Graphical Abstract
Abstract
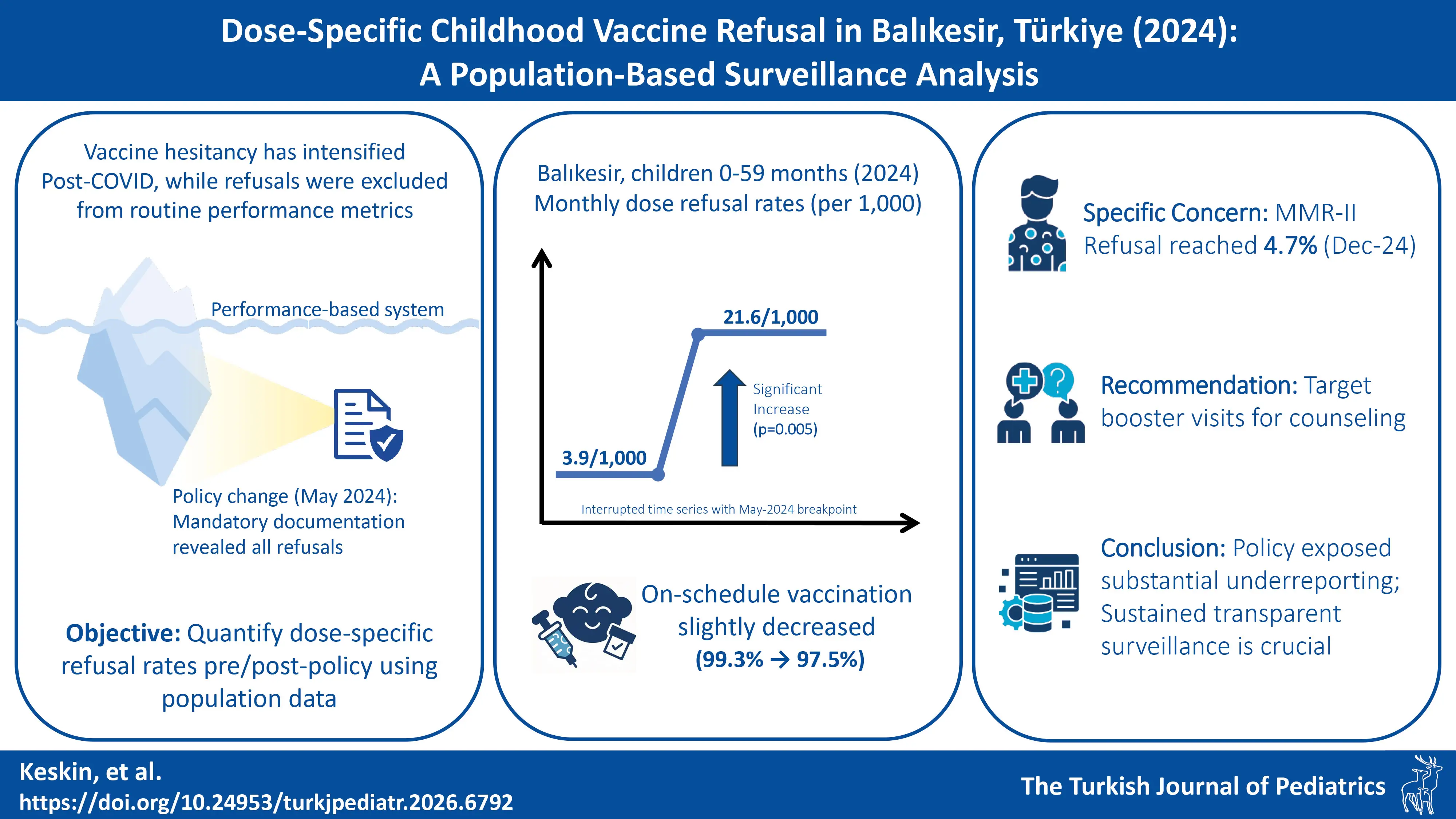
Background. Vaccine hesitancy has intensified since COVID-19. In Türkiye, a performance-based family medicine system provides free vaccines. Before May 2024, physicians could remove refusals from metrics tied to pay deductions, obscuring true rates. A new policy mandating documentation created a natural experiment. We used population-based data to quantify these previously hidden dose-specific refusal rates across the childhood schedule.
Methods. We conducted a population-based ecological analysis of monthly dose-specific refusals among children aged 0–59 months in Balıkesir Province, in 2024. Refusals were extracted from Performance Exception Forms. Rates were calculated per 1,000 target children (Wilson 95% confidence intervals [CI]s), using populations derived from Turkish Statistical Institute counts. To quantify the policy’s impact, we fitted an interrupted time-series regression with a fixed breakpoint at May 2024. This model estimated the baseline level and slope (January-April) and compared them to the immediate level shift and new monthly slope post-reform (May–December).
Results. Of 3,584 missed-dose exceptions, 3,002 (83.8%) were refusals. The aggregate refusal rate was 3.9 per 1,000 (January-April) versus 21.6 per 1,000 (May–December), while the rate of on-schedule vaccinations decreased from 99.3% to 97.5%. Segmented regression indicated a baseline of 2.6 per 1,000 and a post-policy level increase of 13.7 per 1,000 (p=0.005); pre- and post-policy monthly slopes were not significant. Refusals concentrated in booster and later-scheduled doses (18–48 months), whereas primary-series doses showed lower, comparatively stable rates. Notably, refusal for the second measles-mumps-rubella dose (MMR-II, 48 months) rose toward year-end, reaching 47.4 per 1,000 in December (95% CI, 35.1–63.6).
Conclusions. Mandatory documentation exposed substantial under-ascertainment of parental refusal in performance-based records. Refusal is disproportionately clustered at booster and later-childhood visits, with MMR-II approaching herd-immunity margins. Programmatically, pediatric booster encounters are key leverage points for targeted counseling, and sustained, transparent surveillance as enabled by the new policy, is essential for early signal detection and response.
