Abstract
Background. The coexistence of rheumatologic diseases (RD) and inflammatory bowel disease (IBD) in children remains underexplored. This study aimed to assess the frequency of RD and its clinical impact in pediatric IBD patients.
Methods. This retrospective cohort study included pediatric IBD patients followed at the Department of Pediatric Gastroenterology, Hepatology and Nutrition at Hacettepe University, Ankara, Türkiye between November 2008 and December 2020. Demographic characteristics, disease activity scores, inflammatory markers, and treatment modalities were compared between patients with and without concomitant RD; an additional analysis was performed in the familial Mediterranean fever (FMF) subgroup.
Results. A total of 88 patients (35 females, 53 males; median age 14.6 years) were analyzed. RD was identified in 28 patients (31.8%), with FMF being the most frequent (19/28, 67.9%; overall 21.6%). Although patients with RD had lower disease activity at diagnosis (p = 0.010), they required more frequent biologic therapy during follow-up (35.7% vs. 16.7%, p = 0.047). In the FMF subgroup, disease activity scores were significantly lower at diagnosis and higher at the last follow-up compared with patients without RD. There were no significant differences in inflammatory markers between the groups at diagnosis and last follow-up.
Conclusions. RD, particularly FMF, is commonly observed in pediatric IBD, with a prevalence of 31.8% for RD and 21.6% for FMF. The presence of RD is associated with an increased need for biologic therapy despite lower initial disease activity. Children with IBD should be systematically evaluated for RD, especially in regions with high MEFV mutation prevalence, to support more personalized management strategies.
Keywords: biologic therapy, familial Mediterranean fever, inflammatory bowel disease, rheumatologic diseases
Introduction
Inflammatory bowel disease (IBD), including Crohn’s disease (CD), ulcerative colitis (UC), and unclassified inflammatory bowel disease (IBDU), represents a chronic inflammatory condition affecting the gastrointestinal tract.1 Pediatric IBD is frequently associated with extraintestinal manifestations, particularly rheumatologic diseases (RD), such as familial Mediterranean fever (FMF) and enthesitis-related arthritis (ERA) among the juvenile idiopathic arthritis (JIA) subgroups.2
FMF is an inherited autoinflammatory disorder, especially common in populations from the Mediterranean region, and is characterized by recurrent febrile episodes, serositis, and systemic inflammation.3 Recent studies have reported a higher prevalence of MEFV gene mutations among pediatric IBD patients, suggesting a genetic predisposition that may influence disease phenotype and severity.4-7 In a Japanese cohort, MEFV-mutated pediatric IBDU patients demonstrated distinct mucosal phenotypes and responded favorably to colchicine, implying that MEFV-related autoinflammation may shape intestinal immune responses.8 In another cohort from Armenia, pediatric UC patients with coexisting FMF were reported to require earlier initiation of biologic therapy compared to those without FMF.9
Despite growing evidence, the clinical implications of FMF in pediatric IBD have not been fully elucidated. Whether the coexistence of FMF alters the disease course or affects treatment responsiveness remains unclear.
Therefore, this study aims to assess the frequency of FMF among pediatric IBD patients and to evaluate its impact on disease activity, inflammatory markers, and the need for biologic therapy. We hypothesize that the presence of FMF may be associated with an altered disease course and increased likelihood of biologic therapy.
