Graphical Abstract
Abstract
Background. Flexible bronchoscopy (FB) is a critical diagnostic and therapeutic tool in respiratory diseases, enabling airway assessment, sample collection, and therapeutic interventions. Despite international guidelines, practices vary widely across centers. This study aimed to assess current FB practices and to identify variations among pediatric pulmonologists in Türkiye.
Methods. A descriptive cross-sectional survey was distributed via email to clinical directors of 19 centers performing FB in March 2023. The survey comprised 80 questions across seven domains: demographics, patient preparation, bronchoscopy procedure, sedation/anesthesia, discharge, bronchoscope cleaning, and respondent comments. Participants were asked to provide accurate and objective data on FB practices at their centers.
Results. All 19 centers participated in the survey, achieving a 100% response rate. The median FB experience was 12.5 years (IQR: 1-30), with a median 210 procedures per year per center (min-max: 30-500; IQR: 80-250). The most frequently reported indications were atelectasis (84.2%), bronchiectasis (78.9%), suspected foreign body aspiration (52.6%), and to obtain bronchoalveolar lavage (BAL) in patients with immunodeficiency (52.6%). Nine centers (47.4%) indicated performing advanced bronchoscopic procedures, including endobronchial biopsy (31.6%), bronchoscopic intubations (26.3%), and tracheal/bronchial stent insertion (10.5%). General anesthesia (84.2%) was the predominant sedation method, and propofol (89.5%) the most frequently used agent. Premedication was used in 13 centers (68.4%), with midazolam being the most commonly used agent (76.9%). Twelve centers (63.2%) also used topical lidocaine for local anesthesia. Respondents reported the need for developing interventional procedures and increasing the number of trained bronchoscopy teams.
Conclusions. This first comprehensive national survey of pediatric FB practices in Türkiye, encompassing all relevant centers, revealed significant heterogeneity in procedural approaches, particularly regarding sedation protocols, BAL techniques, and interventional capabilities. The findings underscore the importance of developing interventional procedures and enhancing the training of pediatric pulmonologists and interdisciplinary teams practicing pediatric bronchoscopy to improve patient outcomes and procedural consistency.
Keywords: flexible bronchoscopy, pediatric, survey
Introduction
Flexible bronchoscopy (FB) is an essential endoscopic technique in pediatric pulmonology since its introduction in the late 20th century.1 It allows for direct visualization of the anatomical and functional aspects of the airway for diagnostic and therapeutic purposes, including sample collection for cytological and microbiological studies, and therapeutic interventions.1-3 With the improvement of bronchoscopy equipment and technology, FB has gradually been applied to neonates and high-risk patients, including those undergoing lung transplantation and extracorporeal membrane oxygenation in intensive care units (ICU).4,5 Moreover, anesthesia management lacks standardization, with different methods chosen based on the indications for bronchoscopy.1,6,7 The growing need for an interdisciplinary approach to FB emphasizes the importance of establishing national standards and practice policies. Although international and national guidelines on FB indications, management, and techniques exist1-3,6,7, recent European Respiratory Society Task Force statements and survey studies highlight significant inconsistencies in clinical practice.3,8-13
Survey studies offer a cost-effective and efficient way to collect large-scale data, identify best practices, and highlight challenges, ultimately aiding in the development of evidence-based national guidelines.14 In Türkiye, pediatric FB has been increasingly adopted in both tertiary and secondary centers. However, a comprehensive understanding of current practices, including adherence to international standards, technical preferences, and procedural safety, is lacking. While a limited number of surveys have assessed pediatric FB practices8-11, no such data are currently available from Türkiye. To address this gap, we conducted a nationwide survey among pediatric pulmonologists in Türkiye to evaluate the current approaches to FB, identify areas of variability, and highlight opportunities for standardization and improvement in clinical practice. This survey may also serve as a reference for new bronchoscopy units and help clinicians refer patients to centers with established FB programs.
Materials and Methods
Study design
This descriptive cross-sectional study was conducted using an online questionnaire created via Google Forms. The survey was developed based on existing bronchoscopy guidelines and previous pediatric bronchoscopy surveys.1-3,8-10 It consisted of 80 questions divided into seven categories: demographics, patient preparation and preprocedural tests, bronchoscopy procedure, sedation/anesthesia, discharge process, bronchoscope cleaning, and comments. Participants responded using yes/no options, multiple-choice answers, or free-text (e.g., indications) responses where applicable. They could also provide comments and suggestions at the end of the questionnaire. No patient data were collected. The study was approved by the Ege University Faculty of Medicine Ethics Committee (Approval No: 23-5T/40, Date: 05.11.2023).
Since pediatric pulmonology is a relatively new subspecialty in Türkiye and the number of specialists performing flexible bronchoscopy is limited, we invited all 19 pediatric pulmonology centers known to perform the procedure to participate. The survey link, information sheet, and an electronic informed consent form were emailed to the clinical directors of these centers in March 2023. Access to the questionnaire was enabled only after the consent box was checked. Completion of the questionnaire indicated voluntary participation and constituted informed consent. Participants were asked to provide accurate and objective data on FB practices at their centers.
Only the city of each medical center was disclosed, while the center names remained confidential. Participation was voluntary, with no incentives offered. To improve the response rate, a follow-up email was sent four weeks after the initial request. Data collection lasted three months, and all participants approved participation in the study.
Statistical analysis
The answers extracted from Microsoft Excel spreadsheets were processed, and analyzed using Statistical Package for Social Sciences (SPSS for Windows Version 26) software. Descriptive data are given as numbers (n) and the percentage values (%). Values for continuous variables were given as either mean ± standard deviation (SD) or as median (interquartile range [IQR]), based on the normality of their distribution.
