Abstract
Background. Pseudomonas aeruginosa is an opportunistic pathogen that plays a critical role in chronic lung infections in patients with cystic fibrosis (CF), primarily due to its ability to form biofilms and develop antibiotic resistance. This study aimed to evaluate the biofilm-forming ability and antibiotic resistance profiles of P. aeruginosa isolates obtained from patients with CF, and to investigate the relationship between biofilm production and antimicrobial resistance.
Methods. 151 P. aeruginosa isolates were collected from patients with CF attending a university hospital. Antibiotic susceptibility testing was performed using both broth microdilution and gradient diffusion methods. Phenotypic determination of virulence factors was performed using standard plate assays. Biofilm production was quantified using the crystal violet microtiter plate assay and Minimum Biofilm Eradication Concentration (MBEC) assay. Statistical analysis was performed to evaluate the association between biofilm formation and antibiotic resistance.
Results. The median age of patients with CF was 11.5 years, with 51.7% being female. Although resistance to certain antibiotics was observed, overall resistance rates remained relatively low, with the highest rate being 11%. A total of 30 (19.9%) P. aeruginosa isolates, showing intra-zone growth, were positive for antibiotic tolerance, while 10 (6.6%) of the 151 isolates exhibited hypermutator phenotypes based on the phenotypic hypermutation test. Biofilm evaluation showed that 14% of isolates were strong biofilm producers, 35.8% moderate, and 21.9% weak. 75 P. aeruginosa isolates were assessed for antibiofilm activity using the MBEC assay. Diallyl disulfide alone showed no significant effect. Combined with ciprofloxacin, it reduced minimum biofilm inhibitory concentration (MBIC) in 16% of isolates, while 28% showed increased MBIC, suggesting antagonism. With tobramycin, 22.3% of isolates showed enhanced antibiofilm activity, indicated by a decrease in MBIC.
Conclusion. In our study, while a high level of biofilm production was observed among P. aeruginosa isolates from patients with CF, antibiotic resistance rates were found to be low. These results highlight the need for therapeutic strategies targeting biofilms to improve treatment outcomes in CF-related P. aeruginosa infections. Additionally, our data indicate that low ceftazidime resistance in this cohort supports the use of beta-lactam–based empirical strategies and carbapenem-sparing approaches, while recognizing that these findings may not be directly generalizable beyond the local context.
Keywords: biofilm, crystal violet, cystic fibrosis, MBEC assay, P. aeruginosa, virulence
Introduction
Cystic fibrosis (CF) is the most common life-limiting autosomal recessive genetic disorder with an estimated birth incidence of approximately 1 in 3200.1 CF arises from mutations in both alleles of the cystic fibrosis transmembrane conductance regulator (CFTR) gene, located on the long arm of chromosome 7.2 The defective CFTR protein, which functions as a chloride channel, leads to recurrent and chronic sinus and pulmonary infections in patients with CF.3 Impaired CFTR function in the respiratory tract results in thickened mucus that cannot be effectively cleared by the mucociliary system. Consequently, this promotes chronic infections and triggers persistent inflammation. The accumulation of inflammatory cytokines and secreted products contributes to lung damage and the development of bronchiectasis.4 Throughout their lives, patients with CF experience recurrent respiratory infections, with the spectrum of pathogens evolving over time and with age.5 Staphylococcus aureus is most frequently detected in young children whereas Pseudomonas aeruginosa, Achromobacter spp., Stenotrophomonas maltophilia, and Burkholderia cepacia complex (Bcc) species are more commonly isolated in children and adults. While these bacteria are traditionally considered classical CF pathogens, there is growing recognition of the significance and pathogenic potential of mycobacteria, fungi, and viruses in CF respiratory infections.6