Materials and Methods
This retrospective cohort study was conducted in the Department of Pediatric Gastroenterology, Hepatology and Nutrition at Hacettepe University, encompassing cases from November 2008 to December 2020. Pediatric patients diagnosed with IBD (CD, UC and IBDU) were included. IBD diagnosis was established based on clinical presentation, laboratory parameters, endoscopic appearance, histopathologic features, and radiologic findings, in accordance with the revised European Society for Paediatric Gastroenterology Hepatology and Nutrition (ESPGHAN) Porto criteria.10
Patients were categorized into two groups based on the presence or absence of coexisting RD, including FMF and JIA (including the ERA subtype), or other RD such as chronic recurrent multifocal osteomyelitis (CRMO), IgG4-related disease, or IgA vasculitis. The diagnosis of FMF was established according to the Eurofever/PRINTO criteria.11 JIA and ERA were identified using the International League of Associations for Rheumatology (ILAR) classification criteria12, while CRMO was diagnosed based on the Jansson criteria.13 IgA vasculitis was defined according to the EULAR/PRINTO/PRES Ankara 2008 criteria.14 The diagnosis of IgG4-related disease was based on the 2020 Revised Comprehensive Criteria for IgG4-RD.15
Data included demographic variables, clinical features, disease activity scores (Pediatric Crohn’s Disease Activity Index [PCDAI] and Pediatric Ulcerative Colitis Activity Index [PUCAI]), inflammatory markers (C-reactive protein [CRP], erythrocyte sedimentation rate [ESR], albumin, platelet count, fecal calprotectin), and treatment history, including biologic therapy.
Indications for initiating biologic therapy followed standard pediatric IBD practice consistent with ESPGHAN recommendations, including steroid-refractory or steroid-dependent disease, moderate-to-severe activity, fistulizing disease, or inadequate response to immunomodulators.
Statistical analyses were conducted using SPSS for Windows, version 22.0. Continuous variables were presented as median (Q1-Q3) or mean ± standard deviation based on data distribution, while categorical variables were expressed as counts and percentages. Between-group comparisons were performed using the Mann-Whitney U test for continuous variables and chi-square or Fisher’s exact test for categorical variables. A two-tailed p-value of <0.05 was considered statistically significant.
Given the retrospective and exploratory nature of the study, no a priori sample size or power calculation was performed. Due to variable data completeness, a formal post-hoc power analysis could not be applied to all parameters, and the results should therefore be interpreted with caution.
The study received approval from the Hacettepe University Institutional Review Board and was conducted in accordance with the ethical principles outlined in the Declaration of Helsinki. A waiver of informed consent was granted by the Board owing to the retrospective nature of the study.
Results
Patient demographics and clinical features
A total of 88 pediatric patients diagnosed with IBD were included, comprising 35 females (39.8%) and 53 males (60.2%) with a median age of 14.6 years (10.7-17.9 years). Disease subtypes included CD in 46 patients (52.3%), UC in 37 (42%), and IBDU in 5 (5.7%).
A family history of IBD was noted in 10 patients (11.4%), FMF in 3 (3.4%), and both diseases in 3 (3.4%). Parental consanguinity was present in 34 patients (38.6%), with no significant difference observed between the RD and non-RD groups (p = 0.93).
A comprehensive comparison of demographic features, disease subtype, baseline disease activity, inflammatory markers at diagnosis, time to biologic therapy, and treatment outcomes between patients with RD and those without RD is presented in Table I. A separate comparison between patients with FMF and those without RD is shown in Table II. The proportion of patients with CD was significantly higher in the RD group compared with patients without RD (71.4% vs. 43.3%, p = 0.024). Within the FMF subgroup, CD was also more frequent than in patients without RD (63.1% vs. 43.3%); however, this difference did not reach statistical significance (p = 0.101). Regarding disease activity, scores at diagnosis were significantly lower both in patients with RD compared with those without RD (p = 0.010) and in the FMF subgroup compared with patients without RD (p = 0.011). However, at the last follow-up, disease activity scores were significantly higher in the FMF group than in patients without RD (p = 0.015).