Results
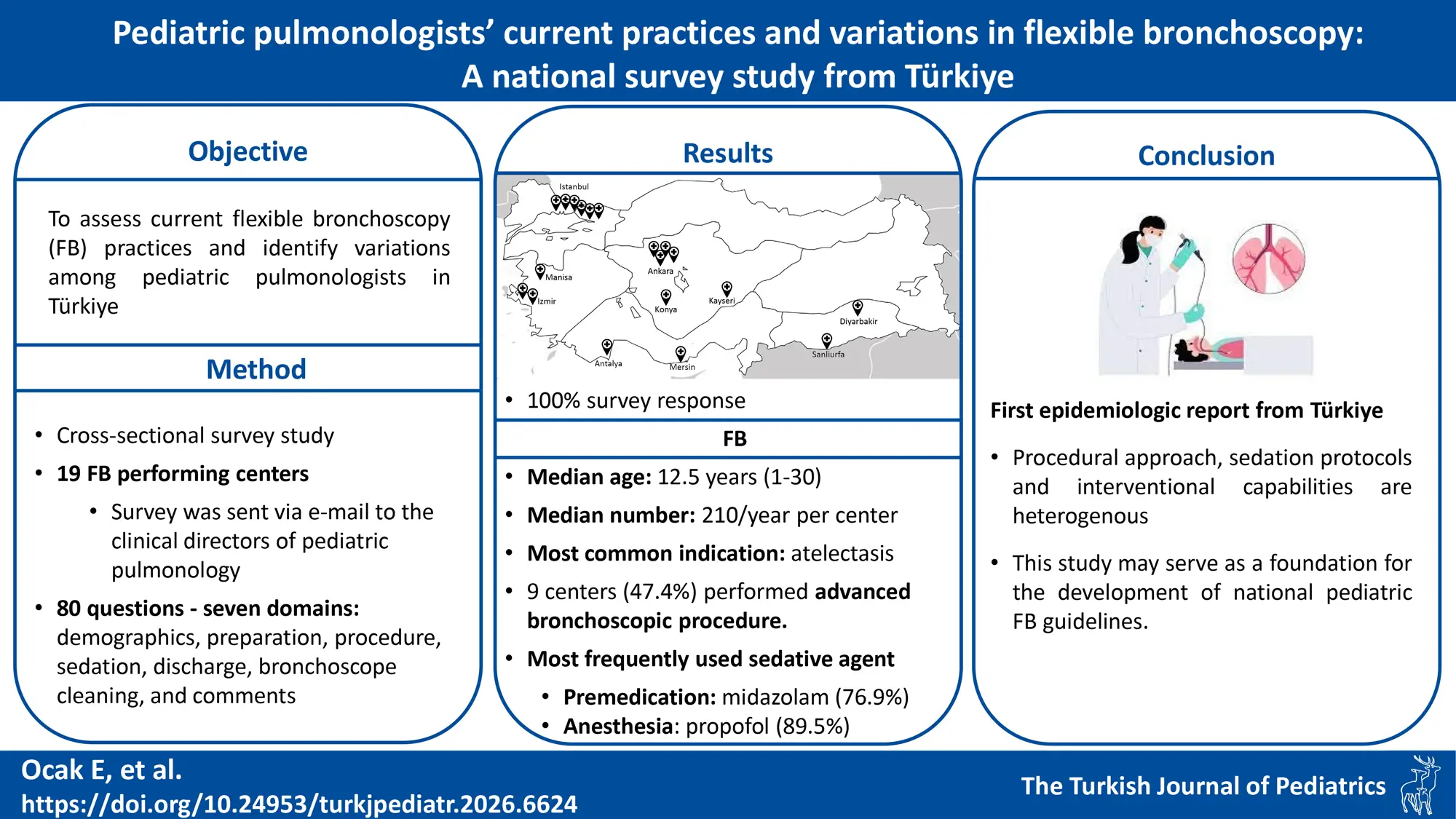
Clinical directors from 19 pediatric pulmonology departments performing FB participated in the study. The median number of clinicians per center was nine (IQR: 1-13), with 76% female. Twelve centers (63.2%) were located in Türkiye’s three most populous metropolitan cities (Fig. 1). The median FB experience was 12.5 years (IQR: 1-30). Three centers (15.8%) had less than three years of FB experience, four (21.1%) had 6-10 years, six (31.6%) had 11-15 years, five (26.3%) had 16–20 years, and one (5.3%) had 30 years of experience.
A total of 3,118 FB procedures were performed in the previous year. Annual pediatric FBs per center had a median of 210 (min-max: 30-500; IQR: 80-250).
Indications
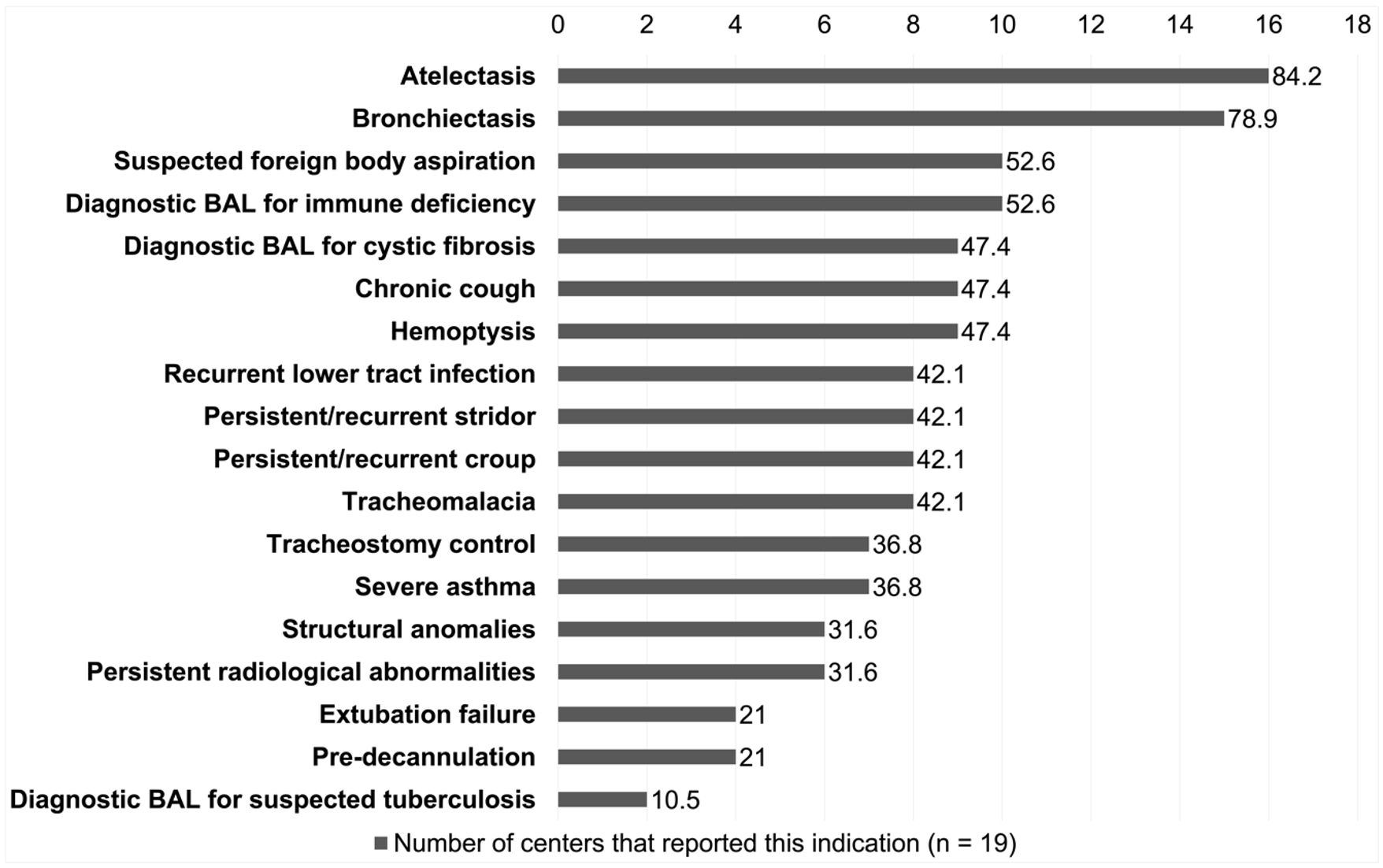
Parameters related to bronchoscopy are shown in Table I. The most frequently reported indications were atelectasis (16 centers, 84.2%), bronchiectasis (15 centers, 78.9%), suspected foreign body aspiration (10 centers, 52.6%), and obtaining BAL in patients with immune deficiency (10 centers, 52.6%) (Fig. 2).
