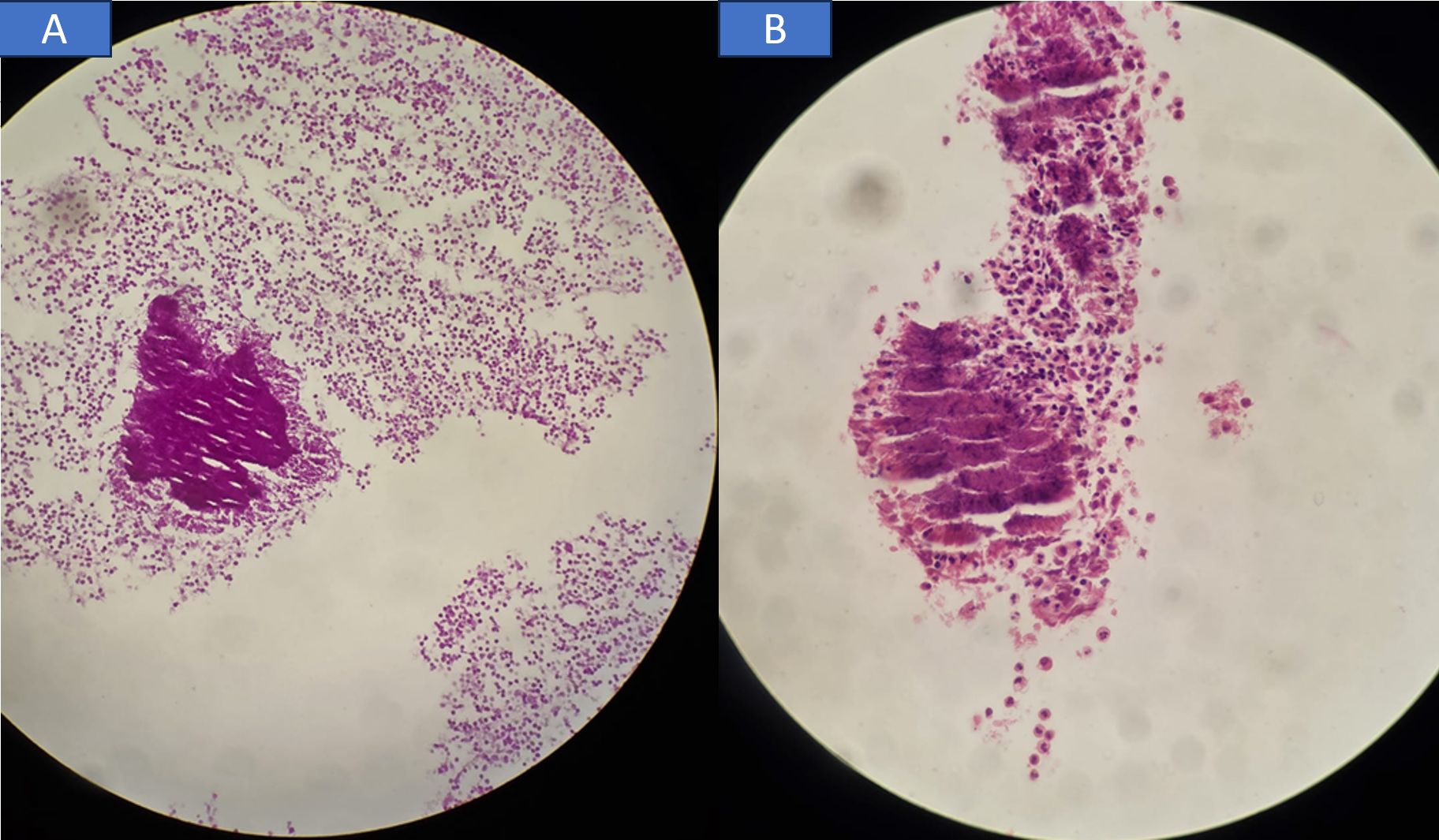
Wound cultures showed no bacterial growth. Direct Gram staining of the drained fluid did not demonstrate identifiable organisms. Histopathological examination of the abscess material revealed abundant polymorphonuclear leukocytes and histiocytes, along with nonspecific inflammatory infiltrates and vascular proliferation. A cluster of filamentous microorganisms morphologically suggestive of Actinomyces species was identified, further supported by positive periodic acid–Schiff (PAS) staining (Fig. 3). Modified acid-fast staining was negative. This finding argues against Nocardia spp., which typically demonstrate partial acid-fast positivity due to the presence of mycolic acids in their cell walls. Therefore, the absence of acid-fast staining favored Actinomyces over Nocardia in the differential diagnosis. Blood cultures remained negative. Upon consultation with the microbiology department, it was noted that anaerobic cultures had not been performed. On day 21, follow-up ultrasonography demonstrated marked regression of the inflammatory process, with only a 9×3 mm residual hypoechoic lesion in the anterior cervical region. In light of the confirmed Actinomyces infection and mandibular osteomyelitis, the patient completed a total of four weeks of intravenous antibiotic therapy. Although Actinomyces species are highly susceptible to penicillin, de-escalation was not pursued in our patient due to the severity of infection, suspected mandibular osteomyelitis, and ongoing clinical instability at the time of histopathologic confirmation. Broad-spectrum therapy was therefore maintained to ensure adequate coverage until clinical resolution was achieved.
Follow-up MRI on day 28 demonstrated near-complete resolution of cellulitis, with no evidence of abscess formation. The patient was discharged on oral antibiotic therapy to complete a total of six weeks of treatment (Fig. 1). A written informed consent was obtained from the parents of the patient for this publication.
Discussion
Pediatric actinomycosis is a rare but challenging infection, often presenting as deep neck infections caused by Actinomyces species, Gram-positive, filamentous, anaerobic bacteria found in the oral cavity, gastrointestinal, and genitourinary tracts.4 While these infections typically involve soft tissues with a chronic course, bone involvement is uncommon.5 Poor oral hygiene and antecedent oropharyngeal disease increase susceptibility; in our patient, the presence of untreated dental caries likely contributed to the disruption of mucosal barriers, facilitating the invasion of Actinomyces and subsequent mandibular involvement.6 A 