|
*Values are presented as median (Q1-Q3). †0=Remission, 1=Mild, 2=Moderate, 3=Severe Note: Some variables contain missing values; analyses reflect available cases. ADA: adalimumab, CRP: C-reactive protein, ESR: erythrocyte sedimentation rate, IBD: inflammatory bowel disease, IBDU: unclassified IBD, IFX: infliximab, RD: rheumatologic disease. |
|||
| Table I. Comparison of clinical and laboratory characteristics of pediatric inflammatory bowel disease patients with and without concomitant rheumatologic diseases | |||
|
|
|
|
|
| Age at IBD diagnosis, years* |
|
|
|
| Female sex, n (%) |
|
|
|
| Parental consanguinity, n (%) |
|
|
|
| IBD subtype, n (%) |
|
||
| Crohn’s disease |
|
|
|
| Ulcerative colitis |
|
|
|
| Unclassified (IBDU) |
|
|
|
| IBD activity at diagnosis*† |
|
|
|
| CRP at diagnosis, mg/dL* |
|
|
|
| ESR at diagnosis, mm/h* |
|
|
|
| Serum albumin at diagnosis, g/dL* |
|
|
|
| Platelet count at diagnosis, ×10³/µL* |
|
|
|
| Fecal calprotectin at diagnosis, µg/g* |
|
|
|
| Biologic therapy for IBD, n (%) |
|
|
|
| IFX |
|
|
|
| ADA |
|
|
|
| IFX→ADA |
|
|
|
| ADA→IFX |
|
|
|
| Time to use of biologics for IBD, months* |
|
|
|
| Outcome (IBD activity at last follow-up)* † |
|
|
|
|
*Values are presented as median (Q1-Q3). †0=Remission, 1=Mild, 2=Moderate, 3=Severe Note: Some variables contain missing values; analyses reflect available cases. ADA: adalimumab, CRP: C-reactive protein, ESR: erythrocyte sedimentation rate, FMF: familial Mediterranean fever, IBD: inflammatory bowel disease, IBDU: unclassified IBD, IFX: infliximab, RD: rheumatologic disease. |
|||
| Table II. Subgroup analysis comparing patients with familial Mediterranean fever to patients without rheumatologic disease | |||
|
|
|
|
|
| Age at IBD diagnosis, years* |
|
|
|
| Female sex, n (%) |
|
|
|
| Parental consanguinity, n (%) |
|
|
|
| IBD subtype, n (%) |
|
||
| Crohn’s disease |
|
|
|
| Ulcerative colitis |
|
|
|
| Unclassified (IBDU) |
|
|
|
| IBD activity at diagnosis*† |
|
|
|
| CRP at diagnosis, mg/dL* |
|
|
|
| ESR at diagnosis, mm/h* |
|
|
|
| Serum albumin at diagnosis, g/dL* |
|
|
|
| Platelet count at diagnosis, ×10³/µL* |
|
|
|
| Fecal calprotectin at diagnosis, µg/g* |
|
|
|
| Biologic therapy for IBD, n (%) |
|
|
|
| IFX |
|
|
|
| ADA |
|
|
|
| IFX→ADA |
|
|
|
| ADA→IFX |
|
|
|
| Time to use of biologics for IBD, months* |
|
|
|
| Outcome (IBD activity at last follow-up), median*† |
|
|
|
Inflammatory markers
At diagnosis, the median CRP level was 3.1 mg/dL (0.9-7.6), and the median ESR was 42 mm/h (20-58). At final follow-up, these values decreased to a median CRP of 0.32 mg/dL (0.16-0.80) and ESR of 13 mm/h (7-23.5), indicating overall improvement in inflammatory burden. Fecal calprotectin levels were available in 47 patients at diagnosis, with a median value of 300 µg/g (300-682), and in 10 patients at the final follow-up, with a median of 87.9 µg/g (2.5-326), supporting a reduction in intestinal inflammation over time. There were no statistically significant differences in CRP, ESR, or fecal calprotectin levels between patients with and without RD at either diagnosis or final follow-up.
