Graphical Abstract
Abstract
Background. Adenovirus infections are commonly associated with respiratory tract infections, conjunctivitis, gastrointestinal disturbances, and in some cases, more severe illnesses such as pneumonia. Guillain-Barré syndrome (GBS), a rare autoimmune disorder that causes progressive peripheral nerve inflammation and paralysis, has been linked to various infections, including viral ones.
Case Presentation. We describe two newly diagnosed pediatric cases of GBS following adenovirus infection. Both patients were previously healthy boys, aged 3 and 6 years. Adenovirus was detected by nasopharyngeal PCR shortly preceding the neurological manifestations. Both were treated with intravenous immunoglobulin (IVIG); one also required plasma exchange and respiratory support. Both recovered fully during follow-up. Additionally, a relevant case from the literature was reviewed for comparison. Treatment included respiratory support, IVIG and plasma exchange therapy. During the 12-month follow-up no severe complications were observed in our patients.
Conclusions. In conclusion, while GBS is a well-established complication of several viral infections, including adenovirus, the association between adenovirus and GBS remains relatively underexplored. Our case reports, along with a review of the literature, highlight the rare but plausible link between adenovirus infection and the development of GBS. Although adenovirus infections are common, the occurrence of GBS following these infections appears to be uncommon, suggesting that additional factors may be at play. However, given the potential severity of GBS and the growing body of evidence, it is important for clinicians to remain vigilant for this rare complication in patients with a history of adenovirus infection.
Keywords: Guillain-Barré syndrome, adenovirus, respiratory tract infection
Introduction
Adenovirus infections are commonly associated with respiratory tract infections, conjunctivitis, gastrointestinal disturbances, and in some cases, more severe illnesses such as pneumonia. Guillain-Barré syndrome (GBS), a rare autoimmune disorder that causes progressive peripheral nerve inflammation and paralysis, has been linked to various infections, including viral ones. Although the precise mechanisms are not entirely understood, there is evidence suggesting a possible association between adenovirus infection and the development of GBS. The relationship between adenovirus and GBS has been explored in a limited number of case reports and cohort studies. GBS is often triggered by viral infections, and adenovirus, as a known cause of respiratory and gastrointestinal illnesses, has been suggested as one of the possible viral triggers. The pathophysiology behind this connection may involve molecular mimicry, in which the immune system’s response to the adenovirus infection inadvertently targets peripheral nerves, leading to autoimmune damage.1,2 A few case studies have reported the development of GBS shortly after adenovirus infection, supporting the hypothesis that adenovirus can act as a potential trigger. For instance, a study by Loni et al.3 described a case where a patient presented with a bulbar predominant atypical GBS variant following an adenovirus infection, although such reports remain relatively rare. Additionally, a review by Jacobs et al.4 highlighted viral infections, including adenovirus, as potential antecedents for GBS, although they also emphasized that the exact mechanisms remain unclear.
In this study, we compiled data from our own patients diagnosed with GBS potentially associated with adenovirus infection, as well as cases reported in the literature. Our aim was to highlight the possible association between adenovirus and GBS emphasizing the need for further consideration of this relationship.
Case Presentation
Case 1
A 3-year-9-month-old male patient with no prior chronic conditions presented to our hospital on May 5, 2023, with increasing difficulty walking, weakness, and reluctance to stand. Symptoms began on April 20 with eye discharge, cough, and fever. By April 30, he exhibited back and leg pain, altered gait and difficulty walking independently. On admission, his vital signs were within normal limits. Neurological examination revealed no cranial nerve involvement. Mild weakness was present in the lower extremities with a Medical Research Council (MRC) scale of 4/5, and deep tendon reflexes were absent. No pathological reflexes were elicited. His gait was wide-based, and he exhibited difficulty maintaining balance. Cerebellar tests were normal. Superficial reflexes, including abdominal and cremasteric reflexes, were intact. No sensory level was detected. The spinal tap was traumatic, and cerebrospinal fluid (CSF) analysis revealed increased erythrocyte counts concurrent with the presence of blood and elevated levels of protein (354 mg/dL). Brain and spinal magnetic resonance imaging (MRI) were conducted, revealing no brain pathology. The spinal MRI was suboptimal due to wiggling and limited cooperation of the child. Electromyography (EMG) revealed demyelinating involvement in motor and sensory nerves, affecting both upper and lower extremities and was compatible with acute inflammatory demyelinating polyneuropathy (AIDP). GBS was considered based on the clinical and EMG findings. Adenovirus was detected in nasopharyngeal swab samples on admission. Respiratory support was not necessary during treatment. The patient received intravenous immunoglobulin (IVIG) therapy, and subsequent monitoring revealed improvement with ongoing rehabilitation.