16-year retrospective study by Chew et al. identified 14 pediatric cases, mostly cervicofacial, followed by thoracic and abdominopelvic forms.6 The mandible’s poor vascularity may contribute to its susceptibility.7,8 A broader review of the literature reveals that pediatric cases of actinomycotic osteomyelitis are extremely rare, with only a limited number published to date.9-20 These cases, summarized in Table I, highlight the variability in age, infection site, microbiologic findings, and management strategies and emphasize the diagnostic challenges associated with this unusual clinical entity.
| AMC: amoxicillin / clavulanic acid, F: female, M: male. | |||||
| Table I. Overview of pediatric osteomyelitis cases attributed to Actinomyces: clinical presentation, diagnosis, and management. | |||||
| Author (year) | Age (yr) / sex | Location | Histopathologic diagnostic method | Culture result | Antibiotic treatment |
| Chew et al.6 (2023) | 9 / F | Cervical spine | Histopathologic diagnosis | Anaerobic culture negative | Ampicillin → AMC |
| Chew et al.6 (2023) | 9/ F | Mandible | Excision debridement | Anaerobic culture negative | AMC |
| Chew et al.6 (2023) | 9/ F | Mandible | Incision and drainage of the abscess | Actinomyces israelii | AMC and ceftriaxone |
| Chew et al.6 (2023) | 12/ F | Mastoid | Incision and drainage, mastoidectomy | Anaerobic culture negative | AMC |
| Conley et al.14 (2022) | 17 / M | Anterior skull base | Medial orbitotomy for drainage | Actinomyces | Clindamycin, ceftriaxone |
| Iwai et al.11 (2021) | 14 / F | Parotid-masseter | Histopathologic diagnosis | Anaerobic culture not reported | Ampicillin, later amoxicillin |
| Mou et al.12 (2021) | 5 / F | Right lower extremity | Debridement and drainage | Actinomycetes europaeus | Ampicillin-sulbactam |
| Boorman et al.9 (2020) | 7 / M | Nasofrontal region | Sequestrum, debridement | Actinomyces funkei | Chloramphenicol |
| Saarinen et al.16 (2011) | 13 / M | Mandible | Debridement | Actinomyces | Penicillin |
| Saarinen et al.16 (2011) | 11 / F | Mandible | No | Actinomyces turicencis | Penicillin |
| Saarinen et al.16 (2011) | 5 / F | Mandible | No | Actinomyces | Penicillin |
| Saarinen et al.16 (2011) | 17 / F | Mandible | No | Actinomyces | Amoxicillin |
| Catalano-pons et al.17 (2007) | 6/ F | Iliac bone | Histopathologic diagnosis | Actinomyces israelii | Amoxicillin |
| Catalano-pons et al.17 (2007) | 7/ M | Elbow and the mandible | Histopathologic diagnosis | Actinomyces | Metronidazole and amphotericin B, followed by amoxicillin alone |
| Robinson et al.15 (2005) | 4/ F | Ramus and angle of the jaw | Debridement, sequestrectomy, | Actinomyces israelii | Amoxicillin-clavulanate |