Rheumatologic disease distribution
RD were identified in 28 of the 88 patients (31.8%), while the remaining 60 patients (68.2%) had no RD. FMF was the most common RD, observed in 19 patients, representing 21.6% of the total cohort and 67.9% of those with RD. Of the 19 patients with FMF, 11 had already been diagnosed with FMF and were receiving colchicine at the time of IBD diagnosis, whereas the remaining 8 patients were diagnosed with FMF after IBD onset and started colchicine thereafter. Among the 19 patients with FMF, MEFV mutation analysis revealed a predominance of pathogenic biallelic genotypes (52.6%), with M694V/M694V being the most frequent genotype (47.3%), followed by M694V/M680I (5.3%). Heterozygous pathogenic variants included M694V, M680I, and V726A. Variants of uncertain significance (VUS) included E148Q and A744S. The overall MEFV mutation spectrum is summarized in Table III, and individual patient-level genotypes are provided in Table IV.
| VUS: variant(s) of uncertain significance. | |
| Table III. MEFV mutation spectrum in familial Mediterranean fever patients | |
| MEFV mutation |
|
| Two pathogenic variants |
|
| M694V/M694V |
|
| M694V/M680I |
|
| One pathogenic variant |
|
| M694V/- |
|
| M680I/- |
|
| V726A/- |
|
| One pathogenic variant / one VUS | |
| M694V/E148Q |
|
| Two VUS | |
| E148Q/E148Q |
|
| One VUS | |
| A744S/- |
|
| Table IV. Individual MEFV genotypes of familial Mediterranean fever patients | ||
|
|
|
|
| Patient 1 |
|
|
| Patient 2 |
|
|
| Patient 3 |
|
|
| Patient 4 |
|
|
| Patient 5 |
|
|
| Patient 6 |
|
|
| Patient 7 |
|
|
| Patient 8 |
|
|
| Patient 9 |
|
|
| Patient 10 |
|
|
| Patient 11 |
|
|
| Patient 12 |
|
|
| Patient 13 |
|
|
| Patient 14 |
|
|
| Patient 15 |
|
|
| Patient 16 |
|
|
| Patient 17 |
|
|
| Patient 18 |
|
|
| Patient 19 |
|
|
FMF was diagnosed as an isolated condition in 14 patients (15.9%) and in combination with other RD in 5 patients (5.7%), including 4 with ERA and 1 with oligoarticular JIA. ERA was identified in 8 patients (9.1%), of whom 4 (4.5%) had isolated ERA and 4 (4.5%) had ERA coexisting with FMF. Other RDs included CRMO in 2 patients (2.3%), isolated oligoarticular JIA in 1 patient (1.1%), IgG4-related disease in 1 patient (1.1%), and IgA vasculitis in 1 patient (1.1%). The overlap between FMF, ERA, and oligoarticular JIA within the RD group is illustrated in Fig. 1.