Case 2
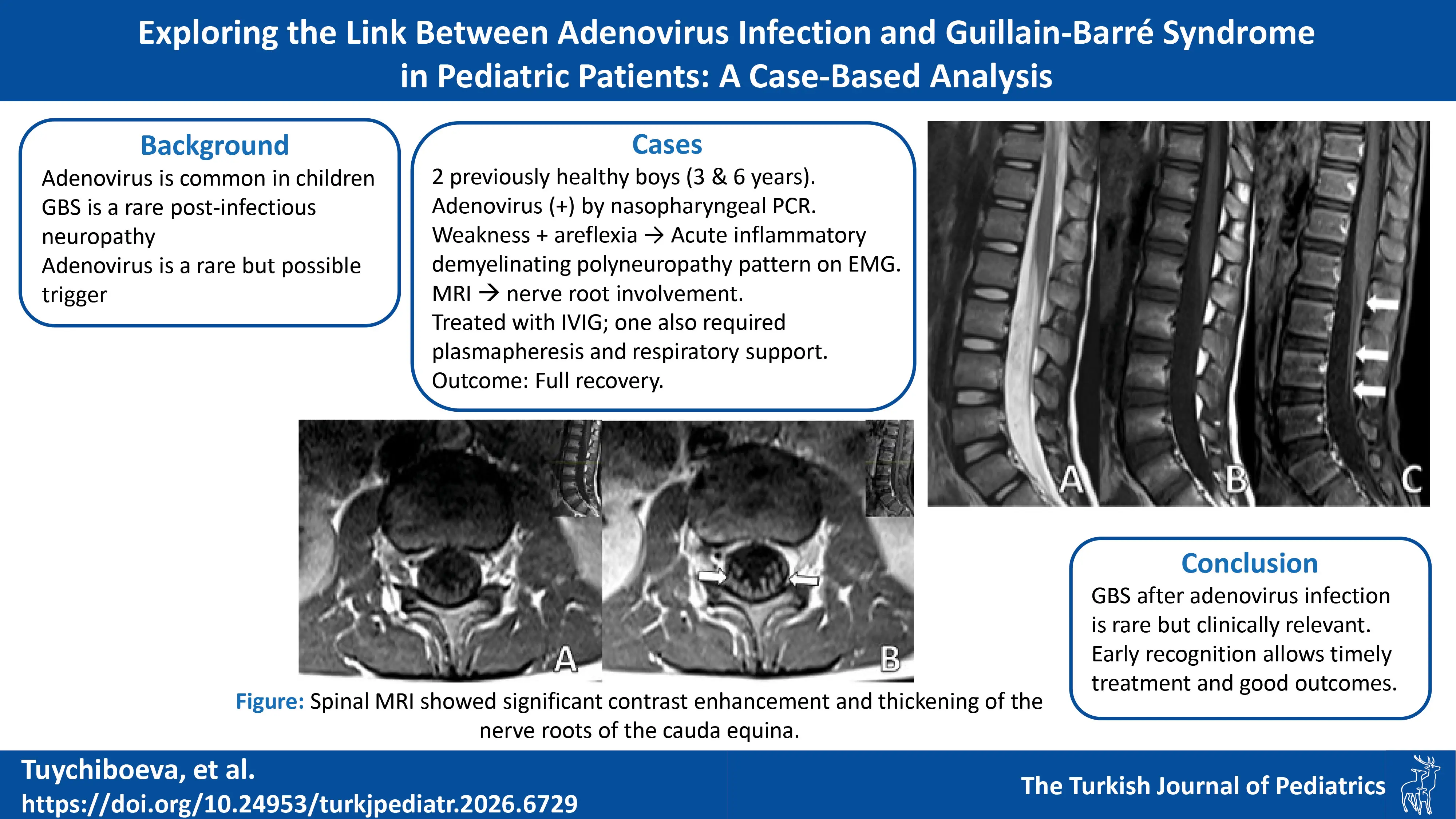
A 6-year-5-month-old male patient with no history of chronic illness presented with difficulty speaking, dyspnea, dysphagia, gait disturbance, and lower limb weakness in January 2022. He was referred to our center with those symptoms on the same day. Five days earlier, he had visited a local hospital for severe abdominal pain, nausea, and vomiting, diagnosed as acute gastroenteritis. On admission to our hospital, his vital signs were within normal limits. Neurological examination revealed no cranial nerve involvement, reduced muscle strength on both upper and lower extremities with a MRC scale of 3/5, and absent reflexes. No pathological reflexes were found. Abdominal and cremasteric superficial reflexes were intact and no sensory level was detected. Cerebellar function was assessed to be normal within the limits of the examination. CSF analysis showed an elevated level of protein (117 mg/dL). Spinal MRI showed significant contrast enhancement and thickening of the nerve roots of the cauda equina (Fig. 1 and Fig. 2).