Keywords: booster doses, childhood immunization, health policy, interrupted time-series, surveillance, vaccine hesitancy
Introduction
Childhood immunization is the cornerstone of public health, known for preventing millions of deaths globally each year.1 However, a new trend among parents has emerged. Vaccine hesitancy, defined as the delay in acceptance, or refusal of vaccination despite service availability, has been declared a top-ten global health threat by the World Health Organization (WHO), contributing to resurgences of diseases like measles.2 In the post-COVID-19 era, vaccine hesitancy has intensified globally. European research, for instance, highlights a decline in vaccination coverage that has occurred alongside growing parental concerns about routine childhood immunizations.3 On a wider scale, global meta-analyses estimate that over 21% of parents exhibit hesitancy regarding routine childhood vaccines, though the prevalence varies significantly across regions.4
Türkiye’s vaccination landscape has undergone a significant transformation over the past decade, shaped by both policy reforms and evolving public attitudes. While the country has made well-documented progress—reducing zero-dose prevalence from 3.2% in 1993 to 0.9% in 2018, despite persistent regional inequities5 —a paradoxical trend has emerged. During this same period, vaccine hesitancy has surged, escalating from rare events to over 23,000 families by 2018, a trend potentially linked to intensified public debate and a 2015 legal decision on parental consent.6,7
This trend unfolded within Türkiye’s national immunization programme which provides vaccines free of charge through an easy-to-access nationwide, performance-based family medicine system. All health facilities are connected through an integrated information technology (IT) infrastructure, enabling the vaccine tracing system to track immunizations administered in any setting—including hospitals and private clinics—beyond the family physician (FP) network.8 However, if system records indicate that a child is unvaccinated, the FP is required to document an explanation to avoid pay deductions. Thus, the performance-based system mandates FPs to certify their non-accountability for every unmet preventive service -including missed vaccinations- in a monthly performance exception form.9 Despite this accountability mechanism, the system’s financial incentives created a surveillance blind spot. Until May 2024, FPs could record a missed dose as “parental refusal” to remove it from the performance metrics used to calculate pay deductions, meaning that many refusals never reached the national surveillance database. The new regulation implemented in May 2024 reversed this, closing the systemic gap by making all such events visible to the national system and mandating that physicians obtain a wet-signed parental document for each refusal as the condition for avoiding a performance deduction.
This policy change created a natural experiment to address the gap in systematic documentation. While national surveys like the Turkish Demographic Health Survey10 provide some population estimates (e.g., 3.4% unvaccinated among children aged 24-35 months in 2018), these figures represent multiple reasons for non-vaccination and not solely explicit refusal. Conversely, recent community-based studies in Türkiye have suggested substantially higher rates of vaccine hesitancy, with some regions reporting hesitancy rates exceeding 15%, highlighting the discrepancy between survey-based estimates and administratively flawed surveillance data.11,12 Earlier attempts to quantify refusal through FP reporting documented comparatively low rates13, indicating a notable gap in the systematic documentation of refusal rates.
Therefore, this study aims to quantify dose-specific vaccine refusal rates among children aged 0–59 months in Balıkesir (2024) and to test the hypothesis that the May 2024 mandatory documentation policy created an immediate, significant increase in recorded refusals, thereby exposing the true, previously under-ascertained burden.
Materials and Methods
Study design
Adopting an ecological cross-sectional approach, we examined monthly dose-specific refusals of vaccines included in Türkiye’s national immunization programme among children aged 0–59 months in 2024.
Study population, data collection, and variables
The study population comprised children aged 0–59 months residing in Balıkesir, a province in the Marmara Region of Türkiye. By December 2024, the province had approximately 1.2 million residents, with an estimated 55,000 falling within the target age band.14 Missed vaccine data were collected from the publicly available anonymized ‘family medicine practice performance exception forms’ published monthly by the Balıkesir Provincial Health Directorate from January 2024 to December 2024.15 Free-text reasons for missed vaccination were coded and cross-validated by the authors, then grouped into four standard categories (vaccine refusal, migration, logistics, and other), enabling the identification of explicit refusals linked to specific doses. The 12 monthly files were then merged into a single dataset.
Because the study focused on doses routinely delivered in the family medicine system, hepatitis B dose I (administered at birth in hospitals) and the tetanus-diphtheria (Td) booster (Td-R) given at age 13 through school services were excluded. The national immunization schedule is outlined in Table I.
|
*Balıkesir does not need an extra dose of MMR at 9 months. Hepatitis B I and Td are excluded from the study. BCG: Bacille Calmette-Guérin vaccine, DaPT-IPV: Quadrivalent diphtheria and tetanus toxoid with acellular pertussis + inactivated polio vaccine, DaPT-IPV-HIB: Pentavalent Diphtheria and tetanus toxoid with acellular pertussis + inactivated polio vaccine + Haemophilus influenzae type b pediatric dose vaccine, Hepatitis A: Hepatitis A pediatric dose vaccine, Hepatitis B: Hepatitis B pediatric dose vaccine, MMR: Measles mumps rubella vaccine, Mo: At the end of months, OPV: Oral polio vaccine, PCV: Pneumococcal conjugate vaccine, R: Rappel (Booster), Td: Tetanus-diphtheria vaccine, Varicella: Varicella vaccine, Yr: At the end of the year. |
||||||||||
| Table I. National immunization schedule for vaccines by the Ministry of Health of Türkiye (2024) | ||||||||||
| Vaccine |
|
|
|
|
|
|
|
|
|
|
| Hepatitis B |
|
|
|
|||||||
| BCG |
|
|||||||||
| DTaP-IPV-Hib |
|
|
|
|
||||||
| PCV |
|
|
|
|||||||
| MMR* |
|
|
||||||||
| DTaP-IPV |
|
|||||||||
| OPV |
|
|
||||||||
| Td |
|
|||||||||
| Hepatitis A |
|
|
||||||||
| Varicella |
|
|||||||||
Estimating monthly target populations
Monthly target population numbers were derived by disaggregating annual single-age population counts according to observed birth distributions, thereby addressing seasonal variation (e.g., peaks in January and summer). Monthly live-birth counts for each province (2019-2023) were obtained from Turkish Statistical Institute (TURKSTAT) birth statistics (Supplementary Table S1).16 For each calendar month, we divided the corresponding birth count by that year’s total births to derive within-year monthly proportions (e.g., January 2020: 932/11,444 = 0.081), presented in Supplementary Table S2. Single-year age populations at the provincial level were retrieved from the 2024 TURKSTAT address-based population registration system dated 31 December 2024 (Supplementary Table S3).14 Because monthly birth statistics for 2024 were not yet available, we projected the 2024 monthly birth distribution by applying the 2023 proportions to the 2024 annual birth estimate. The monthly target population calculation method is further exemplified in the Supplementary Tables S3 and S4.
Measurements and analysis
Dose-specific vaccine refusal rates were computed by dividing the monthly vaccine refusal counts by the corresponding projected monthly target population numbers, expressed per thousand individuals; illustrative example of this calculation is provided in Supplementary Tables S5-S7. Robust 95% confidence intervals (CIs) were generated with the Wilson method, which is widely regarded as more reliable for low proportions. Temporal trends were visualized using raw monthly rates to explicitly display month-to-month variability. To quantify the policy-related change, we fitted an interrupted time-series regression with a fixed break at May 2024. Model 1 assessed the aggregate trend, while Model 2 was fitted to the full disaggregated panel dataset (18 vaccine types X 12 months; N=216) to estimate vaccine-specific effects with sufficient degrees of freedom. Given the short pre-intervention window, these models were specified primarily to estimate the baseline level (January-April) and the immediate level shift post-reform (May-December), while treating slope estimates descriptively. Statistical analyses were conducted using R software version 4.2.1.