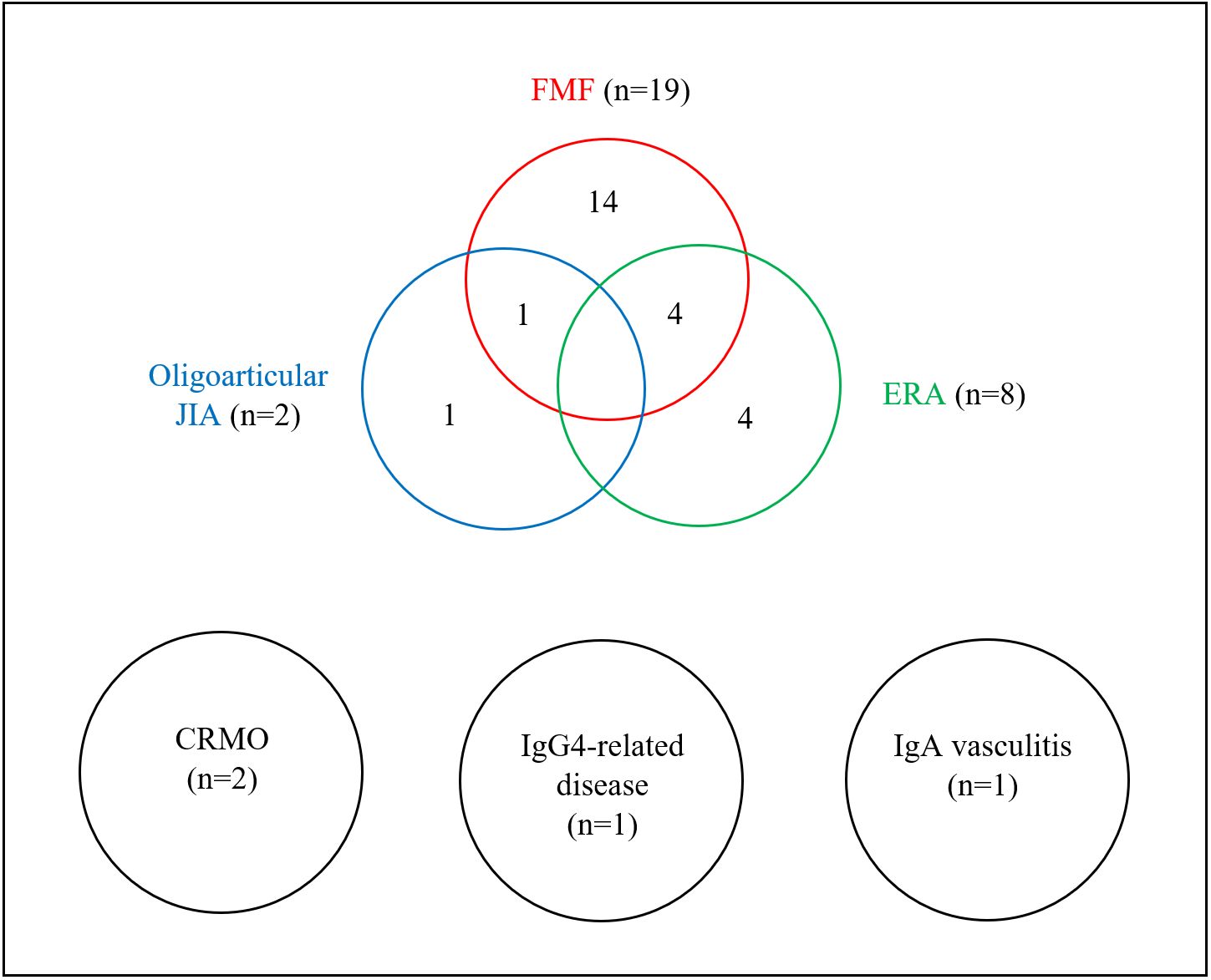
CRMO: chronic recurrent multifocal osteomyelitis, ERA: enthesitis-related arthritis, FMF: familial Mediterranean fever, JIA: juvenile idiopathic arthritis.
Biologic therapy for IBD
Biologic therapy for IBD was administered to 20 patients (22.7%) in the cohort. Among them, 10 received infliximab (IFX), 4 received adalimumab (ADA), 4 were switched from IFX to ADA, and 2 from ADA to IFX. To avoid misclassification, only biologic agents initiated specifically for IBD were included in this analysis; biologic therapies prescribed solely for rheumatologic indications were recorded separately.
Among patients with CD, 5 received IFX, 2 received ADA, 2 were switched from IFX to ADA, and 2 from ADA to IFX. In patients with UC, 5 received IFX, 2 received ADA, and 2 received sequential IFX followed by ADA. Biologic agents were used more frequently in patients with RD (10/28, 35.7%) compared to those without RD (10/60, 16.7%) (p = 0.047). In the FMF subgroup, the frequency of biologic therapy use was higher, although not statistically significant, compared with patients without RD (p = 0.194). Notably, 3 patients received biologic therapy for both IBD and RD: 1 with CD, FMF, and ERA; 1 with CD and FMF; and 1 with CD and ERA.
Biologic therapy for rheumatologic disease
Biologic therapy specifically targeting RD was administered to 8 patients (28.6%) within the RD group. Among these, 3 also required biologic agents for concomitant IBD, as previously noted.
Canakinumab was used in 3 patients, 1 of whom had previously received anakinra. One patient was switched from anakinra to IFX, and another was treated with IFX without prior biologics. ADA was administered to 2 patients, 1 following prior etanercept therapy and the other without any prior biologic treatment. Anakinra was used as initial therapy in 1 patient.