|
Percentages are based on 19 centers unless otherwise specified. *“Fiberoptic bronchoscope” refers to traditional scopes using optical fiber bundles for illumination and image transmission, “video bronchoscope” denotes scopes with a built-in digital camera and processor, and “video-assisted fiberoptic bronchoscope” describes a fiberoptic model connected to an external camera for monitor viewing. ** “Endobronchial biopsy” refers to sampling that includes the bronchial wall and submucosa for structural/pathologic evaluation, whereas “mucosal biopsy” denotes a superficial epithelial sampling primarily for histopathologic or inflammatory assessment. *** na (available) = the test could be performed at the center when clinically indicated; nr (routine) = the test was regularly performed in all BAL samples as part of standard diagnostic practice. %a represents the proportion of centers with test availability among all 19 centers (%a = na/19). %r represents the proportion of centers performing the test routinely among those in which the test was available (%r = nr/na). BAL: Bronchoalveolar lavage |
||
| Table I. Data about bronchoscopy and BAL method. | ||
| Variables |
|
|
| Frequency of bronchoscopy sessions | ||
| Once a week |
|
|
| 2-3 times a week |
|
|
| Every day |
|
|
| Type of bronchoscopes* | ||
| Fiberoptic bronchoscope |
|
|
| Video bronchoscope |
|
|
| Video- assisted fiberoptic bronchoscope |
|
|
| Diagnostic procedures** | ||
| Evaluation of upper and lower airway |
|
|
| BAL |
|
|
| Endobronchial biopsy |
|
|
| Mucosal biopsy |
|
|
| Transbronchial biopsy |
|
|
| Therapeutic interventions | ||
| Removal of mucus plug |
|
|
| Partial lung lavage |
|
|
| Whole lung lavage |
|
|
| Bronchoscopic intubation |
|
|
| Foreign body removal |
|
|
| Tracheal/bronchial stent insertion |
|
|
| Balloon dilatation |
|
|
| Dominant hand holding the bronchoscope | ||
| Right |
|
|
| Left |
|
|
| Either |
|
|
| Route of bronchoscope insertion | ||
| Via the laryngeal mask airway |
|
|
| Via the nasal route |
|
|
| Via the oral route without an airway device |
|
|
| Routes of oxygen supplementation | ||
| Via nasal cannula |
|
|
| Via mechanic ventilation |
|
|
| Quantity of saline used for BAL in each aliquot | ||
| 10 mL |
|
|
| 10-20 mL |
|
|
| 1 mL/kg, max 10 mL |
|
|
| 1 mL/kg, max 20 mL |
|
|
| Routine BAL segments in non-focal disease | ||
| Right middle lobe and lingula |
|
|
| Right middle lobe |
|
|
| Instruments used to aspirate BAL fluid | ||
| Electric portable machine |
|
|
| Wall mounted |
|
|
| 50 mL syringe |
|
|
| Availability and routine use of BAL fluid analyses |
|
|
| Bacterial culture |
|
|
| Cytology |
|
|
| Cultures for tuberculosis |
|
|
| Fungal culture |
|
|
| Lipid-laden alveolar macrophages |
|
|
| Lymphocyte panel (immunology) |
|
|
| Differential cell counts |
|
|
| Histopathology |
|
|
| Virology |
|
|
| Hemosiderin-laden macrophages |
|
|
Interventional bronchoscopy
Based on participant reports, therapeutic FB constituted a median 35% (range 2–80%) of all FB procedures, indicating that diagnostic FB was performed approximately twice as often as therapeutic FB. Moreover, of the nine centers (47.4%) indicating that they can perform advanced bronchoscopic procedures, endobronchial and mucosal biopsy was reported by six centers (31.6%), bronchoscopic intubations by five centers (26.3%), foreign body removal by three centers (15.8%), and tracheal/bronchial stent insertion by two centers (10.5%). Detailed information on bronchoscopic interventions is presented in Table I.
Equipment and personnel
Sixteen centers (84.2%) used video-assisted flexible bronchoscopes. Every center had at least two bronchoscopes with different outer diameters (ODs). Seven centers (36.8%) had three different ODs available, while one center (5.3%) had four. The three commonly reported bronchoscope ODs were 4.9 mm (12 centers, 63.2%), 3.6 mm (6 centers, 31.6%), and 2.8 mm (5 centers, 26.3%).
When the availability of experienced, dedicated staff for pediatric FB (nurses, anesthesiologists, and anesthesia technicians) was assessed across 19 centers, three (15.8%) reported having none, four (21.1%) had a nurse only, one (5.3%) had an anesthesiologist only, one (5.3%) had an anesthesia technician only, three (15.8%) had a nurse and an anesthesiologist, and seven (36.8%) had all three dedicated staff members.
Twelve centers (63.2%) reported that bronchoscopies were mainly performed in operating rooms; in four centers (21.1%) bronchoscopies were carried out in bronchoscopy suites, and one center (5.3%) used an airway endoscopy suite. In two centers, more than one site was available. Additionally, nine centers (47.4%) performed FB for critically ill patients in the ICU.