Ethics approval
This study constitutes a secondary analysis of publicly available, anonymized, and aggregated data. The datasets are made accessible through the official website of the Ministry of Health in accordance with applicable regulations and legislation17; specific data links are available in the Supplementary Table S8. Given the use of non-identifiable administrative surveillance data that are publicly disclosed, this research is exempt from formal ethics committee approval.
Results
In 2024, 3,584 unmet vaccine-dose exceptions were reported in Balıkesir. Before the May policy change (January–April), 423 exceptions occurred alongside a 99.3% on-schedule vaccination rate; refusals accounted for 60.0% (n=254) of these exceptions, followed by ‘other’ reasons (21.5%, n=91) and migration (18.4%, n=78). Post-policy (May–December), exceptions increased to 3,161 while the on-schedule rate slightly decreased to 97.5%. Recorded refusals increased to 2,748, comprising 86.9% of post-reform exceptions. Other reasons included ‘other’ (7.4%, n=234), migration (5.3%, n=169), and logistical issues (0.3%, n=10). Overall, parental refusal (n=3,002) constituted 83.8% of all yearly exceptions, establishing it as the primary driver of missed doses, far exceeding other factors. Dose-specific refusal details are presented in Table II. Vaccine-group-specific monthly refusal counts and refusal rates are summarized in Supplementary Tables S9 and S10. Correspondingly, the aggregate refusal rate rose from 3.9 per 1,000 (95% CI: 3.4–4.4) in the first four months to 21.6 per 1,000 (95% CI: 20.8–22.4) in the subsequent eight-month period. This increase was consistent across all age-specific vaccination schedules.
|
Mo: At the end of months. Projected: Projected target population count for the specific age milestone during the specified period. Rate (‰): Refusal rate calculated per 1,000 projected target children (Refusal count / Projected × 1,000). |
||||||
| Table II. Childhood vaccine refusal counts and rates (per thousand target children) by age milestone and period | ||||||
| Month |
|
|
||||
|
|
|
|
|
|
|
|
| 1 Mo |
|
|
|
|
|
|
| 2 Mo |
|
|
|
|
|
|
| 4 Mo |
|
|
|
|
|
|
| 6 Mo |
|
|
|
|
|
|
| 12 Mo |
|
|
|
|
|
|
| 18 Mo |
|
|
|
|
|
|
| 24 Mo |
|
|
|
|
|
|
| 48 Mo |
|
|
|
|
|
|
| Total |
|
|
|
|
|
|
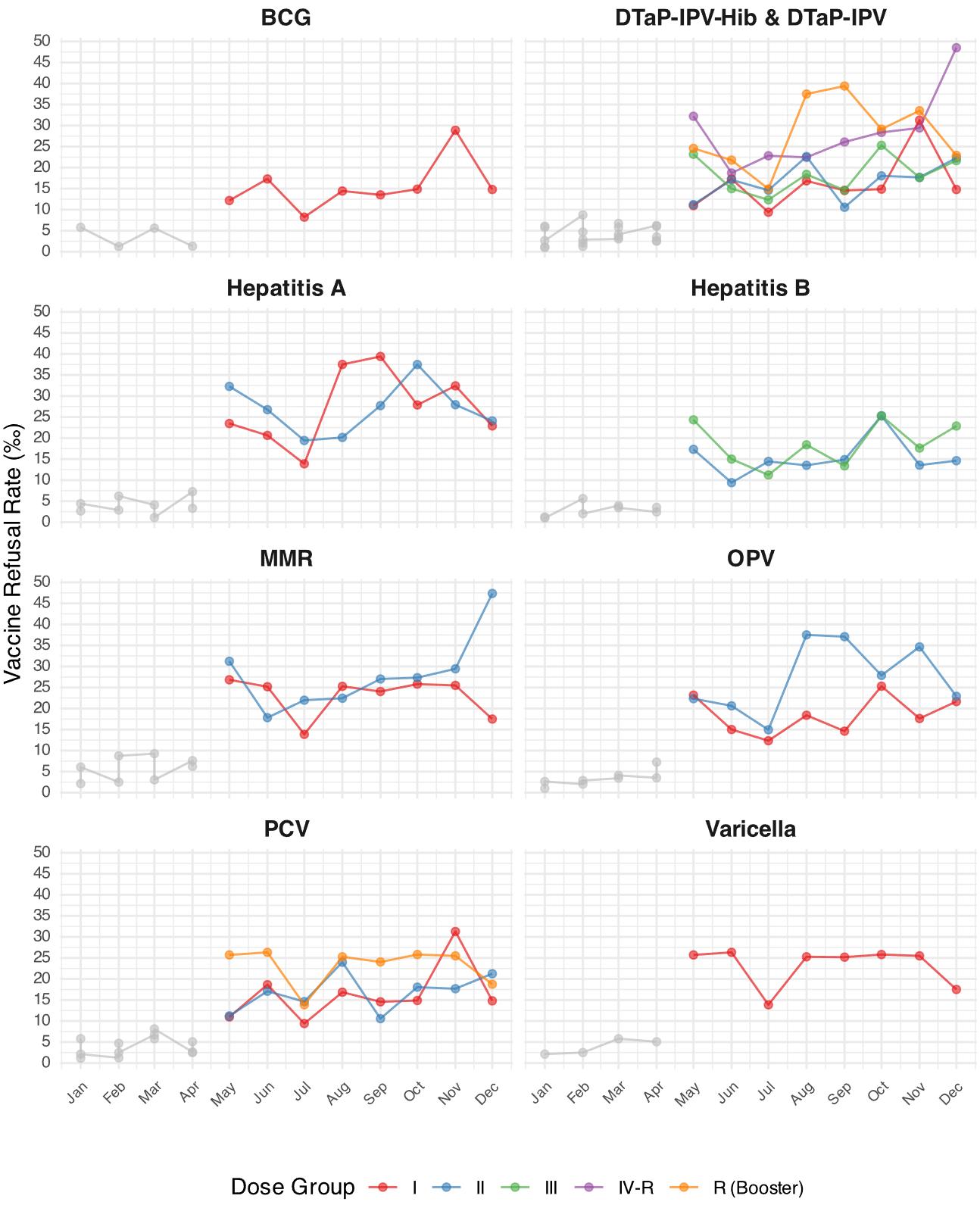
Monthly refusal rates during the May-December 2024 period revealed dose-specific variations (Table III). Primary-series vaccines such as Bacille Calmette-Guérin (BCG) I, pneumococcal conjugate vaccine (PCV) I, and diphtheria and tetanus toxoid with acellular pertussis, inactivated polio vaccine, and Haemophilus influenzae type b vaccine (DTaP-IPV-Hib) I generally exhibited lower refusal rates than their corresponding boosters. Refusal rates peaked for DTaP-IPV-Hib rappel (booster) dose (R) (39.4‰ in September) and particularly for diphtheria and tetanus toxoid with acellular pertussis and inactivated polio vaccine (DTaP-IPV) R (48.5‰ in December). Temporal trends had variations across vaccines; refusal rates for measles, mumps, and rubella vaccine (MMR) II and the DTaP-IPV R booster showed increases towards the end of the year, peaking in December at 47.4‰ (95% CI: 35.1–63.6) and 48.5‰ (95% CI: 36.1–64.9), respectively. Conversely, rates for some early doses, such as Hepatitis B II and BCG I, remained comparatively stable, varying between 8.2‰ and 28.9‰.