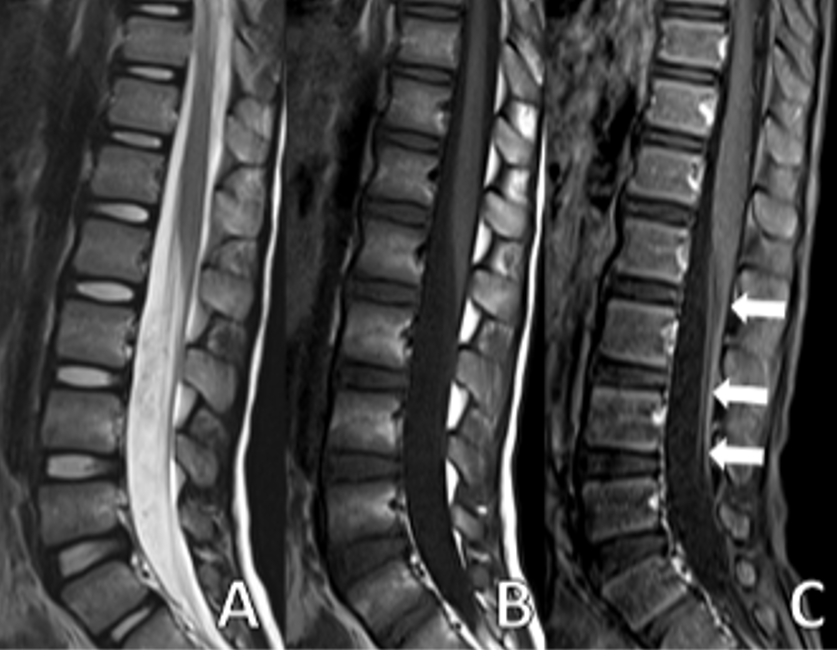
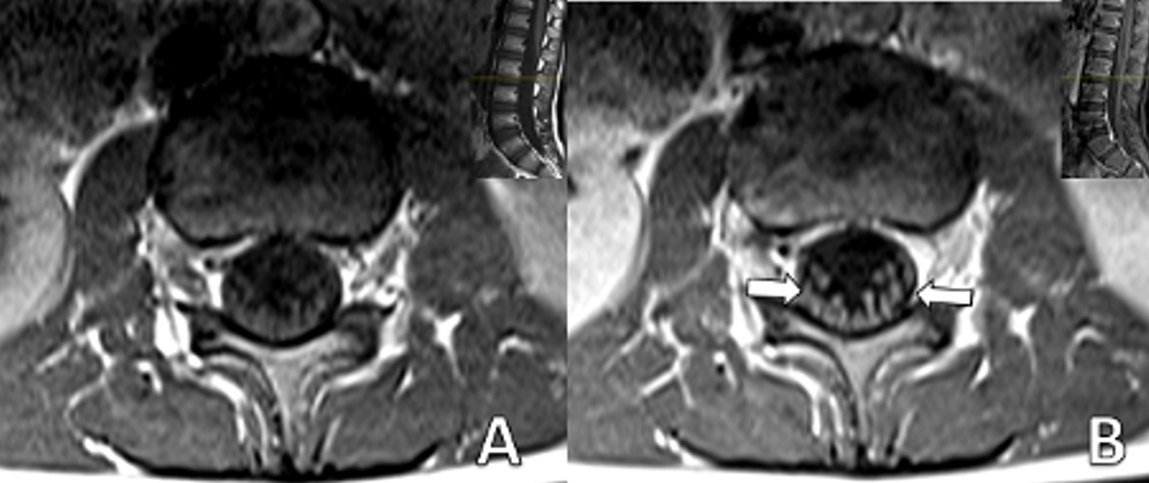
EMG revealed a severe demyelinating involvement in motor and sensory nerves. The clinical and laboratory findings were consistent with a diagnosis of GBS. The patient received IVIG and plasma exchange therapy, further improvement noted in his neurological status. Adenovirus was detected in nasopharyngeal swab samples on admission. The patient received respiratory support for 10 days during hospitalization. After intensive treatment, including 7 days of IVIG and plasma exchange, the patient’s condition improved sufficiently for discharge.