Preprocedural evaluations
Routine preprocedural testing was conducted in 18 centers (94.7%): complete blood count was obtained in 16 centers (84.2%), coagulation parameters in 14 centers (73.7%), chest X-ray in 12 centers (63.2%), pulmonary function tests in four centers (21.1%), and serologic testing for hepatitis viruses, syphilis, and Human immunodeficiency virus (HIV) antibodies in four centers (21.1%). All respondents confirmed obtaining written informed consent from caregivers before FB. Additionally, 13 centers (68.4%) required notifying the bronchoscopy team or operating room personnel at least one day before the procedure.
Bronchoalveolar lavage
During FB, the most commonly used personal protective equipment included sterile gloves (89.5%), N95 masks (52.6%), and safety glasses (36.8%). Additionally, 63.2% of respondents reported performing the procedure while positioned behind the patient’s head—near the airway—facing the bronchoscopy monitor. The remaining respondents reported standing at the patient’s side during the procedure.
The aliquot counts for microbiological and cytological examinations were two (8 centers, 42.1%), three (5 centers, 26.3%), six (3 centers, 15.8%), one (2 centers, 10.5%), and four (1 center, 5.3%). Table I illustrates the variations in BAL techniques, and BAL fluid analysis. Cytology and tuberculosis tests were available in all centers; however, they were performed routinely in 11 centers (57.9%) and 13 centers (68.4%), respectively.
Cleaning and disinfection of bronchoscopes
For bronchoscope reprocessing, automated cleaning was reported by nine centers (47.4%), automated disinfection by 10 centers (52.6%), and manual drying by nine centers (47.4%) (Table II). Moreover, 15 centers (78.9%) sterilized the bronchoscopes and 14 centers (73.7%) sent random bacterial surveillance culture samples to reference laboratories.
| Percentages are based on 19 centers, unless otherwise specified. | ||
| Table II. Cleaning, disinfection and storage of bronchoscopes. | ||
| Variables |
|
|
| Cleaning methods of bronchoscopes | ||
| Manual |
|
|
| Automated |
|
|
| Both |
|
|
| Disinfection methods of bronchoscopes | ||
| Manual |
|
|
| Automated |
|
|
| Both |
|
|
| Methods of drying bronchoscopes | ||
| Manual |
|
|
| Compressed air |
|
|
| Bronchoscope drying cabinet |
|
|
| 70% alcohol solution |
|
|
| Storage containers for bronchoscopes | ||
| Storage cabinet |
|
|
| Bronchoscope suitcase |
|
|
| Sterile surgical drape |
|
|
| Disinfectant solutions used for manual disinfection of bronchoscopes (n = 9) | ||
| Peracetic acid and hydrogen peroxide |
|
|
| 2% glutaraldehyde solution |
|
|
| Peracetic acid |
|
|
Premedication and sedation
Thirteen centers (68.4%) implemented premedication, with considerable variability in drug selection, procedural approaches, and patient monitoring methods, as shown in Table III. Three centers (15.8%) routinely administered inhaled bronchodilators as preprocedural medication, while one center (5.3%) additionally used intranasal adrenergic drops, and another center (5.3%) used intravenous antihistamines in addition to bronchodilators. Pre-oxygenation was routinely performed prior to general anesthesia (GA). Among the three centers using moderate conscious sedation, two reported performing pre-oxygenation.
|
Percentages are based on 19 centers unless otherwise specified. *Some centers use more than one drug for premedication. ** Used in selected patients (e.g., with asthma or airway hyperreactivity). *** Sedation adequacy was self-reported. “Always sufficient” = clinically acceptable depth without escalation in nearly all cases; “Often sufficient” = clinically acceptable depth without escalation in most cases. ECG; electrocardiography, EtCO2; end-tidal CO₂, NIBP; non-invasive blood pressure, SPO2; oxygen saturation measured using pulse oxymetry. |
||
| Table III. Methods of premedication and sedation. | ||
| Variables |
|
|
| Premedication given by (n = 13) | ||
| Anesthesiologist |
|
|
| Nurse |
|
|
| Anesthesia technician |
|
|
| Pediatric pulmonologist |
|
|
| Drugs used for premedication (n = 13)* | ||
| Midazolam |
|
|
| Lidocaine |
|
|
| Methylprednisolone (as needed) ** |
|
|
| Fentanyl |
|
|
| Ketamine |
|
|
| Routinely used medications before procedure | ||
| None |
|
|
| Inhaled bronchodilators |
|
|
| Inhaled bronchodilators + Intranasal adrenergic drops |
|
|
| Inhaled bronchodilators + Intravenous antihistamines |
|
|
| Methods of sedation | ||
| Moderate conscious sedation combined with local anesthesia |
|
|
| General anesthesia |
|
|
| General anesthesia combined with local anesthesia |
|
|
| Drugs used for induction of anesthesia | ||
| Propofol |
|
|
| Midazolam |
|
|
| Fentanyl |
|
|
| Remifentanil |
|
|
| Ketamine |
|
|
| Atropine |
|
|
| Lidocaine |
|
|
| Rocuronium |
|
|
| Drugs used for maintenance of anesthesia | ||
| Sevoflurane alone |
|
|
| Propofol infusion alone |
|
|
| Propofol infusion + sevoflurane |
|
|
| Propofol infusion + remifentanil infusion |
|
|
| Topical lidocaine applications (n = 12) | ||
| Pharyngeal |
|
|
| Intratracheal |
|
|
| Pharyngeal plus intratracheal |
|
|
| Combined used intranasal + pharyngeal + intratracheal |
|
|
| Endotracheal lidocaine injection sites (n = 11) | ||
| Epiglottis |
|
|
| Carina |
|
|
| Routinely used medications after procedure | ||
| None |
|
|
| Nebulization with bronchodilator |
|
|
| Intravenous methylprednisolone |
|
|
| Nebulization with adrenalin |
|
|
| Cool mist |
|
|
| Parameters monitored during procedure | ||
| SpO2, ECG, EtCO2, NIBP |
|
|
| SpO2, ECG, EtCO2 |
|
|
| SpO2 |
|
|
| SpO2, ECG, NIBP |
|
|
| SpO2, ECG |
|
|
| Adequacy of sedation provided during the procedure*** | ||
| Always sufficient |
|
|
| Often sufficient |
|
|
Anesthesiologists evaluated patients the day before the procedure in 16 centers (84.2%), and on the same day in three centers (15.8%). FB was predominantly performed under GA combined with local anesthesia (9 centers, 47.4%). However, three centers (15.8%) opted for moderate conscious sedation combined with lidocaine spray for local anesthesia. Of the 16 centers (84.2%) utilizing GA, nine (47.4%) also administered local anesthesia. Lidocaine was the sole agent used for local anesthesia, though the instillation sites varied (Table III).