| BCG: Bacille Calmette-Guérin vaccine, DaPT-IPV: Quadrivalent diphtheria and tetanus toxoid with acellular pertussis + inactivated polio vaccine, DaPT-IPV-HIB: Pentavalent diphtheria and tetanus toxoid with acellular pertussis + inactivated polio vaccine + Haemophilus influenzae type b pediatric dose vaccine, Hepatitis A: Hepatitis A pediatric dose vaccine, Hepatitis B: Hepatitis B pediatric dose vaccine, MMR: Measles mumps rubella vaccine, Mo: At the end of months, OPV: Oral polio vaccine, PCV: Pneumococcal conjugate vaccine, R: Rappel (Booster), Varicella: Varicella vaccine. | |||||||||
| Table III. Dose-specific monthly vaccine refusal rates per thousand, May-December 2024 | |||||||||
| Schedule | Vaccine | May 24 | Jun 24 | Jul 24 | Aug 24 | Sep 24 | Oct 24 | Nov 24 | Dec 24 |
| 1 Mo | Hepatitis B II | 17.3 (10.1-29.4) | 9.4 (4.8-18.4) | 14.4 (8.3-25.1) | 13.5 (7.9-23.0) | 14.9 (8.9-24.8) | 25.3 (16.6-38.3) | 13.6 (7.6-24.1) | 14.6 (8.4-25.4) |
| 6 Mo | Hepatitis B III | 24.3 (16.0-36.9) | 15.0 (8.6-26.0) | 11.2 (6.1-20.5) | 18.4 (11.0-30.6) | 13.4 (7.5-23.8) | 25.3 (16.3-39.2) | 17.6 (10.7-28.9) | 22.9 (14.7-35.4) |
| 2 Mo | BCG I | 12.2 (6.6-22.3) | 17.3 (10.1-29.4) | 8.2 (4.0-16.9) | 14.4 (8.3-25.1) | 13.5 (7.9-23.0) | 14.9 (8.9-24.8) | 28.9 (19.5-42.6) | 14.8 (8.5-25.7) |
| 2 Mo | DTaP-IPV-Hib I | 11.0 (5.8-20.7) | 17.3 (10.1-29.4) | 9.4 (4.8-18.4) | 16.8 (10.1-28.1) | 14.6 (8.7-24.3) | 14.9 (8.9-24.8) | 31.3 (21.4-45.4) | 14.8 (8.5-25.7) |
| 4 Mo | DTaP-IPV-Hib II | 11.2 (6.1-20.5) | 17.1 (10.0-29.0) | 14.6 (8.4-25.4) | 22.6 (14.2-36.0) | 10.6 (5.6-20.0) | 18.1 (11.0-29.6) | 17.7 (11.1-28.1) | 22.3 (14.6-33.9) |
| 6 Mo | DTaP-IPV-Hib III | 23.2 (15.1-35.5) | 15.0 (8.6-26.0) | 12.3 (6.9-22.0) | 18.4 (11.0-30.6) | 14.6 (8.4-25.4) | 25.3 (16.3-39.2) | 17.6 (10.7-28.9) | 21.7 (13.7-34.0) |
| 18 Mo | DTaP-IPV-Hib R | 24.6 (16.3-36.9) | 21.8 (14.0-33.8) | 14.9 (8.9-24.9) | 37.5 (26.4-53.0) | 39.4 (28.3-54.6) | 29.1 (19.5-43.3) | 33.5 (23.6-47.5) | 22.9 (14.9-35.1) |
| 48 Mo | DTaP-IPV R | 32.2 (23.0-44.9) | 18.7 (12.3-28.4) | 22.8 (15.7-33.0) | 22.4 (15.4-32.7) | 26.1 (18.1-37.5) | 28.4 (19.6-41.0) | 29.4 (20.4-42.2) | 48.5 (36.1-64.9) |
| 2 Mo | PCV I | 11.0 (5.8-20.7) | 18.6 (11.1-31.1) | 9.4 (4.8-18.4) | 16.8 (10.1-28.1) | 14.6 (8.7-24.3) | 14.9 (8.9-24.8) | 31.3 (21.4-45.4) | 14.8 (8.5-25.7) |
| 4 Mo | PCV II | 11.2 (6.1-20.5) | 17.1 (10.0-29.0) | 14.6 (8.4-25.4) | 24.0 (15.2-37.6) | 10.6 (5.6-20.0) | 18.1 (11.0-29.6) | 17.7 (11.1-28.1) | 21.3 (13.8-32.6) |
| 12 Mo | PCV R | 25.7 (17.2-38.3) | 26.3 (17.6-39.2) | 13.9 (8.3-23.1) | 25.3 (17.2-37.0) | 24.0 (15.8-36.5) | 25.8 (17.1-38.8) | 25.5 (16.9-38.3) | 18.8 (11.4-30.7) |
| 6 Mo | OPV I | 23.2 (15.1-35.5) | 15.0 (8.6-26.0) | 12.3 (6.9-22.0) | 18.4 (11.0-30.6) | 14.6 (8.4-25.4) | 25.3 (16.3-39.2) | 17.6 (10.7-28.9) | 21.7 (13.7-34.0) |
| 18 Mo | OPV II | 22.3 (14.5-34.3) | 20.6 (13.1-32.4) | 14.9 (8.9-24.9) | 37.5 (26.4-53.0) | 37.1 (26.4-51.9) | 27.9 (18.5-41.8) | 34.7 (24.5-48.8) | 22.9 (14.9-35.1) |
| 12 Mo | MMR I | 26.8 (18.1-39.6) | 25.2 (16.7-37.8) | 13.9 (8.3-23.1) | 25.3 (17.2-37.0) | 24.0 (15.8-36.5) | 25.8 (17.1-38.8) | 25.5 (16.9-38.3) | 17.5 (10.5-29.2) |
| 48 Mo | MMR II | 31.2 (22.2-43.8) | 17.8 (11.6-27.4) | 22.0 (15.0-32.0) | 22.4 (15.4-32.7) | 27.0 (18.9-38.5) | 27.3 (18.7-39.7) | 29.4 (20.4-42.2) | 47.4 (35.1-63.6) |
| 12 Mo | Varicella I | 25.7 (17.2-38.3) | 26.3 (17.6-39.2) | 13.9 (8.3-23.1) | 25.3 (17.2-37.0) | 25.2 (16.7-37.8) | 25.8 (17.1-38.8) | 25.5 (16.9-38.3) | 17.5 (10.5-29.2) |
| 18 Mo | Hepatitis A I | 23.5 (15.4-35.6) | 20.6 (13.1-32.4) | 13.9 (8.1-23.6) | 37.5 (26.4-53.0) | 39.4 (28.3-54.6) | 27.9 (18.5-41.8) | 32.4 (22.7-46.2) | 22.9 (14.9-35.1) |
| 24 Mo | Hepatitis A II | 32.3 (22.7-45.7) | 26.7 (18.8-37.9) | 19.4 (12.9-29.2) | 20.1 (13.2-30.6) | 27.7 (19.1-40.0) | 37.5 (26.5-52.7) | 27.9 (19.0-40.9) | 24.1 (15.8-36.5) |
Fig. 1 illustrates the monthly temporal trends in dose-specific refusal rates throughout 2024. A visual shift occurs across all vaccine groups starting in May 2024, coinciding with the implementation of the mandatory refusal reporting system and revealing higher rates thereafter. Post-May, later doses within a series and boosters regularly attracted more refusals than initial doses, whereas month-to-month volatility persisted. End-of-the-year surge for the 48-month boosters (DTaP-IPV R and MMR II) is particularly pronounced.