| Robinson et al.15 (2005) | 3/ M | Mandible | Sequestrectomy | Actinomyces naeslundii | Penicillin → clindamycin, then amoxicillin |
| Sobol et al.13 (2004) | 14/ F | Temporal bone | Tympanomastoidectomy | Anaerobic culture not reported | Oral penicillin |
| Thisted et al.19 (1987) | 3/ M | Mandible | Puncture | Actinomyces spp. | Ampicillin, metronidazole |
| Vannier et al.20 (1986) | 13/ F | Skull and atlas | Puncture, drainage | Actinomyces israelii | Erythromycin and metronidazole |
| Walker, et al.18 (1981) | 7/ M | Mandible | Curettage | Actinomyces israelii | Penicillin |
| Present case | 15/ M | Mandible | Drainage | Anaerobic culture not performed, aerobic culture negative | Vancomycin, metronidazole, cefotaxime → teicoplanin, piperacillin/tazobactam → meropenem, teicoplanin → at discharge: oral AMC |
This case is notable for its acute presentation, with trismus and a rapidly enlarging mass—features that mimicked malignancy. This aligns with prior studies showing that pediatric actinomycosis may resemble tumors or granulomatous diseases, leading to misdiagnosis or delayed treatment.6 Although Actinomyces infections are typically chronic, they may present acutely in children, complicating diagnosis. Clinical and radiological findings are often nonspecific and may mimic malignancy or chronic infections. Chew et al.6 similarly reported that pediatric actinomycosis can resemble neoplastic or granulomatous diseases, leading to delayed diagnosis and treatment.
Despite negative wound cultures, the diagnosis was ultimately confirmed via histopathologic analysis. This reflects existing challenges in isolating Actinomyces due to their slow growth and anaerobic nature.4,7,8 Consequently, histopathology—particularly the detection of sulfur granules and filamentous Gram-positive organisms—remains a cornerstone in diagnosis. Although histopathology confirmed the presence of Actinomyces, anaerobic cultures were not performed in this case. This major limitation resulted in delays in both definitive diagnosis and initiation of targeted therapy, underscoring the critical role of anaerobic cultures when actinomycosis is suspected.
Surgical management of actinomycotic osteomyelitis, including curettage, sequestrectomy, and peripheral osteotomy, plays a critical role in both diagnosis and treatment.21 Although penicillin is generally effective against Actinomyces, broad-spectrum antibiotics were initially preferred in our patient due to the infection’s severity and poor early response. Chew et al.6 recommend tailoring antibiotic regimens to the infection’s extent, location, and course, supporting this approach.