We have obtained informed consent from the patients of these patients for the publication of this case report. The summary of these two cases, along with the case of atypical GBS reported by Loni et al.3 is presented in Table I.
| None of the three patients had an underlying disease; *Spinal MRI evaluation was limited, as post-contrast sequences covered only the upper spinal levels (C1–T9); AIDP: Acute inflammatory demyelinating polyneuropathy, CSF: Cerebrospinal fluid, DTR: deep tendon reflexes, EMG: Electromyography, IVIG: intravenous immunoglobulin, MP: methylprednisolone, MRI: Magnetic Resonance Imaging, RBC: red blood cells, WBC: white blood cell. | ||||||
| Table I. The demographic and clinical data of patients with Guillain-Barré syndrome and adenovirus infection | ||||||
| Patient, year, age, sex | History and examination | CSF | MRI | EMG | Treatment | Outcome |
| Case 1 in present report, 2023, 3 yr, M | Lower limb weakness, walking difficulty, absent DTR | Glucose: 68 mg/dL, Protein: 354.5 mg/dL, Albumin: 87.6 mg/dL, Many RBC (traumatic puncture), No WBC |
Brain MRI normal. Spinal MRI: Normal* |
AIDP in the lower and upper extremities | IVIG at a dose of 0.4 gr/kg/d for 5 days | Recovery |
| Case 2 in present report, 2022, 6 yr, M | Speaking, swallowing, walking difficulties, urinary incontinence weakness, absent DTR | Glucose: 72 mg/dL, Protein: 117 mg/dL, Albumin: 60.9 mg/dL, No cells |
Contrast enhancement and thickening of the nerve roots of the cauda equinaf | Severe AIDP of sensory and motor fibers | 2 doses of IVIG 1 g/kg/dose, and 3 doses at of 0.4 g/kg/dose. Plasmapheresis x5. | Recovery |
| Report by Loni et al, 2024.3, 6 yr, M | Swallowing and speaking difficulties, drooling, weak gag and cough reflexes, hyperreflexia without clonus, upgoing Babinski reflexes, and axial hypotonia with preserved limb tone. | Glucose: 77 mg/dL, Protein total: 14.54 mg/dL, Albumin: 95.6 mg/L, No cells |
Brain MRI: Millimeter-sized nonspecific white matter lesions. Spinal MRI: Axial T1 weighted fat-saturated image with contrast shows thickened enhancing roots of cauda equina |
Not performed. | MP for 3 days, followed by IVIG 2 g/kg over 48 hr in 2 divided doses. | Residual sequelae |
Discussion
The exact mechanism by which adenovirus could trigger GBS is not well understood, but it is thought to involve a dysregulated immune response. During viral infections, the body’s immune system may produce antibodies that cross-react with neuronal gangliosides, components of nerve cell membranes, resulting in demyelination and nerve damage.2 This phenomenon, known as molecular mimicry, is a proposed mechanism for the development of GBS following infections.
Molecular mimicry refers to a phenomenon in which prior infection with microorganisms leads to the incidental sharing of identical epitopes between microbial and neural structures, thereby triggering a cellular immune response. Several microorganisms have been notably linked to GBS, including Campylobacter jejuni, Mycoplasma pneumoniae, Haemophilus influenzae, Epstein-Barr virus, cytomegalovirus (CMV), varicella-zoster virus, and influenza virus. Among these, C. jejuni is the most frequently associated pathogen.4 Other infections that have been associated with GBS include the Zika virus, influenza A virus, hepatitis E, hepatitis B, and human immunodeficiency virus (HIV). Despite the absence of a clear consensus, GBS has been strongly linked to severe acute respiratory distress syndrome coronavirus 2 (SARS-CoV-2) infection (coronavirus disease 2019, COVID-19). The precise pathophysiological mechanisms underlying COVID-19 as a potential cause of GBS remain unclear. Furthermore, adenovirus-based vaccines have been implicated in an increased risk of developing GBS.5
A recent systematic review reported that the incidence of GBS following COVID-19 vaccination was 8.1 cases per 1,000,000 vaccinations. The review further revealed that this increased risk was more strongly associated with adenovirus vector vaccines than with mRNA vaccines.6 It is possible that the higher incidence of GBS in adenovirus vector-based vaccines may be related to the direct GBS-triggering properties of the adenovirus itself. However, with the current available data, this remains speculative. Nevertheless, in the current study, the possibility of an association between adenovirus and GBS in patients who had no underlying conditions and for whom no other causal factors could be identified during their illness cannot be disregarded. Adenovirus detection by nasopharyngeal PCR should be interpreted with caution, as positivity may occasionally reflect asymptomatic carriage or prolonged shedding.7 However, in our patient, alternative causes were excluded through negative blood and CSF cultures and a comprehensive respiratory viral panel. In this context, adenovirus remains a clinically relevant finding and a plausible trigger for GBS, although causality cannot be proven solely by PCR positivity. While adenovirus infections are common, the occurrence of GBS following adenoviral infection is rare. However, the role of adenovirus as an occasional but plausible trigger for GBS warrants further investigation, particularly in specific patient populations or during outbreaks.