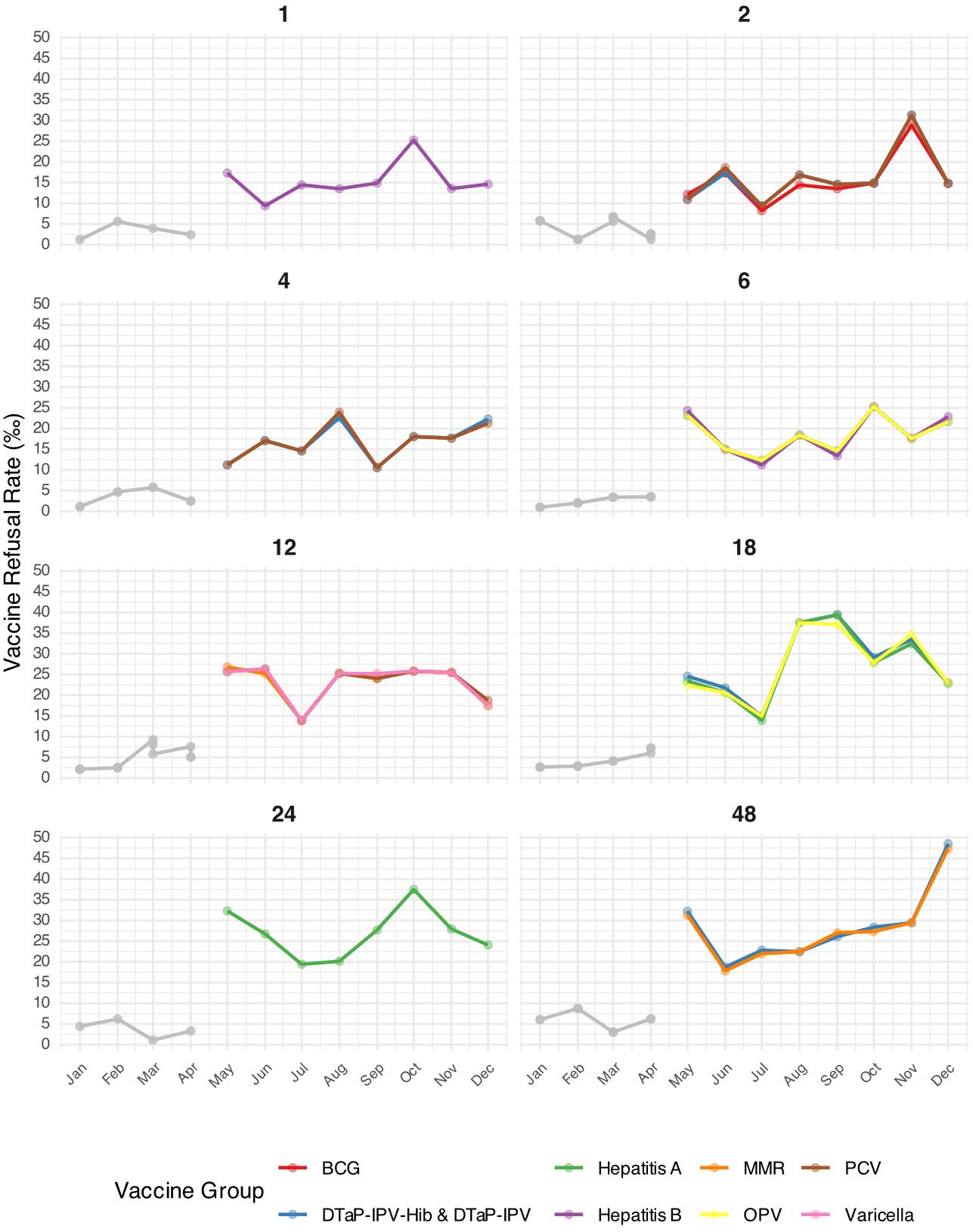
Grouping refusals by scheduled age (Fig. 2) yields a similar hierarchy: the 18-, 24-, and 48-month milestones generally attracted the highest rates once routine mandatory reporting began, whereas vaccines scheduled in early infancy were refused less frequently. When several vaccines were co-administered at a single visit, their monthly trajectories tended to align with one another.