The respiratory tract of patients with CF is particularly prone to colonization by P. aeruginosa which is the most significant and prevalent gram-negative pathogen in this population P. aeruginosa is a rod shaped bacterium belonging to the family Pseudomonadaceae. As an opportunistic pathogen it rarely causes disease in healthy individuals but proliferates easily in immunocompromised patients.7 Metabolically, P. aeruginosa is oxidase positive and does not ferment lactose. However, under anaerobic conditions, such as those present in the CF lung, it can utilize nitrite or nitrate as terminal electron acceptors. In the early stages of infection, P. aeruginosa typically exists as a nonmucoid strain that can either be cleared by the host’s immune system or eradicated through antibiotic therapy.8,9 Over time, however, P. aeruginosa undergoes a phenotypic switch, producing alginate and forming robust biofilms.10 Once biofilms are established, they present a significant challenge to standard antibiotic treatments due to their protective matrix. Therefore, strategies are available to eliminate early infection by using inhaled antibiotics with or without oral quinolones. Consequently, therapeutic strategies have been developed to target early infections, including the use of inhaled antibiotics with or without oral quinolones8. Additionally, the biofilm environment facilitates the emergence of multidrug-tolerant persistent cells which are implicated in the persistence of chronic and recurrent infections in patients with CF.11
A biofilm is a structured community of microorganisms that attaches to both living and nonliving surfaces, encased within a self-produced matrix of extracellular polymeric substances. This matrix is composed of exopolysaccharides, proteins, metabolites, and extracellular DNA. Microbial cells within biofilms exhibit significantly reduced susceptibility to antimicrobial agents and host immune defenses compared to their planktonic counterparts, which grow in liquid suspension.12 In patients with CF, P. aeruginosa is a major cause of chronic lung infections, forming resilient biofilms on lung epithelial cell surfaces. This process involves the secretion of DNA, proteins, and exopolysaccharides, which contribute to the protective nature of the biofilm and the persistence of the infection.13
The aim of this study was to evaluate the antibiotic susceptibility, biofilm-forming capacity, and antibiofilm activity (tobramycin, ciprofloxacin, and garlic extract) of P. aeruginosa isolates obtained from respiratory tract samples (throat swab, sputum, and bronchoalveolar lavage) of patients with CF during routine hospital visits, as well as to identify hypermutator and antibiotic-tolerant phenotypes among the isolates and to assess their associations with relevant clinical parameters.
Materials and Methods
This study was approved by Hacettepe University Health Sciences Research Ethics Committee (Date: March 30, 2021, Decision No: 2021/07-11).
Patient population
Demographic characteristics of patients and clinical samples (age, gender, follow-up period of patients, body mass index, sample type) and potential risk factors (diagnosis of concomitant infection, diagnosis of other concomitant disease (diabetes, etc.), antibiotic use (frequency of antibiotic use within the past year), number of acute pulmonary exacerbations in a year and history of hospitalization, date of application to CF outpatient clinic) at the time of the first P. aeruginosa isolation were prospectively recorded for each patient using a standardized patient information form. Pediatric patients with CF who had either an initial P. aeruginosa isolation or established P. aeruginosa colonization were included in the study.
Definition and referencing of clinical severity
In this study, we used the definition for Pulmonary Exacerbation based on signs and symptoms characterized as major criteria or minor criteria, those who required hospitalization and intravenous (IV) antibiotic and/or oxygen/respiratory support treatment were considered to have severe and those treated at home with oral antibiotics were defined as mild exacerbations.14
Imaging methods and bronchiectasis ascertainment
Thorax high-resolution computed tomography (HRCT) is the gold standard for the detection and characterization of bronchiectasis and atelectasis in patients with CF. In this study, HRCT examinations were routinely performed between January 2021 and May 2023 and interpreted by two experienced pediatric radiologists.
Bacterial isolates
This prospective study included 151 patients with CF and P. aeruginosa (n=151) isolates grown in respiratory tract samples (throat swab, sputum) taken during both disease episodes and routine hospital visits in patients who were followed up at the Department of Pediatric Pulmonology at Hacettepe University Children’s Hospital between January 2021 and May 2023. Species identification of P. aeruginosa was performed with the matrix-assisted laser desorption/ionization time of flight mass spectrometry (MALDI-TOF MS/Phoenix/ (Bruker, Germany)) system. Species identification in all isolates was confirmed with conventional methods (Gram staining, oxidase test and inoculation in triple sugar iron agar). Following species identification and confirmation of P. aeruginosa isolates, all isolates were stored in tryptic soy broth supplemented with glycerol and stored at -20 °C for the duration of the study.