All patients in this case series underwent brain and spinal MRIs. Radiological studies are essential to rule out other potential causes when nerve conduction studies and CSF examinations yield inconclusive results. An MRI of the spine is particularly helpful, as it can help exclude other conditions, such as transverse myelitis and compressive causes of polyradiculopathy. If a non-contrast MRI is performed, it is important to administer contrast media if a specific diagnosis is suspected, as the non-contrast sequences are usually normal.8 The most common site of enhancement in GBS is the anterior nerve roots, though enhancement of the posterior nerve roots can also occur.
In conclusion, while GBS is a well-established complication of several viral infections, including adenovirus, the association between adenovirus and GBS remains relatively underexplored. Our case reports, along with a review of the literature, highlight the rare but plausible link between adenovirus infection and the development of GBS. Although adenovirus infections are common, the occurrence of GBS following these infections appears to be uncommon, suggesting that additional factors may be at play. However, given the potential severity of GBS and the growing body of evidence, it is important for clinicians to remain vigilant for this rare complication in patients with a history of adenovirus infection.
The main limitation of this report is the small number of cases, which limits the generalizability of our observations. Larger case series are needed to better clarify the relationship between adenovirus infection and GBS.
Ethical approval
We have obtained informed consent from the patients for publication of the data.
Source of funding
The authors declare the study received no funding.
Conflict of interest
The authors declare that there is no conflict of interest.
References
- Willison HJ, Jacobs BC, van Doorn PA. Guillain-Barré syndrome. Lancet 2016; 388: 717-727. https://doi.org/10.1016/S0140-6736(16)00339-1
- Shastri A, Al Aiyan A, Kishore U, Farrugia ME. Immune-mediated neuropathies: pathophysiology and management. Int J Mol Sci 2023; 24: 7288. https://doi.org/10.3390/ijms24087288
- Loni R, Alsindi AM, Alfaw AA, et al. Acute encephalitis with flaccid paralysis following an adenoviral infection presenting as a rare variant of Guillain-Barre syndrome in a school-age child. Int J Contemp Pediatr 2024; 11: 1123-8. https://doi.org/10.18203/2349-3291.ijcp20241944
- Jacobs BC, Rothbarth PH, van der Meché FG, et al. The spectrum of antecedent infections in Guillain-Barré syndrome: a case-control study. Neurology 1998; 51: 1110-1115. https://doi.org/10.1212/wnl.51.4.1110
- Valaparla VL, Rane SP, Patel C, Li X. Guillain-Barre syndrome and link with COVID-19 infection and vaccination: a review of literature. Front Neurol 2024; 15: 1396642. https://doi.org/10.3389/fneur.2024.1396642
- Ogunjimi OB, Tsalamandris G, Paladini A, Varrassi G, Zis P. Guillain-Barré syndrome induced by vaccination against COVID-19: a systematic review and meta-analysis. Cureus 2023; 15: e37578. https://doi.org/10.7759/cureus.37578
- Kalu SU, Loeffelholz M, Beck E, et al. Persistence of adenovirus nucleic acids in nasopharyngeal secretions: a diagnostic conundrum. Pediatr Infect Dis J 2010; 29: 746-750. https://doi.org/10.1097/INF.0b013e3181d743c8
- Alkan O, Yildirim T, Tokmak N, Tan M. Spinal MRI findings of guillain-barré syndrome. J Radiol Case Rep 2009; 3: 25-28. https://doi.org/10.3941/jrcr.v3i3.153
License
Copyright (c) 2026 The Author(s). This is an open access article distributed under the Creative Commons Attribution License (CC BY), which permits unrestricted use, distribution, and reproduction in any medium or format, provided the original work is properly cited.