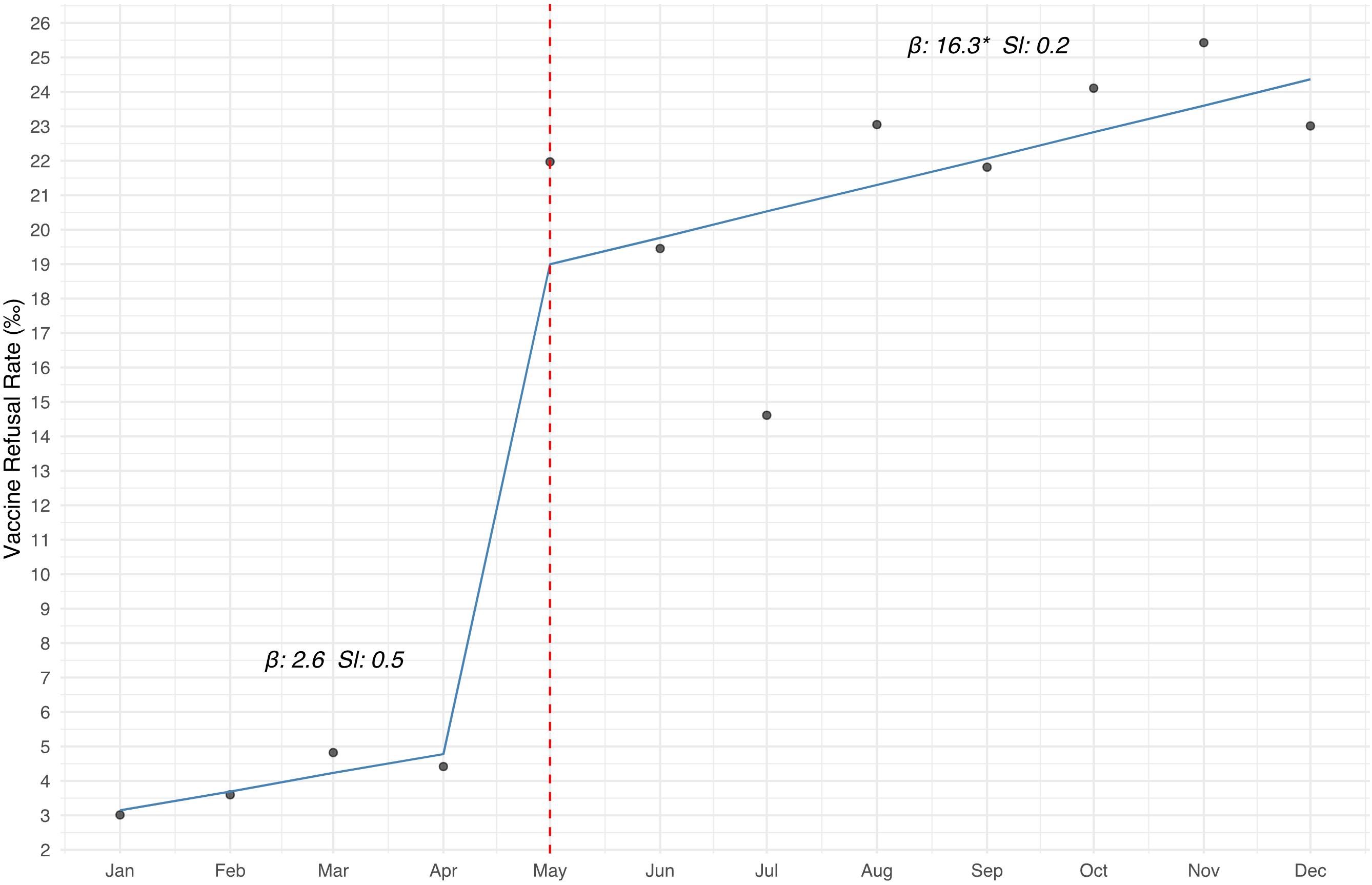
Finally, segmented-regression (Model 1) output quantifies the regulatory effect (Fig. 3). Prior to May, the baseline level was beta coefficient (β) = 2.6‰. After the reform, the level increased significantly to β = 16.3‰, a net rise of 13.7 per mille points (p = 0.005). The within-period slopes, which test whether rates continued to climb from the beginning to the end of each segment, were 0.5‰ and 0.2‰ per month, respectively, and neither reached statistical significance. Complete parameters for Model 1 and the vaccine-type-specific Model 2 are presented in the Supplementary Table S11.
Discussion
This study provides the first comprehensive quantification of vaccine refusal rates using systematically documented health system data from Türkiye. Following the implementation of the policy, vaccine refusals were documented at a rate of 2.1%. This represented a substantial increase in refusals, suggesting that previous underreporting rather than a genuine escalation was the cause, as evidenced by the stable pre- and post-policy slopes. Refusal rates were higher for booster and later-scheduled doses compared to primary series vaccinations. Vaccine refusals accounted for 83.8% of all documented vaccination exceptions, underscoring their major contribution to immunization gaps.
Our study reveals markedly higher vaccine refusal rates in Balıkesir during May-December 2024 (aggregate 21.6‰) compared to previous family medicine-based surveillance studies in Türkiye, which typically reported rates between 2‰–6‰.7,13,18,19 Pre-policy (January-April) rates (3.9‰) aligned with these earlier estimates. The sharp May increase suggests that mandatory reporting uncovered substantial pre-existing refusal previously underrepresented in performance-based data. As pre-policy records might be systematically unreliable, extending the baseline backward was unfeasible; however, this sharp level shift itself serves to quantify the magnitude of the previous surveillance gap. Post-May rates11,12 converge with community-based surveys, indicating higher underlying refusal, and highlighting the importance of systematic surveillance over voluntary reporting systems.20
From a health management perspective, the May 2024 reform exposed flaws in Türkiye’s performance-based financing (PBF) system. International evidence suggests that PBF incentives alone are often costly and unreliable, supporting verifiable data over pay-based exemptions.21 While intended to improve quality, performance mechanisms undermined data authenticity when clinicians faced pressure without support, triggering resistance regarding feasibility.22 This tension highlights broader challenges: immunization monitoring faces indicator overload and reporting burden, yet disease threats demand stable surveillance.23 Patients need trustworthy protection, providers reasonable accountability, and administrators reliable systems. Navigating these competing interests requires the system to evolve, prioritizing the overriding benefit of public health.
Regarding international contextualization, our documented 2.1% vaccine dose refusal rate demands evaluation within contemporary global vaccination challenges. Our findings align closely with the Netherlands (approximately 2%)24 while remaining substantially below rates reported in Austria (2.7–5.9%)25, Brazil (2.7%)26, and Pakistan (22%).27 WHO European Region surveillance data demonstrate concerning regional patterns, with MMR second-dose coverage declining from 92% to 91% between 2019–202428, while more than half of member states now fail to meet the 95% herd immunity elimination target, with some countries reporting coverage below 82%.29 While direct comparisons have limitations due to methodological differences, within this regional context, our refusal rate of 2.1%, appears relatively well-contained. Nevertheless, even modest increases in these rates could erode existing herd immunity margins and compromise disease elimination efforts.
While unmet vaccination reasons in Canada and Brazil primarily involve access barriers or vaccine shortages, our study identified refusal as the predominant factor.30,31 For instance, timely vaccination rates for free vaccines in Austria have been reported to range from 6.3% to 48.8% by vaccine type25, whereas our analysis indicates that post-policy timely vaccination coverage in Türkiye remains over 97.5%. This high rate of on-schedule vaccination suggests that transportation and vaccine logistics challenges have been relatively resolved, reinforcing that refusal is the most significant remaining challenge, particularly following post-COVID-19 trends in hesitancy.3 Our findings further demonstrate that refusal rates were consistently higher for booster and later-scheduled doses compared to the primary series. This pattern carries particular public health significance for measles-containing vaccines, where our data revealed 2.7% refusal for 48-month vaccinations after May, escalating to 4.7% for MMR II by December 2024. Given that coverage rates exceeding 95% are critical for maintaining measles elimination status32, these refusal trends approach thresholds that could compromise herd immunity, potentially creating outbreak-susceptible populations not only in Balıkesir but likely across other Turkish regions. This concerning trajectory likely reflects several interconnected factors identified in prior literature, including parents’ diminished perception of disease risk as children age33, accumulated concerns about safety and potential side effects from previous doses34, and the emergence of vaccine fatigue35 following repeated immunization encounters. Moreover, Turkish qualitative research reveals that vaccine refusal often clusters with broader rejection of healthcare services36, suggesting systematic mistrust that extends beyond vaccination decisions. This pattern may be further amplified in vulnerable populations, as specialized clinic studies demonstrate a fourfold increase in refusal among younger siblings of children with autism spectrum disorder37, indicating how specific family experiences can cascade into broader immunization hesitancy.