Actinomyces species are slow-growing, with low metabolic activity, and form dense tissue abscesses and sinus tracts, which limit antibiotic penetration and delay clinical response.2 Consequently, prolonged antibiotic therapy is required to achieve adequate tissue concentrations and prevent relapse. The standard regimen consists of high-dose intravenous penicillin G for 2–6 weeks.1,4 This extended treatment is particularly important in cases with deep tissue or bone involvement, such as mandibular osteomyelitis. Shorter courses have been associated with higher relapse rates and suboptimal outcomes.
Although high-dose intravenous penicillin remains the standard of care, our patient received broad-spectrum antibiotics. This decision was based on the severity of the infection, the presence of mandibular osteomyelitis, and clinical instability at the time of histopathologic confirmation. Given these factors, de-escalation to penicillin was not pursued, and therapy was maintained with broad-spectrum coverage until clinical resolution. The literature consistently supports prolonged penicillin therapy as first-line treatment; however, tailored regimens may be necessary in severe, refractory, or complicated cases.
Imaging techniques, including CT and MRI, were essential for both diagnosis and monitoring.22 These modalities help distinguish infection from neoplasm and track disease progression. On day 28, follow-up imaging demonstrated near-complete resolution. This aligns with literature emphasizing the value of serial imaging to guide treatment duration and detect complications.22,23
In conclusion, this case highlights the need for a multidisciplinary approach including infectious disease specialists, radiologists, otolaryngologists, microbiologists, and pathologists. Pediatric cervicofacial actinomycosis, though rare, should be considered in persistent infections with negative cultures and osteomyelitis. Multidisciplinary care improves diagnostic accuracy and treatment outcomes.
Ethical approval
A written informed consent was obtained from the parents of the patient for this publication.
Source of funding
The authors declare the study received no funding.
Conflict of interest
The authors declare that there is no conflict of interest.
References
- Wong VK, Turmezei TD, Weston VC. Actinomycosis. BMJ 2011; 343: d6099. https://doi.org/10.1136/bmj.d6099
- Valour F, Sénéchal A, Dupieux C, et al. Actinomycosis: etiology, clinical features, diagnosis, treatment, and management. Infect Drug Resist 2014; 7: 183-197. https://doi.org/10.2147/IDR.S39601
- Bartkowski SB, Zapala J, Heczko P, Szuta M. Actinomycotic osteomyelitis of the mandible: review of 15 cases. J Craniomaxillofac Surg 1998; 26: 63-67. https://doi.org/10.1016/s1010-5182(98)80037-1
- Mandell GL, Bennett JE, Dolin R, editors. Mandell, Douglas, and Bennett’s principles and practice of infectious diseases. 7th ed. Philadelphia, PA: Churchill Livingstone Elsevier; 2010.
- Sharkawy AA. Cervicofacial actinomycosis and mandibular osteomyelitis. Infect Dis Clin North Am 2007; 21: 543-56, viii. https://doi.org/10.1016/j.idc.2007.03.007
- Chew SJ, Low KB, Chong CY, et al. Paediatric actinomycosis: a 16-year, single-institution retrospective review of cases. J Paediatr Child Health 2023; 59: 833-839. https://doi.org/10.1111/jpc.16400
- Figueiredo LMG, Trindade SC, Sarmento VA, de Oliveira TFL, Muniz WR, Valente RODH. Actinomycotic osteomyelitis of the mandible: an unusual case. Oral Maxillofac Surg 2013; 17: 299-302. https://doi.org/10.1007/s10006-012-0381-2
- Simre SS, Jadhav AA, Patil CS. Actinomycotic osteomyelitis of the mandible - a rare case report. Ann Maxillofac Surg 2020; 10: 525-528. https://doi.org/10.4103/ams.ams_99_20
- Boorman S, Boone L, Weatherall K, Caldwell F. Actinomyces as a cause of osteomyelitis of the nasofrontal suture in a gelding. J Equine Vet Sci 2020; 91: 103148. https://doi.org/10.1016/j.jevs.2020.103148
- Bulut G, Bayram Y, Bulut MD, Garça MF, Bayram İ. Mandibular actinomyces infection mimicking a malignancy: case report. Turk Patoloji Derg 2017; 33: 256-258. https://doi.org/10.5146/tjpath.2014.01276
- Iwai T, Ohashi N, Sugiyama S, et al. Actinomycotic osteomyelitis with proliferative periostitis arising in the mandibular ramus: an unusual case with spontaneous bone regeneration after coronoidectomy. Oral Radiol 2021; 37: 137-145. https://doi.org/10.1007/s11282-020-00462-x
- Mou Y, Jiao Q, Wang Y, et al. Musculoskeletal actinomycosis in children: a case report. BMC Infect Dis 2021; 21: 1220. https://doi.org/10.1186/s12879-021-06890-2
- Sobol SE, Samadi DS, Wetmore RF. Actinomycosis of the temporal bone: a report of a case. Ear Nose Throat J 2004; 83: 327-329.