Patient discharge
The discharge protocols for elective outpatient FB showed that four centers (21.1%) discharged patients the following day, whereas the remaining 15 (78.9%) performed same-day discharge—seven centers (36.8%) within 1–3 hours and eight centers (42.1%) within 6–12 hours post-procedure. None of the centers prescribed routine medications at discharge; however, five centers (26.3%) routinely performed a post-procedural chest X-ray.
Respondent feedback
Five respondents (26.3%) emphasized the need to enhance interventional procedures and increase the number of trained anesthesiologists and nurses. Additionally, three respondents (15.8%) advocated for the immediate development of national pediatric FB guidelines to standardize procedural approaches.
Discussion
This study presents the first nationwide epidemiologic overview of pediatric FB practices among pediatric pulmonologists in Türkiye. As all centers performing pediatric FB participated, our findings provide a realistic representation of current national practices. The survey revealed substantial heterogeneity in procedural approaches, highlighting the need for national standards to improve consistency, training, and patient outcomes.
Flexible bronchoscopy has been performed by pediatric pulmonologists in Türkiye for nearly three decades. However, the median number of procedures per center per year (210 in 19 centers) is markedly higher than that reported from Europe (35 in 198 centers), China (158 in 47 centers), and India (75 in 24 centers).2,8,9 A detailed comparison with international survey data is provided in Table IV. Despite the high number of procedures per center, only 10 out of 81 provinces offer FB, meaning each center serves about eight provinces. These findings indicate challenges in referral pathways and raise concerns about service capacity. Whether this high number of procedures in a few centers results from serving a large population or reflects advanced expertise will become clearer through future comparative studies.
| BAL; bronchoalveolar lavage, CBC; complete blood count, CXR; chest X-ray, ECG; electrocardiography, GA; general anesthesia, ICU; intensive care units, IQR; interquartile range, LA; local anesthesia, LLAM; lipid-laden alveolar macrophages, OD; outer diameter, PFT; pulmonary function test, SpO₂; oxygen saturation. | ||||
| Table IV. Comparison of pediatric flexible bronchoscopy (FB) practices: Türkiye vs. international multicenter surveys. | ||||
| Parameter | Schramm et al., 20172 | Lin et al., 20209 | Jat et al., 20228 | Present study, 2026 |
| Study year and country | 2015 and European countries | 2018 and Western China | 2019 and India | 2023 and Türkiye |
| Total centers and response rate |
198 centers in 33 countries Response rate 75% (33/44) |
47 centers in 32 cities Response rate 87% (47/54) |
24 centers in 14 cities Response rate 5.2% (24/455) |
19 centers in 10 cities Response rate 100% (19/19) |
| Performed FBs per center per year (median) | 96 | 158 | 75 | 210 |
| Main indication for FB |
-Aspiration (72.7%) -Infection (48.5%) -Radiographic abnormalities (48.5%) |
Pooled rate; -Atelectasis/lobar pneumonia (95% CI, 25-30%), -Persistent/recurrent wheeze (95% CI, 17-20%) -Chronic cough (95% CI, 11-15%) |
-Suspected tuberculosis (87.5%) -Atelectasis (87.5%) -Suspected airway malacia (83.3%) -Infection other than tuberculosis (83.3%) -Persistent/recurrent pneumonia (83.3%) |
-Atelectasis (84.2%) -Bronchiectasis (78.9%) -Immune deficiency (52.6%) -Suspected foreign body aspiration (52.6%) |
| Advanced bronchoscopic procedures |
-Endobronchial biopsy (73.2%) -Transbronchial biopsy (37.8%) -Balloon dilatation (21.2%) -Stent placement (9.5%) |
-Cryoablation (23.4%) -Balloon dilatation (21.2%) -Thermal ablation (17%) -Stent placement (0%) |
-Endobronchial biopsy (37.5%) - Transbronchial biopsy (12.5%) -Mucosal biopsy (16.6%) -Balloon dilatation (0%) -Stent placement (0%) -Bronchoscopic intubation (45.8%) -Foreign body removal (41%) |
-Endobronchial biopsy (31.6%) - Transbronchial biopsy (10.5%) -Mucosal biopsy (31.6%) -Balloon dilatation (5.3%) -Stent placement (10.5%) -Bronchoscopic intubation (26.3%) -Foreign body removal (15.8%) |
| Bronchoscope types and OD |
OD 4.9 mm and 3.6 mm were most common; counts/percentages not reported. Bronchoscope types not reported. |
Not reported |
OD not reported. -Video bronchoscope (41%) -Fiberoptic bronchoscope (33%) - Video- assisted fiberoptic bronchoscope (21%) |
OD 4.9 mm (63.2%), 3.6 mm (31.6%), and 2.8 mm (26.3%) were most common. - Video- assisted fiberoptic bronchoscope (84.2%) -Fiberoptic bronchoscope (10.5%) - Video bronchoscope (5.3 %) |
| Dedicated staff for pediatric FB | “Most of the centers reported three qualified bronchoscopists and two dedicated nurses.” (counts/percentages not reported). |
-47 centers (100%) had median 4 (IQR; 1-19) dedicated nurses. -24 centers (51.1%) had median 1 (IQR; 0–5) anesthesiologists. |
“Healthcare personnel involved in the procedure included a trained pediatric pulmonologist, trained nurse, pediatric resident, pediatric pulmonology fellow, laboratory technician, or anesthetist.” (counts/percentages not reported). |
-Nurse (21.1%) -Anesthesiologist (5.3%) -Anesthesia technician (5.3%) -Nurse and anesthesiologist (15.8%) - Nurse, anesthesiologist, and anesthesia technician (36.8%) -None (15.8%) |
| Site of bronchoscopy |
-Bronchoscopy suite (87.3%) -Operating room (74.7%) -ICU (62.1%) |
-ICU (66%) -Endoscopy suite (61.7%) -Bronchoscopy suite (4%) -More than one site (79.2%) |
-Bronchoscopy suite (58.3%) -Operating room (25%) -ICU (12.5%) -Endoscopy suite (4.2%) |
-Operating room (63.2%) -ICU (47.4%) -Bronchoscopy suite (21.1%) -Endoscopy suite (5.3%) -more than one site (10.5%) |
| Routine pre-procedure tests | Not reported |
-CBC (100%) -CXR (97.9%) -Coagulation function (97.9%) -PFT (40%) -Serologic testing for hepatitis viruses, syphilis, and HIV antibodies (100%) |
Not reported |