This study’s major strength lies in providing the first systematic, population-based quantification of dose-specific vaccine refusal rates using comprehensive health system surveillance data, unlike previous survey-based approaches. The interrupted time-series regression analysis enabled the precise assessment of temporal trends following policy implementation. However, several limitations warrant consideration. The ecological study design and single province setting limit the generalizability of our findings to the broader Turkish population. Additionally, denominators were based on projections, which could affect rate precision, though they were anchored to known annual population data. The data also lacked sociodemographic details, preventing an analysis of refusals across different strata. Future individual-level research is suggested to explore these factors and validate the findings nationally. Furthermore, the exclusion of hospital-administered Hepatitis B I and Td vaccines may underestimate the complete picture of refusal patterns across all delivery settings. Finally, while the four-month baseline might be sufficient to detect the substantial level shift, the limited statistical power for trend analysis warrants cautious interpretation of slope estimates.
Vaccine refusals constitute a substantial proportion of immunization gaps, making their resolution critical for improving coverage. This study highlights the importance of the post-May regulatory reform in Türkiye, which enabled more systematic detection of vaccine refusals. In an era of declining vaccine confidence, comprehensive surveillance systems become indispensable for enabling early trend detection, rapid misinformation response, and tailored interventions for at-risk populations.38 Enhanced interventions targeting refusals should leverage integrated healthcare approaches, as other research suggests that postnatal care utilization and maternal education strongly correlate with vaccine acceptance—making these more effective than vaccination-only strategies.39
While our analysis suggests overall stability during the post-May period, refusal rates for certain later doses approached critical herd immunity thresholds. This pattern raises legitimate concerns about the potential impact of increasing vaccine hesitancy, particularly within the Balıkesir region. Enhanced interventions are needed for later doses, and nationwide long-term surveillance is essential to determine broader patterns, as refusal rates likely vary across Türkiye’s diverse socioeconomic regions. Future nationwide surveillance should incorporate sociodemographic stratification to identify regional variations and enable geographically targeted public health responses.
Supplementary materials
Supplementary materials for this article are available online at https://doi.org/10.24953/turkjpediatr.2026.6792.
Ethical approval
This study constitutes a secondary analysis of publicly available, anonymized, and aggregated data. The datasets are made accessible through the official website of the Ministry of Health in accordance with applicable regulations and legislation; specific data links are available in the supplementary material. Given the use of non-identifiable administrative surveillance data that are publicly disclosed, this research is exempt from formal ethics committee approval.
Source of funding
The authors declare the study received no funding.
Conflict of interest
The authors declare that there is no conflict of interest.
References
- Shattock AJ, Johnson HC, Sim SY, et al. Contribution of vaccination to improved survival and health: modelling 50 years of the Expanded Programme on Immunization. Lancet 2024; 403: 2307-2316. https://doi.org/10.1016/S0140-6736(24)00850-X
- Galagali PM, Kinikar AA, Kumar VS. Vaccine Hesitancy: Obstacles and Challenges. Curr Pediatr Rep 2022; 10: 241-248. https://doi.org/10.1007/s40124-022-00278-9
- Aguinaga-Ontoso I, Guillen-Aguinaga S, Guillen-Aguinaga L, et al. The Impact of COVID-19 on DTP3 Vaccination Coverage in Europe (2012-2023). Vaccines (Basel) 2024; 13: 6. https://doi.org/10.3390/vaccines13010006
- Abenova M, Shaltynov A, Jamedinova U, Semenova Y. Worldwide Child Routine Vaccination Hesitancy Rate among Parents of Children Aged 0-6 Years: A Systematic Review and Meta-Analysis of Cross-Sectional Studies. Vaccines (Basel) 2023; 12: 31. https://doi.org/10.3390/vaccines12010031
- Eryurt MA, Yalçin SS. Zero-dose children in Turkey: regional comparison of pooled data for the period 1990 to 2018. BMC Infect Dis 2022; 22: 421. https://doi.org/10.1186/s12879-022-07416-0
- Gür E. Vaccine hesitancy - vaccine refusal. Turk Pediatri Ars 2019; 54: 1-2. https://doi.org/10.14744/TurkPediatriArs.2019.79990
- Kurt O, Küçükkelepçe O, Öz E, Doğan Tiryaki H, Parlak ME. Childhood Vaccine Attitude and Refusal among Turkish Parents. Vaccines (Basel) 2023; 11: 1285. https://doi.org/10.3390/vaccines11081285
- Birinci Ş. A Digital Opportunity for Patients to Manage Their Health: Turkey National Personal Health Record System (The e-Nabız). Balkan Med J 2023; 40: 215-221. https://doi.org/10.4274/balkanmedj.galenos.2023.2023-2-77
- Karaşahin EF, Turfan O, İbişoğlu B, Karaşahin Ö, Bedir G. A Rural-Urban Comparison of Performance-Based Primary Health Care Services in Erzurum. International Journal of Health Services Research and Policy 2023; 8: 126-138. https://doi.org/10.33457/ijhsrp.1294640
- Hacettepe University Institute of Population Studies. 2018 Turkey Demographic and Health Survey. 2019. Available at: https://dhsprogram.com/publications/publication-FR372-DHS-Final-Reports.cfm (Accessed on July 22, 2025).
- Özlem A, Özlem A, Koçak M, Mehmet N. Prevalence of Turkish Parent Refusal and Hesitance Rate and Its Associated Factors in Childhood Vaccination. Malaysian Journal of Health Sciences 2022; 20: 11-22. https://doi.org/10.17576/JSKM-2022-2002-02
- Soysal G, Akdur R. Investigating Vaccine Hesitancy and Refusal Among Parents of Children Under Five: A Community-based Study. The Journal of Current Pediatrics 2022; 20: 339-348. https://doi.org/10.4274/jcp.2022.01488
- Yalçin SS, Kömürlüoğlu A, Topaç O. Rates of childhood vaccine refusal in Turkey during 2016-2017: Regional causes and solutions. Arch Pediatr 2022; 29: 594-598. https://doi.org/10.1016/j.arcped.2022.06.005
- TURKSTAT. The Results of Address Based Population Registration System, 2024. 2025. Available at: https://data.tuik.gov.tr/Bulten/Index?p=The-Results-of-Address-Based-Population-Registration-System-2024-53783
- Health Directorate of Balıkesir. Performance Exception Forms. 2025. Available at: https://balikesirism.saglik.gov.tr/arama?_Dil=1&p=performans (Accessed on Mar 24, 2025).