- Conley W, Cox RE, Robey T. Rhinocerebral mucormycosis associated with anterior skull base actinomyces osteomyelitis in a pediatric patient with type 1 diabetes. Cureus 2022; 14: e24311. https://doi.org/10.7759/cureus.24311
- Robinson JL, Vaudry WL, Dobrovolsky W. Actinomycosis presenting as osteomyelitis in the pediatric population. Pediatr Infect Dis J 2005; 24: 365-369. https://doi.org/10.1097/01.inf.0000157215.15152.c2
- Saarinen RT, Kolho KL, Kontio R, Saat R, Salo E, Pitkäranta A. Mandibular osteomyelitis in children mimicking juvenile recurrent parotitis. Int J Pediatr Otorhinolaryngol 2011; 75: 811-814. https://doi.org/10.1016/j.ijporl.2011.03.013
- Catalano-Pons C, Raymond J, Chalumeau M, Armengaud JB, Kalifa G, Gendrel D. Case 2: paediatric chronic osteomyelitis: report of two cases. Case 1 diagnosis: pulmonary TB complicated by pneumomediastinum. Case 2 diagnosis: osteomyelitis caused by actinomyces. Acta Paediatr 2007; 96: 1849-1852. https://doi.org/10.1111/j.1651-2227.2007.00547.x
- Walker S, Middelkamp JN, Sclaroff A. Mandibular osteomyelitis caused by actinomyces israelii. Oral Surg Oral Med Oral Pathol 1981; 51: 243-244. https://doi.org/10.1016/0030-4220(81)90052-9
- Thisted E, Poulsen P, Christensen PO. Actinomycotic osteomyelitis in a child. J Laryngol Otol 1987; 101: 746-748. https://doi.org/10.1017/s0022215100102658
- Vannier JP, Schaison G, George B, Casin I. Actinomycotic osteomyelitis of the skull and atlas with late dissemination. a case of transient neurosurgical syndrome. Eur J Pediatr 1986; 145: 316-318. https://doi.org/10.1007/BF00439411
- Mufeed A, Reshma VJ, Anwar S, Ashir KR, Ahmed A. Actinomycotic osteomyelitis of the mandible. Journal of Oral and Maxillofacial Surgery, Medicine, and Pathology 2017; 29: 166-169. https://doi.org/10.1016/j.ajoms.2016.10.005
- Lau L, Darraj M, Keynan Y. Mandibular actinomyces osteomyelitis mimicking osteosarcoma. JMM Case Reports 2016; 3: 1-3. https://doi.org/10.1099/jmmcr.0.005073
- An CH, An SY, Choi BR, et al. Hard and soft tissue changes of osteomyelitis of the jaws on CT images. Oral Surg Oral Med Oral Pathol Oral Radiol 2012; 114: 118-126. https://doi.org/10.1016/j.oooo.2012.01.009
License
Copyright (c) 2026 The Author(s). This is an open access article distributed under the Creative Commons Attribution License (CC BY), which permits unrestricted use, distribution, and reproduction in any medium or format, provided the original work is properly cited.