The primary indications for biologic therapy were FMF in 4 patients (3 treated with canakinumab and 1 with anakinra), ERA in 3 patients (1 treated with IFX and 2 with ADA), and coexisting FMF and ERA in 1 patient treated with IFX.
Discussion
This study investigated the prevalence and clinical impact of RD, particularly FMF, in children with IBD. We found that RD was present in approximately one-third of the cohort, with FMF being the most common, observed in 21.6% of all patients and 67.9% of those with concomitant RD. Although patients with RD had significantly lower disease activity scores at diagnosis, they were more likely to require biologic therapy during follow-up. Our findings align with previous studies reporting a high prevalence of FMF among pediatric IBD patients, particularly in populations with a high frequency of MEFV mutations, such as Turkey and Armenia.6,9 For example, Uslu et al. reported that 21.2% of Turkish children with IBD had FMF6, while Amaryan et al. observed an even higher prevalence in Armenian pediatric patients with UC.9 These results support a regional and possibly genetic predisposition for FMF-IBD coexistence. In our cohort, CD was significantly more common in patients with RD than in those without RD. A similar trend was observed in the FMF subgroup; however, this difference did not reach statistical significance.
The coexistence of FMF and IBD has also raised questions about whether MEFV mutations could modify the course of pediatric IBD. Several studies have investigated the prevalence of MEFV variants in IBD patients and reported significantly higher rates than in the general population.5,6 A multicenter Turkish study detected MEFV mutations in 41.9% of pediatric IBD patients, suggesting a possible genetic overlap or modifying effect.5
It has been proposed that MEFV mutations may influence the clinical course of IBD by enhancing innate immune activation, potentially contributing to treatment-refractory or atypical disease phenotypes.5,16 However, results across studies remain inconsistent. In our cohort, patients with FMF did not exhibit more severe inflammatory markers at diagnosis, and although the frequency of biologic therapy use was higher, this difference did not reach statistical significance. CRP and ESR were also similar between groups, which should be interpreted cautiously, given the relatively small sample size. Notably, however, disease activity scores at the last follow-up were significantly higher in the FMF group, suggesting that disease control over time may be more difficult to achieve in these patients despite comparable baseline inflammatory markers.
Although some reports have suggested that MEFV mutations may underlie atypical colitis phenotypes categorized as IBDU16, our study specifically included patients with confirmed FMF and identified different treatment patterns despite comparable baseline inflammatory markers. This observation is also consistent with a recent genetic study of our cohort, which identified MEFV mutations as the most frequent monogenic cause of early-onset IBD and highlighted their potential role in modifying disease phenotypes and possibly influencing treatment responses in children with IBD.17
In our cohort, patients with FMF presented with lower clinical disease activity scores at diagnosis compared with patients without RD. This finding may be partially explained by prior colchicine use, as 11 of the 19 FMF patients had been diagnosed with FMF before their IBD onset and were already receiving colchicine therapy. Colchicine, the cornerstone of FMF treatment, prevents attacks and suppresses subclinical inflammation through inhibition of microtubule polymerization, neutrophil activation, and downstream inflammatory pathways.3 Its sustained anti-inflammatory effects may mask FMF-related systemic symptoms and attenuate inflammatory biomarkers (e.g., CRP, ESR), potentially leading to underestimation of IBD activity by standard indices such as PCDAI.
Indeed, several case reports have documented infantile or early-onset colitis and CD that responded dramatically to colchicine initiation, highlighting its capacity to modify gastrointestinal inflammatory phenotypes.18-20 However, while colchicine may dampen clinical and laboratory signs, it does not necessarily prevent progressive intestinal inflammation, which may ultimately require escalation to biologic therapy.21 Therefore, the anti-inflammatory effects of colchicine should be recognized as a potential confounding factor that may obscure true disease activity and influence treatment decisions in patients with overlapping FMF and IBD.