Antibiotic susceptibility testing
Antibiotic susceptibility testing was performed using both broth microdilution and gradient diffusion methods. Mueller-Hinton broth (MHB) (Sigma Aldrich, USA) and Mueller-Hinton agar (MHA) (BD, USA) media were used for antimicrobial susceptibility testing of all isolates. For the broth microdilution method, the following antibiotic powders were utilized: tobramycin (minimum inhibitory concentration [MIC] range = 0.016-256 mg/L, Thermo scientific-USA), meropenem (MIC range: 0.002-32 mg/L, Thermo scientific-USA), ceftazidime (MIC range: 0.016-256 mg/L, Thermo scientific-USA), levofloxacin (MIC range = 0.002-32 mg/L, Cayman-USA) and colistin sulfate (MIC range = 0.125-256 mg/L, Cayman-USA). For gradient diffusion testing, piperacillin-tazobactam (Oxoid, UK) gradient strips were used, and P. aeruginosa ATCC 25853 served as the quality control strain.
Phenotypic determination of virulence
Determination of antibiotic-tolerant phenotypes in P. aeruginosa isolates from CF patients (TD test)
All isolates included in the study (n=151) were adjusted to a bacterial suspension of 106-107 CFU/mL. The suspensions were then inoculated onto Luria-Bertani (LB) agar plates, following a procedure similar to the disk diffusion method. A tobramycin antibiotic disk was placed at the center of each plate and the plates were incubated at 37 °C overnight. After 24 hours, the antibiotic disk was removed and a disk containing 2 mg of glucose was placed in the same position, followed by an additional overnight incubation. The absence of intra-zone bacterial growth following the first incubation and the appearance of intra-zone regrowth after the second incubation were interpreted as the presence of tolerant/persistent cells.15
Determination of hypermutator phenotypes among P. aeruginosa isolates in CF patients
Bacterial isolates were subcultured on blood agar and incubated at 37 °C for 24 hours. Following incubation, bacterial suspensions were prepared and serially diluted from 10-110-8. The undiluted, 10-1 and 10-2 bacterial suspensions were inoculated onto MHA containing 300 µg/mL rifampicin, while the 10-7 and 10-8 dilutions were inoculated onto MHA without antibiotics. All plates were incubated at 37 °C for 24 hours and colony counts were performed. The hypermutation frequency was calculated as the ratio of the total number of colonies on rifampicin-containing plates to the total number of colonies on antibiotic-free plates.16