- TURKSTAT. Birth Statistics, 2023. 2024. Available at: https://data.tuik.gov.tr/Bulten/Index?p=Birth-Statistics-2023-53708
- Republic of Türkiye Ministry of Health. Performance Exception Committee Procedures and Principles “Performans İtiraz Komisyonu Usulü ve Esasları”. 2024. Available at: https://archive.md/zPHda (Accessed on Mar 18, 2025).
- Baysan C, Yavaş SP, Uğur Karabat M. Examination of parents refusing administration of childhood vaccinations: Turkey example. CHILDS Health 2021; 16: 218-224. https://doi.org/10.22141/2224-0551.16.3.2021.233906
- Terzi Ö, Gülen EN, Dündar C. The causes of parental vaccine refusal: results of a survey from Giresun, Turkey. Turk J Pediatr 2021; 63: 618-625. https://doi.org/10.24953/turkjped.2021.04.009
- ECDC. Immunisation information systems in the EU and EEA - Results of a survey on implementation and system characteristics. Stockholm: ECDC; 2017. Available at: https://www.ecdc.europa.eu/en/publications-data/immunisation-information-systems-eu-and-eea
- Turcotte-Tremblay AM, Spagnolo J, De Allegri M, Ridde V. Does performance-based financing increase value for money in low- and middle- income countries? A systematic review. Health Econ Rev 2016; 6: 30. https://doi.org/10.1186/s13561-016-0103-9
- Aile Hekimleri Dernekleri Federasyonu. Aşı Redleri Konusunda Bakanlığa Başvurumuzu Yaptık. 2024. Available at: https://www.ahef.org.tr/detay.aspx?ID=3194 (Accessed on Oct 27, 2025).
- Patel C, Rendell N, Sargent GM, et al. Measuring National Immunization System Performance: A Systematic Assessment of Available Resources. Glob Health Sci Pract 2023; 11: e220055. https://doi.org/10.9745/GHSP-D-22-00555
- Bussink-Voorend D, Hulscher MEJL, Akkermans R, Krom A, Hautvast JLA. Vaccine hesitancy among Dutch parents of newborn children and its associated characteristics, values and uptake of vaccines. Vaccine 2025; 59: 127244. https://doi.org/10.1016/j.vaccine.2025.127244
- Magyar R, Voitl PK, Voitl JJM, Diesner-Treiber SC. Vaccine hesitancy among parents of children in their first two years of life. Front Public Health 2024; 12: 1438737. https://doi.org/10.3389/fpubh.2024.1438737
- Macedo TRO, Borges MFSO, Silva IFD, et al. Vaccination coverage, barriers and vaccine hesitancy in children up to 24 months old: a population survey in a state capital in the Western Amazon. Epidemiol Serv Saude 2024; 33(spe2): e20231295. https://doi.org/10.1590/s2237-96222024v33e20231295.especial2.en
- Hakim M, Ali F, Zala , Pervaiz A, Afaq S, Haq ZU. Prevalence and associated factors of parental refusal rates for routine immunisation: a cross-sectional study in Peshawar, Khyber Pakhtunkhwa, Pakistan-2024. BMC Public Health 2025; 25: 369. https://doi.org/10.1186/s12889-025-21388-1
- Bhatnagar B, Diallo G. Childhood vaccination rates lag in Europe - fueling further resurgence of measles and whooping cough. Copenhagen, Geneva: WHO Joint News Release; 2025. Available at: https://www.who.int/europe/news/item/15-07-2025-childhood-vaccination-rates-lag-in-europe---fueling-further-resurgence-of-measles-and-whooping-cough (Accessed on Aug 7, 2025).
- World Health Organization (WHO). WHO Immunization Data portal - Global: Immunization Data. 2025. Available at: https://immunizationdata.who.int (Accessed on Aug 7, 2025).
- Faria LB, França AP, Moraes JC, Donalisio MR. Vaccination Barriers in Brazil: Exploring Hesitancy, Access, and Missed Opportunities in a Cohort of Children (2017-2018)-National Vaccination Coverage Survey Results (2020-2021). Vaccines (Basel) 2025; 13: 516. https://doi.org/10.3390/vaccines13050516
- MacKay H, Gretton JD, Chyderiotis S, et al. Confidence and barriers: Analysis of factors associated with timely routine childhood vaccination in Canada during the COVID-19 pandemic. Vaccine 2024; 42: 126236. https://doi.org/10.1016/j.vaccine.2024.126236
- Minta AA, Ferrari M, Antoni S, et al. Progress Toward Measles Elimination - Worldwide, 2000-2023. MMWR Morb Mortal Wkly Rep 2024; 73: 1036-1042. https://doi.org/10.15585/mmwr.mm7345a4
- Baumgaertner B, Ridenhour BJ, Justwan F, Carlisle JE, Miller CR. Risk of disease and willingness to vaccinate in the United States: A population-based survey. PLoS Med 2020; 17: e1003354. https://doi.org/10.1371/journal.pmed.1003354
- Gowda C, Dempsey AF. The rise (and fall?) of parental vaccine hesitancy. Hum Vaccin Immunother 2013; 9: 1755-1762. https://doi.org/10.4161/hv.25085
- Su Z, Cheshmehzangi A, McDonnell D, da Veiga CP, Xiang YT. Mind the “Vaccine Fatigue”. Front Immunol 2022; 13: 839433. https://doi.org/10.3389/fimmu.2022.839433
- Yalçin SS, Bakacak AG, Topaç O. Unvaccinated children as community parasites in National Qualitative Study from Turkey. BMC Public Health 2020; 20: 1087. https://doi.org/10.1186/s12889-020-09184-5
- Bağ Ö, Güney SA. Vaccine refusal risk factors among parents of children with autism spectrum disorders. Turk J Pediatr 2023; 65: 218-226. https://doi.org/10.24953/turkjped.2022.291
- Kurpas D, Stefanicka-Wojtas D, Soll-Morka A, et al. Vaccine Hesitancy and Immunization Patterns in Central and Eastern Europe: Sociocultural, Economic, Political, and Digital Influences Across Seven Countries. Risk Manag Healthc Policy 2025; 18: 1911-1934. https://doi.org/10.2147/RMHP.S519479
- Hermawan A, Irawan IR, Widiyanti M, Rosnani R, Arifin H. Association of socio-demographic factors with measles vaccination coverage among Indonesian children aged 12-23 months: a nationwide study. Turk J Pediatr 2025; 67: 304-316. https://doi.org/10.24953/turkjpediatr.2025.5886
License
Copyright (c) 2026 The Author(s). This is an open access article distributed under the Creative Commons Attribution License (CC BY), which permits unrestricted use, distribution, and reproduction in any medium or format, provided the original work is properly cited.