The potential masking effect of colchicine complicates treatment decisions, especially in refractory cases. The management of patients with concomitant IBD and FMF can be challenging, particularly in those who respond inadequately to standard therapies. In our cohort, patients with RD had a higher likelihood of receiving biologic therapy for IBD, and several also required targeted treatment for FMF. IL-1 inhibitors such as anakinra and canakinumab were used in selected cases, particularly among patients with colchicine-resistant FMF. These findings align with previous reports supporting the effectiveness of IL-1 blockade in managing inflammation associated with both isolated FMF and FMF-IBD overlap syndromes.4,7
This study provides additional insight into the interplay between autoinflammatory conditions and pediatric IBD, suggesting that FMF may influence treatment decisions even in the absence of severe initial disease activity. Through a comprehensive analysis of clinical characteristics, inflammatory markers, and treatment patterns, we observed that FMF, as a frequent comorbidity, may be associated with certain differences in disease presentation and treatment approaches.
However, several limitations must be acknowledged. The relatively small cohort limits generalizability, particularly for less common rheumatologic conditions. Additionally, therapeutic decisions may have been influenced by clinician judgment and institutional protocols.
Despite these limitations, our findings highlight the importance of systematically screening for RD, particularly FMF, in pediatric IBD populations, especially in regions with a high prevalence of MEFV mutations. Conversely, clinicians should consider underlying IBD in children with known RD who present with chronic diarrhea, abdominal pain, or growth delay. Future prospective, multicenter studies incorporating genetic analyses and long-term outcome data are warranted to clarify the clinical and mechanistic impact of FMF-IBD overlap. Early recognition of FMF in this context may facilitate more personalized treatment strategies and ultimately improve disease management.
Ethical approval
The study was approved by Hacettepe University Ethics Committee (date: February 23, 2021, number: GO 21/75).
Source of funding
The authors declare the study received no funding.
Conflict of interest
The authors declare that there is no conflict of interest.
References
- Turner D, Ruemmele FM, Orlanski-Meyer E, et al. Management of paediatric ulcerative colitis, part 1: ambulatory care-an evidence-based guideline from European Crohn’s and Colitis Organization and European Society of Paediatric Gastroenterology, Hepatology and Nutrition. J Pediatr Gastroenterol Nutr 2018; 67: 257-291. https://doi.org/10.1097/MPG.0000000000002035
- Jose FA, Garnett EA, Vittinghoff E, et al. Development of extraintestinal manifestations in pediatric patients with inflammatory bowel disease. Inflamm Bowel Dis 2009; 15: 63-68. https://doi.org/10.1002/ibd.20604
- Özen S, Batu ED, Demir S. Familial mediterranean fever: recent developments in pathogenesis and new recommendations for management. Front Immunol 2017; 8: 253. https://doi.org/10.3389/fimmu.2017.00253
- Atas N, Eroglu GA, Sodan HN, et al. Long-term safety and efficacy of anakinra and canakinumab in patients with familial Mediterranean fever: a single-centre real-life study with 101 patients. Clin Exp Rheumatol 2021; 39(Suppl 132): 30-36. https://doi.org/10.55563/clinexprheumatol/815tdt
- Urgancı N, Ozgenc F, Kuloğlu Z, et al. Familial Mediterranean fever mutation analysis in pediatric patients with inflammatory bowel disease: a multicenter study. Turk J Gastroenterol 2021; 32: 248-250. https://doi.org/10.5152/tjg.2021.20057