-None (5.3%) -CBC (84.2%) -Coagulation function (73.7%) -CXR (63.2%) -PFT (21.1%) -Serologic testing for hepatitis viruses, syphilis, and HIV antibodies 21.1% |
| Routine preprocedural medication | Not reported | Not reported |
-Atropine (16.7%) -Nasal adrenergic drops (54.2%) |
-Bronchodilator (26.3%) -Nasal adrenergic drops (5.3%) -Intravenous antihistamines (5.3%) |
| Methods of anesthesia | “Usually performed under GA or moderate sedation/analgesia.” (counts/percentages not reported). |
-Conscious sedation combined with LA (63.8%) -GA with tracheal intubation (42.6%) -GA with LMA (44.7%) -GA with HFV (2.1%) |
-Conscious sedation (79%) -GA (8.4%) -Conscious sedation or GA (12.5%) |
-GA combined with LA (47.4%) -GA (36.8%) -Conscious sedation combined with LA (15.8%) |
| GA medication |
“The main intravenous anesthetic was propofol, the main inhalation anesthetic was sevoflurane.” (counts/percentages not reported). |
-Midazolam (95.8%) -Propofol (31.9%) -Fentanyl (44.7%) |
-Midazolam + fentanyl (37.5%) -Midazolam + propofol (20.8%) |
-Propofol (89.5%) -Sevoflurane (73.7%) -Midazolam (57.9%) -Fentanyl (31.6%) |
| Local anesthesia |
“Local anesthesia during GA was most often provided by lidocaine.” (counts/percentages not reported). |
-Lidocaine 77.1% (sites did not reported) |
-Spray as go lignocaine (71%) -Both nebulized and spray as go lignocaine (25%) -Nebulized lignocaine (4.2%) |
12 centers (63.2%) used lidocaine for LA; -Pharyngeal + intratracheal (41.7%) -Intratracheal (33.3%) -Intranasal + pharyngeal + intratracheal (16.7%) |
| Routine monitoring | Not reported |
-SpO₂ 100% -ECG 100% |
-SpO₂ 100% -Blood pressure 21% -ECG 33.3% |
-SpO₂ 100% -Blood pressure 52.6% -ECG 78.9% -EtCO2 57.9% |
| Availability or routine of BAL |
Availability; -Microbiology (93.9%) -Cytology (82.8%) -Virology (73.7%) -Immunology (70.7%) -Differential cell count (82.8%) -LLAM (54.5%) |
Routine; -Bacterial culture (95.7%) -Cytology (80.9%) -PCR (61.7%) -Histopathological examination for bronchial mucosa (55.3%) |
Not reported |
Availability and routine; -Bacterial culture (100% and 100%) -Cytology (100%, and 57.9%) -Virology (78.9% and 13.3%) -Immunology (89.5% and 11.8%) - Differential cell count (89.5% and 76.5%) -LLAM (94.7% and 50%) |
| BAL aliquots | Not reported | Not reported |
3–4: 8 (33.3%) 2–3: 5 (20.8%) |
Median 2 (range 1–6) |
In our survey, atelectasis, bronchiectasis, suspected foreign body aspiration, and immune deficiency (to obtain BAL for microbiological evaluation) were the most common indications for FB. Compared with European data, FB for immunodeficient patients was more frequently reported in Türkiye.2 This finding may be explained by the higher prevalence of primary immunodeficiencies in regions with increased consanguinity.12,13 A recent study in Türkiye assessed the diagnostic value of FB and BAL in immunosuppressed children, revealing that microscopic examination of BAL specimens identified pathogens in 65.1% of cases despite prior antimicrobial therapy.14 Diagnostic complexity due to pathogen diversity underscores the importance of FB in immunosuppressed children with respiratory symptoms.10,15-20
Advanced interventional procedures were performed less frequently than in European centers, particularly bronchoscopic intubation and transbronchial biopsy.2 While pediatric airway anatomy presents unique challenges, international recommendations emphasize that such procedures should be performed by experienced pediatric bronchoscopists.3 As pediatric pulmonology continues to evolve in Türkiye, the frequency of these procedures is expected to rise with an increasing number of pediatric pulmonologists, well-trained bronchoscopy teams, and the development of national pediatric FB guidelines.2,21
There is no consensus on the optimal sedation method for pediatric FB.1,6-8,10 Mild sedation with topical anesthetics may be sufficient22; however, deeper sedation is often required to suppress cough during FB.23 Although the use of GA has increased with advances in anesthetic techniques6, it may mask dynamic airway abnormalities such as tracheomalacia and bronchomalacia. Our survey found that GA combined with local anesthesia is the most preferred approach. The benefit of combining topical local anesthesia with GA during FB remains controversial, as available evidence shows no clear efficacy in cough suppression and suggests a potential increase in perioperative respiratory adverse events.24,25 Given the frequent use of GA in our country, a multi-center randomized study could assess the efficacy of local anesthesia in FB.
In our study, propofol was the most frequently used agent for both induction and maintenance of anesthesia, consistent with its established safety and efficacy in pediatric FB.23,26 Among 13 centers using propofol for maintenance, eight combined it with sevoflurane, and only one used it with remifentanil. However, evidence remains inconclusive regarding its optimal use alone or in combination27,28, Sedation methods and agent preferences varied widely across centers and international studies.2,8,9 This study highlights differences in anesthesia practices due to varying experiences among bronchoscopists and anesthesiologists. We believe that our findings may contribute to individualized sedation strategies and support efforts toward standardizing FB practices.
Premedication was routinely used in most centers, with midazolam being the preferred agent. Although there is no consensus on premedication use in pediatric FB1,6-8,10, previous studies have examined the effects of premedication agents on cough, anxiety, and recovery time.26-34 Midazolam has been shown to improve procedural tolerance and reduce the incidence and severity of the cough reflex.26,31,35 In addition, combining midazolam with propofol has been shown to decrease propofol’s induction dose, reducing its cardiovascular and respiratory side effects.36,37 Minimizing discomfort before FB improves both child and parental satisfaction, making premedication particularly beneficial in anxious or uncooperative children.