Assessment of biofilm formation using the crystal violet assay
Biofilm formation was assessed using the standard crystal violet microtiter plate assay, as previously described. Briefly, bacterial suspensions were inoculated into 96-well microplates and incubated to allow biofilm formation. After washing to remove non-adherent cells, biofilms were stained with crystal violet, and the optical density was measured at 590 nm to quantify biofilm production. The optical density cutoff value (ODc) was calculated as three standard deviations (SD) above the mean OD value of the negative control. ODc= mean OD of the negative control + (3 x SD negative control). Based on this threshold, biofilm production was categorized as follows: strong biofilm formation (4 x ODc <OD), moderate biofilm formation (2 x ODc <OD≤4 x ODc); weak biofilm formation (ODc <OD≤2 x ODc); and non-biofilm (OD≤ODc) according to their optical densities.17
Evaluation of antibiofilm efficacy using calgary biofilm device
A total of 75 P. aeruginosa isolates, previously categorized based on crystal violet staining as strong, moderate or weak biofilm producers, along with tobramycin-resistant non-biofilm-forming isolates, were selected for assessment of antibiofilm activity against various agents using the Minimum Biofilm Eradication Concentration Assay™ (MBEC Assay) (Physiology and Genetics, P&G, Innovotech Inc., Edmonton, AB, Canada). Initially, all isolates were grown overnight in Tryptic soy broth (TSB) medium at 37 °C. The following day, overnight cultures were adjusted to an optical density at 600 nm (OD600) of 0.1 (105 CFU/mL) using TSB medium and subsequently subjected to a 1:1000 dilution in TSB medium. A volume of 150 µL of the diluted bacterial suspension was added to the wells of sterile 96-well microtiter plates, each fitted with a polystyrene microtiter peg lid (MBEC Assay). Plates were incubated at 37 °C with shaking at 110 rpm for 24 hours to allow biofilm formation on the pegs. After incubation, the peg lids were removed and washed three times in 150 μL sterile distilled water to eliminate planktonic bacteria. The peg lids were transferred to new 96-well microtiter plates (antibiotic challenge plates) containing 200 μL per well of different concentrations of ciprofloxacin, colistin or diallyl