- Uslu N, Yüce A, Demir H, et al. The association of inflammatory bowel disease and Mediterranean fever gene (MEFV) mutations in Turkish children. Dig Dis Sci 2010; 55: 3488-3494. https://doi.org/10.1007/s10620-010-1178-5
- Yücel BB, Aydog O, Nalcacioglu H, Yılmaz A. Effectiveness of canakinumab treatment in colchicine resistant Familial Mediterranean fever cases. Front Pediatr 2021; 9: 710501. https://doi.org/10.3389/fped.2021.710501
- Nakase H, Wagatsuma K, Kobayashi T, et al. Involvement of Mediterranean fever gene mutations in colchicine-responsive enterocolitis: a retrospective cohort study. EBioMedicine 2024; 110: 105454. https://doi.org/10.1016/j.ebiom.2024.105454
- Amaryan G, Sarkisian T, Tadevosyan A, Braegger C. Familial Mediterranean fever in Armenian children with inflammatory bowel disease. Front Pediatr 2024; 11: 1288523. https://doi.org/10.3389/fped.2023.1288523
- Levine A, Koletzko S, Turner D, et al. ESPGHAN revised porto criteria for the diagnosis of inflammatory bowel disease in children and adolescents. J Pediatr Gastroenterol Nutr 2014; 58: 795-806. https://doi.org/10.1097/MPG.0000000000000239
- Gattorno M, Hofer M, Federici S, et al. Classification criteria for autoinflammatory recurrent fevers. Ann Rheum Dis 2019; 78: 1025-1032. https://doi.org/10.1136/annrheumdis-2019-215048
- Petty RE, Southwood TR, Manners P, et al. International League of Associations for Rheumatology classification of juvenile idiopathic arthritis: second revision, Edmonton, 2001. J Rheumatol 2004; 31: 390-392.
- Jansson AF, Müller TH, Gliera L, et al. Clinical score for nonbacterial osteitis in children and adults. Arthritis Rheum 2009; 60: 1152-1159. https://doi.org/10.1002/art.24402
- Ozen S, Pistorio A, Iusan SM, et al. EULAR/PRINTO/PRES criteria for Henoch-Schönlein purpura, childhood polyarteritis nodosa, childhood Wegener granulomatosis and childhood Takayasu arteritis: Ankara 2008. Part II: Final classification criteria. Ann Rheum Dis 2010; 69: 798-806. https://doi.org/10.1136/ard.2009.116657
- Umehara H, Okazaki K, Kawa S, et al. The 2020 revised comprehensive diagnostic (RCD) criteria for IgG4-RD. Mod Rheumatol 2021; 31: 529-533. https://doi.org/10.1080/14397595.2020.1859710
- Saito D, Hibi N, Ozaki R, et al. MEFV gene-related enterocolitis account for some cases diagnosed as inflammatory bowel disease unclassified. Digestion 2020; 101: 785-793. https://doi.org/10.1159/000502640
- Demirtas Guner D, Bildik HN, Demir H, et al. Genetic variants in early-onset inflammatory bowel disease: monogenic causes and clinical implications. Children (Basel) 2025; 12: 536. https://doi.org/10.3390/children12050536
- Sari S, Egritas O, Dalgic B. The familial Mediterranean fever (MEFV) gene may be a modifier factor of inflammatory bowel disease in infancy. Eur J Pediatr 2008; 167: 391-393. https://doi.org/10.1007/s00431-007-0508-x
- Egritas O, Dalgic B. Infantile colitis as a novel presentation of familial Mediterranean fever responding to colchicine therapy. J Pediatr Gastroenterol Nutr 2011; 53: 102-105. https://doi.org/10.1097/MPG.0b013e31820cfab1
- Kuloğlu Z, Kansu A, Ustündağ G, Birsin Özçakar Z, Ensari A, Ekim M. An infant with severe refractory Crohn’s disease and homozygous MEFV mutation who dramatically responded to colchicine. Rheumatol Int 2012; 32: 783-785. https://doi.org/10.1007/s00296-009-1326-4
- Taş Ö, Aydın F, Kuloğlu Z, et al. Inflammatory bowel disease in paediatric rheumatological diseases. Clin Rheumatol 2025; 44: 2043-2052. https://doi.org/10.1007/s10067-025-07424-w
License
Copyright (c) 2026 The Author(s). This is an open access article distributed under the Creative Commons Attribution License (CC BY), which permits unrestricted use, distribution, and reproduction in any medium or format, provided the original work is properly cited.