The optimal number of aliquots, saline volume per aliquot, and sampling sites for BAL remain unclear in the literature.1,6,10 Our findings align with general recommendations, suggesting 1 mL/kg per aliquot (maximum 20 mL) and favoring the right middle lobe and lingula for sampling.1,38 Routine cytological and microbiological analyses were inconsistently performed despite guideline recommendations.1,7 Therefore, a national FB guideline is needed to standardize BAL fluid practices and improve diagnostic yield.
Bronchoscope reprocessing methods also differed, with both manual and automated approaches in use. While most centers performed microbiological surveillance cultures, evidence supporting their routine use remains limited.1,39 Alcohol flushing, which was rarely used, is known to reduce positive culture rates.40 Since bacterial surveillance cultures are time-consuming and costly, alcohol flushing may offer a practical alternative to reduce contamination and drying time.
This study has several limitations. First, self-reported responses introduce potential bias due to differences in interpretation, recall, and reporting. Although we captured anesthesia practices at a center level, the survey did not ask GA-using centers how they modify their approach when dynamic airway disease is suspected, such as preserving spontaneous breathing or opting for minimal sedation. Additionally, the questionnaire did not include details on suction pressures or fluid retrieval percentages during BAL, both of which are essential for diagnostic accuracy. The lack of clarity regarding sample handling—whether initial or subsequent samples were analyzed according to recommended standards—may also impact diagnostic reliability.
In conclusion, current pediatric FB practices in Türkiye demonstrate significant variability across centers, particularly in sedation protocols, interventional procedures, BAL techniques, and reprocessing methods. Moreover, comparison with survey results from other countries allowed us to observe both similarities and differences in procedural approaches. These findings highlight the need for standardized national guidelines, expanded training programs, and multidisciplinary collaboration to improve procedural quality and patient safety.
Acknowledgements
We sincerely thank all participating centers and the clinical directors who completed the national survey and provided valuable data for this study. We are especially grateful to Ayşe Tana Aslan, Ayşen Bingöl, Güzin Cinel, Haluk Çokuğraş, Bülent Karadağ, and Sedat Öktem, the center directors who contributed data but were not involved in data analysis or manuscript preparation. We also appreciate Gülçin Kabaçam, MSc in preparing the figure for the map demonstrating centers performing FB in Turkey.
Ethical approval
The study was approved by Ege University Faculty of Medicine Ethics Committee (date: November 5, 2023, number: 23-5T/40).
Source of funding
The authors declare the study received no funding.
Conflict of interest
The authors declare that there is no conflict of interest.
References
- Faro A, Wood RE, Schechter MS, et al. Official American Thoracic Society technical standards: flexible airway endoscopy in children. Am J Respir Crit Care Med 2015; 191: 1066-1080. https://doi.org/10.1164/rccm.201503-0474ST
- Schramm D, Yu Y, Wiemers A, et al. Pediatric flexible and rigid bronchoscopy in European centers-Availability and current practice. Pediatr Pulmonol 2017; 52: 1502-1508. https://doi.org/10.1002/ppul.23823
- Eber E, Antón-Pacheco JL, de Blic J, et al. ERS statement: interventional bronchoscopy in children. Eur Respir J 2017; 50: 1700901. https://doi.org/10.1183/13993003.00901-2017
- Atag E, Unal F, Yazan H, et al. Pediatric flexible bronchoscopy in the intensive care unit: A multicenter study. Pediatr Pulmonol 2021; 56: 2925-2931. https://doi.org/10.1002/ppul.25566
- Rosner EA, L Parker J, Vandenberg C, et al. Flexible bronchoscopy in pediatric venovenous extracorporeal membrane oxygenation. Respir Care 2022; 67: 688-693. https://doi.org/10.4187/respcare.09243
- Goussard P, Pohunek P, Eber E, et al. Pediatric bronchoscopy: recent advances and clinical challenges. Expert Rev Respir Med 2021; 15: 453-475. https://doi.org/10.1080/17476348.2021.1882854
- Du Rand IA, Blaikley J, Booton R, et al. British Thoracic Society guideline for diagnostic flexible bronchoscopy in adults: accredited by NICE. Thorax 2013; 68(Suppl 1): i1-i44. https://doi.org/10.1136/thoraxjnl-2013-203618
- Jat KR, Agarwal S, Lodha R, Kabra SK. A survey of pediatric flexible bronchoscopy in India. Pediatr Pulmonol 2022; 57: 2674-2680. https://doi.org/10.1002/ppul.26081
- Lin J, Tao X, Xia W, et al. A multicenter survey of pediatric flexible bronchoscopy in western China. Transl Pediatr 2021; 10: 83-91. https://doi.org/10.21037/tp-20-244
- Pérez-Frías J, Moreno Galdó A, Pérez Ruiz E, et al. Pediatric bronchoscopy guidelines. Arch Bronconeumol 2011; 47: 350-360. https://doi.org/10.1016/j.arbres.2011.04.003
- Barbato A, Magarotto M, Crivellaro M, et al. Use of the paediatric bronchoscope, flexible and rigid, in 51 European centres. Eur Respir J 1997; 10: 1761-1766. https://doi.org/10.1183/09031936.97.10081761
- Al-Mousa H, Al-Saud B. Primary Immunodeficiency Diseases in Highly Consanguineous Populations from Middle East and North Africa: Epidemiology, Diagnosis, and Care. Front Immunol 2017; 8: 678. https://doi.org/10.3389/fimmu.2017.00678
- Al-Herz W, Aldhekri H, Barbouche MR, Rezaei N. Consanguinity and primary immunodeficiencies. Hum Hered 2014; 77: 138-143. https://doi.org/10.1159/000357710
- Vardhana Rao MV, Sahu D, Nair S, et al. National guidelines for data quality in surveys: An overview. Indian J Med Res 2022; 156: 715-720. https://doi.org/10.4103/ijmr.ijmr_1261_22
- Eroglu-Ertugrul NG, Yalcin E, Oguz B, et al. The value of flexible bronchoscopy in pulmonary infections of immunosuppressed children. Clin Respir J 2020; 14: 78-84. https://doi.org/10.1111/crj.13103
- Deotare U, Merman E, Pincus D, et al. The utility and safety of flexible bronchoscopy in critically ill acute leukemia patients: a retrospective cohort study. Can J Anaesth 2018; 65: 272-279. https://doi.org/10.1007/s12630-017-1041-7
- Rizik S, Hakim F, Bentur L, Arad-Cohen N, Kassis I. Bronchoscopy and Bronchoalveolar Lavage in the Diagnosis and Management of Pulmonary Infections in Immunocompromised Children. J Pediatr Hematol Oncol 2018; 40: 532-535. https://doi.org/10.1097/MPH.0000000000001283
- Soyer T. The role bronchoscopy in the diagnosis of airway disease in children. J Thorac Dis 2016; 8: 3420-3426. https://doi.org/10.21037/jtd.2016.11.87
- Wood RE. The emerging role of flexible bronchoscopy in pediatrics. Clin Chest Med 2001; 22: 311-7, viii. https://doi.org/10.1016/s0272-5231(05)70045-9
- Demir E, Ozaslan M. Bronkoalveolar lavaj ve endikasyonlar [Bronchoalveolar lavage and indications]. In: Yüksel H, Bingöl A, Gülen F, editors. Pediatrik Fleksibl Bronkoskopi. 1st ed. Ankara: Nobel Tıp Kitabevleri; 2022: [212-217]. [Turkish].