disulfide. In columns 1 to 11, the tested concentration ranges of ciprofloxacin, colistin and diallyl disulfide were 128 mg/L, 128 mg/L and 512 mg/L, respectively. Antibiotic containing plates with peg lids were incubated overnight at 37 °C with shaking at 110 rpm. Following antibiotic exposure, the peg lids were again washed three times in microtiter plates containing 150 μL sterile distilled water and transferred to fresh 96-well plates containing 200 μL TSB medium in each well. Biofilms were dislodged from the pegs via sonication for 30 minutes and plates were sealed with regular flat lids and incubated at 37°C for another 24 hours under the same shaking conditions at 110 rpm (recovery plates). Biofilm regrowth was quantified by measuring optical density at 650 nm (OD650) using a microplate reader enzyme-linked immunosorbent assay (ELISA). Minimum biofilm inhibitory concentration (MBIC) was defined as the lowest concentration of a compound that inhibits biofilm formation.
Statistical analyses
Statistical analyses were performed using the SPSS version 25.0 software package (IBM, SPSS, Chicago, IL, USA). The normality of data distribution was assessed both analytically (Kolmogorov–Smirnov and Shapiro–Wilk tests) and visually (via histograms and probability plots). Categorical variables were presented as absolute and relative frequencies. Continuous variables were expressed as mean ± standard deviation (SD) if normally distributed, or as median and interquartile range (IQR) if not. Changes in forced expiratory volume in 1 second (FEV1; initial and final values) were evaluated using the Wilcoxon signed-rank test, as the data were not normally distributed. The chi-square test was applied to assess the association between antibiotic resistance and biofilm production. A p-value of <0.05 was considered statistically significant.
Results
Patients and demographic characteristics
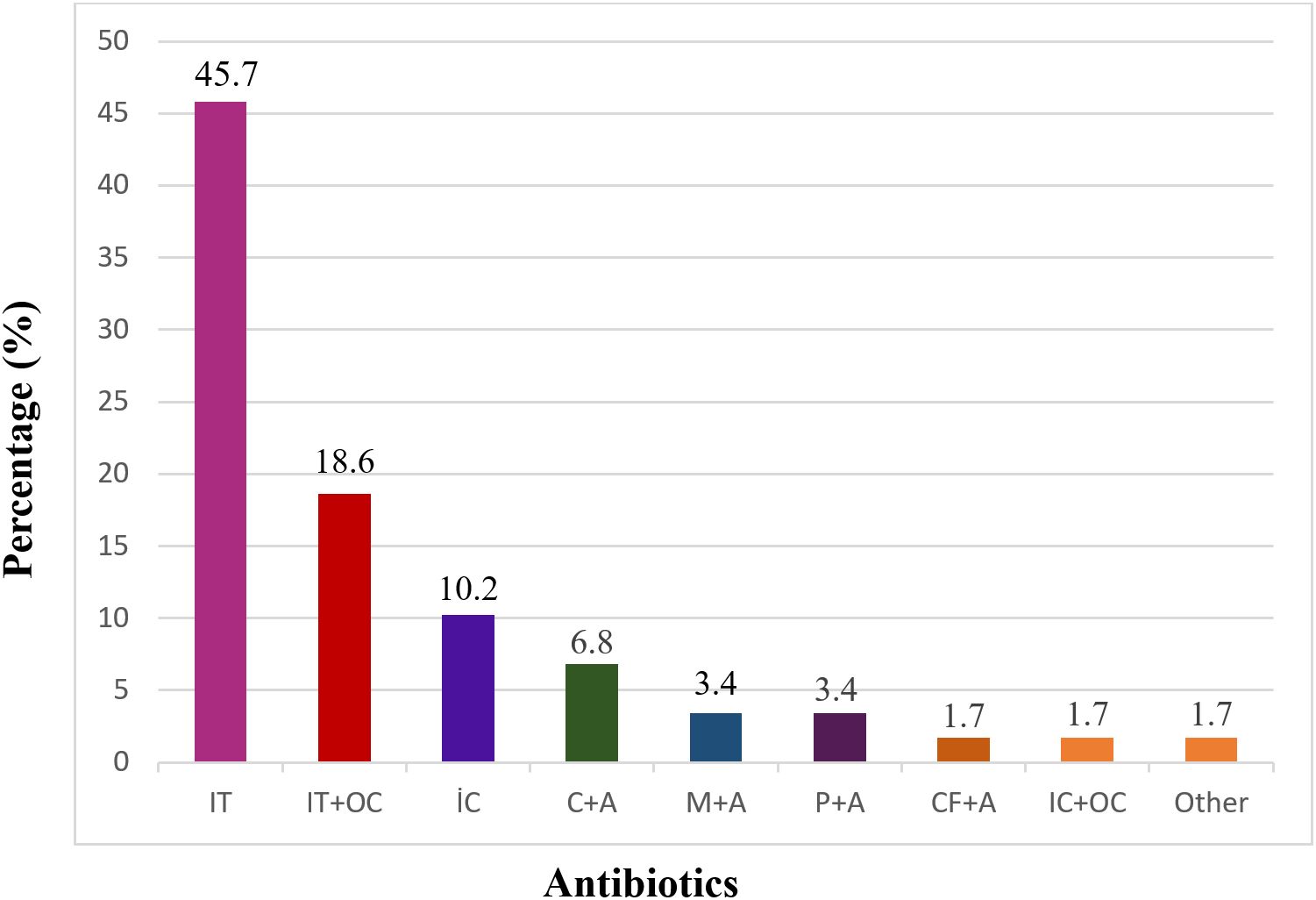
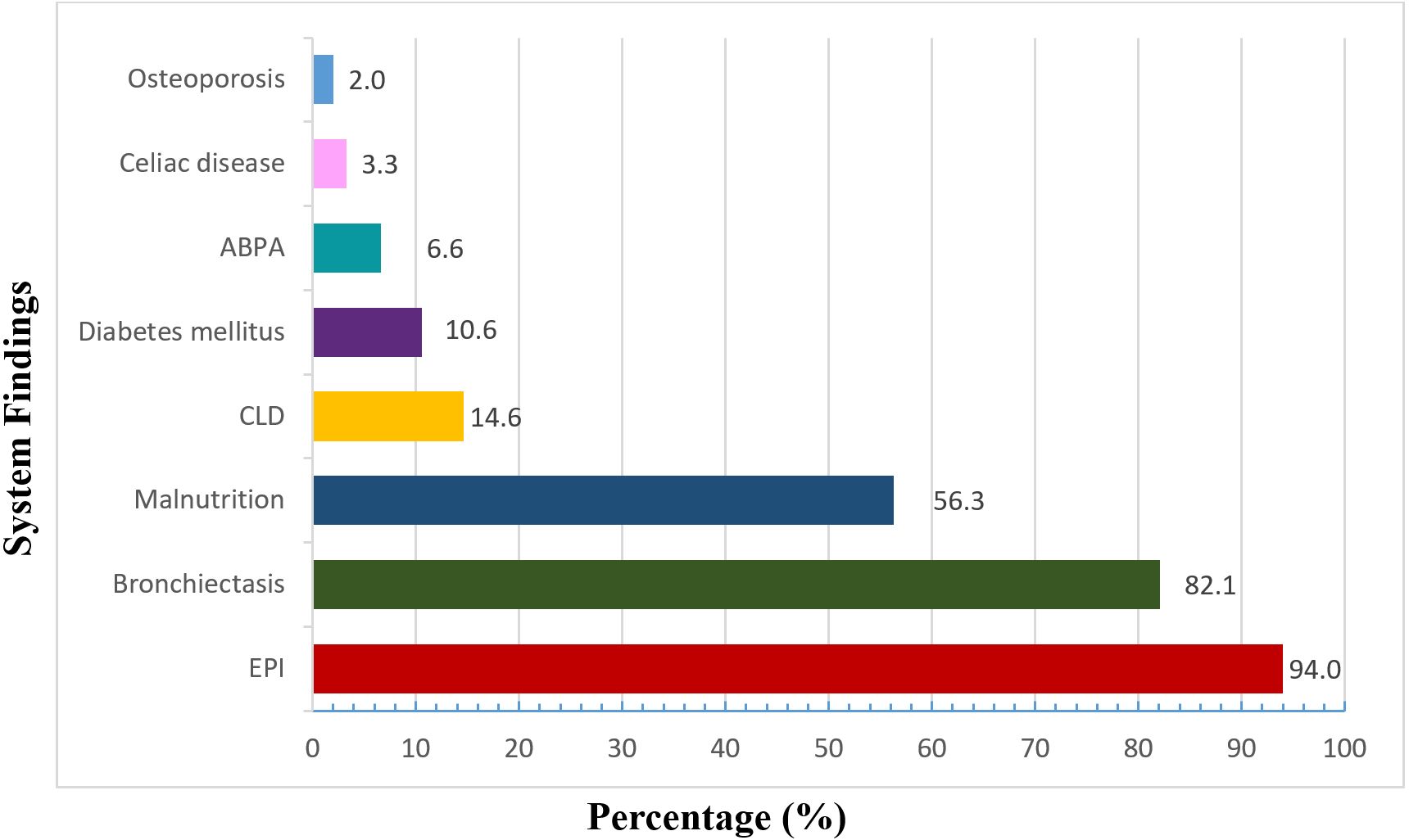
The demographic and clinical characteristics of the pediatric patients with CF are summarized in Table I. Briefly, the cohort consisted of both male and female patients with a median age in childhood and a median body mass index within the expected range for CF. Approximately one-third of the patients experienced at least one acute pulmonary exacerbation during follow-up, most of which were