- Wahidi MM, Jain P, Jantz M, et al. American College of Chest Physicians consensus statement on the use of topical anesthesia, analgesia, and sedation during flexible bronchoscopy in adult patients. Chest 2011; 140: 1342-1350. https://doi.org/10.1378/chest.10-3361
- Londino AV, Jagannathan N. Anesthesia in diagnostic and therapeutic pediatric bronchoscopy. Otolaryngol Clin North Am 2019; 52: 1037-1048. https://doi.org/10.1016/j.otc.2019.08.005
- Berkenbosch JW, Graff GR, Stark JM, Ner Z, Tobias JD. Use of a remifentanil-propofol mixture for pediatric flexible fiberoptic bronchoscopy sedation. Paediatr Anaesth 2004; 14: 941-946. https://doi.org/10.1111/j.1460-9592.2004.01355.x
- Augstkalns I, Bradshaw EG. The value of topical lignocaine for bronchoscopy under general anaesthesia. Anaesthesia 1977; 32: 367-370. https://doi.org/10.1111/j.1365-2044.1977.tb11641.x
- Roberts MH, Gildersleve CD. Lignocaine topicalization of the pediatric airway. Paediatr Anaesth 2016; 26: 337-344. https://doi.org/10.1111/pan.12868
- Reyle-Hahn M, Niggemann B, Max M, Streich R, Rossaint R. Remifentanil and propofol for sedation in children and young adolescents undergoing diagnostic flexible bronchoscopy. Paediatr Anaesth 2000; 10: 59-63. https://doi.org/10.1046/j.1460-9592.2000.00440.x
- Chen L, Yu L, Fan Y, Manyande A. A comparison between total intravenous anaesthesia using propofol plus remifentanil and volatile induction/ maintenance of anaesthesia using sevoflurane in children undergoing flexible fibreoptic bronchoscopy. Anaesth Intensive Care 2013; 41: 742-749. https://doi.org/10.1177/0310057X1304100609
- Costi D, Cyna AM, Ahmed S, et al. Effects of sevoflurane versus other general anaesthesia on emergence agitation in children. Cochrane Database Syst Rev 2014; 2014: CD007084. https://doi.org/10.1002/14651858.CD007084.pub2
- Houghton CM, Raghuram A, Sullivan PJ, O’Driscoll R. Pre-medication for bronchoscopy: a randomised double blind trial comparing alfentanil with midazolam. Respir Med 2004; 98: 1102-1107. https://doi.org/10.1016/j.rmed.2004.03.023
- Poi PJ, Chuah SY, Srinivas P, Liam CK. Common fears of patients undergoing bronchoscopy. Eur Respir J 1998; 11: 1147-1149. https://doi.org/10.1183/09031936.98.11051147
- Putinati S, Ballerin L, Corbetta L, Trevisani L, Potena A. Patient satisfaction with conscious sedation for bronchoscopy. Chest 1999; 115: 1437-1440. https://doi.org/10.1378/chest.115.5.1437
- Panigrahi MK, Datta A, Bhuniya S, Bal SK, Mohapatra PR. Dextromethorphan premedication in the alleviation of cough during flexible bronchoscopy in adults: A randomized double-blind placebo-controlled trial. Respirology 2023; 28: 484-490. https://doi.org/10.1111/resp.14445
- Antoniades N, Worsnop C. Topical lidocaine through the bronchoscope reduces cough rate during bronchoscopy. Respirology 2009; 14: 873-876. https://doi.org/10.1111/j.1440-1843.2009.01587.x
- Bi Y, Ma Y, Ni J, Wu L. Efficacy of premedication with intranasal dexmedetomidine for removal of inhaled foreign bodies in children by flexible fiberoptic bronchoscopy: a randomized, double-blind, placebo-controlled clinical trial. BMC Anesthesiol 2019; 19: 219. https://doi.org/10.1186/s12871-019-0892-6
- Maltais F, Laberge F, Laviolette M. A randomized, double-blind, placebo-controlled study of lorazepam as premedication for bronchoscopy. Chest 1996; 109: 1195-1198. https://doi.org/10.1378/chest.109.5.1195
- Short TG, Chui PT. Propofol and midazolam act synergistically in combination. Br J Anaesth 1991; 67: 539-545. https://doi.org/10.1093/bja/67.5.539
- Cressey DM, Claydon P, Bhaskaran NC, Reilly CS. Effect of midazolam pretreatment on induction dose requirements of propofol in combination with fentanyl in younger and older adults. Anaesthesia 2001; 56: 108-113. https://doi.org/10.1046/j.1365-2044.2001.01789.x
- Meyer KC, Raghu G, Baughman RP, et al. An official American Thoracic Society clinical practice guideline: the clinical utility of bronchoalveolar lavage cellular analysis in interstitial lung disease. Am J Respir Crit Care Med 2012; 185: 1004-1014. https://doi.org/10.1164/rccm.201202-0320ST
- Tunuguntla A, Sullivan MJ. Monitoring quality of flexible endoscope disinfection by microbiologic surveillance cultures. Tenn Med 2004; 97: 453-456.
- Gavaldà L, Olmo AR, Hernández R, et al. Microbiological monitoring of flexible bronchoscopes after high-level disinfection and flushing channels with alcohol: Results and costs. Respir Med 2015; 109: 1079-1085. https://doi.org/10.1016/j.rmed.2015.04.015
License
Copyright (c) 2026 The Author(s). This is an open access article distributed under the Creative Commons Attribution License (CC BY), which permits unrestricted use, distribution, and reproduction in any medium or format, provided the original work is properly cited.