mild. P. aeruginosa was isolated predominantly from sputum samples, and chronic infection was more frequent than initial isolation. No significant difference was observed between initial and final FEV1 values during the follow-up period (p = 0.2). Eradication regimens used in patients with initial P. aeruginosa isolation were summarized in Fig. 1. Inhaled tobramycin, either alone or in combination with oral ciprofloxacin, was the most frequently preferred treatment, while alternative IV and inhaled regimens were used in a smaller proportion of patients. No eradication therapy was initiated in a limited number of cases due to unavailable follow-up data. The distribution of comorbid systemic manifestations among the patients is shown in Fig. 2. Exocrine pancreatic insufficiency (EPI) and bronchiectasis were the most frequently observed conditions, while malnutrition and other systemic comorbidities were detected at lower frequencies.
| FEV1: forced expiratory volume in 1 second, IQR: interquartile range, SD: standard deviation. | |
| Table I. Demographic and clinical characteristics of patients with cystic fibrosis and Pseudomonas aeruginosa infection (N=151). | |
| Gender, n (%) | |
| Male |
|
| Female |
|
| Age, years, (SD: 4.8) |
|
| Acute pulmonary exacerbation, n (%) |
|
| Mild |
|
| Severe |
|
| Specimen type, n (%) | |
| Oropharyngeal swab |
|
| Sputum |
|
| Initial vs. chronic infection | |
| Initial infection, n (%) |
|
| Chronic infection, n (%) |
|
| Colonization time, months, median (IQR) |
|
| FEV1 | |
| Initial FEV1 between 60% and 95% of predicted, n (%) |
|
| Final FEV1 between 60% and 100% of predicted, n (%) |
|
Antibiotic susceptibility testing results
The MIC range, MIC50 and MIC90 values and resistance rates of P. aeruginosa isolates against tobramycin, meropenem, ceftazidime, levofloxacin, colistin sulfate and piperacillin-tazobactam (PIP+TAZ) are given in Table II.
| MIC: minimum inhibitory concentration, MIC50: the lowest antibiotic concentration that inhibits 50% of the tested isolates, MIC90: the lowest antibiotic concentration that inhibits 90% of the tested isolates, PIP+TAZ: piperacillin-tazobactam. | ||||
| Table II. MIC50, MIC90 values, MIC distribution and resistance rate of the isolates obtained from antibiotic susceptibility testing (N = 151). | ||||
| Antimicrobial drug |
|
|
|
|
| Tobramycin |
|
|
|
|
| Ceftazidime |
|
|
|
|
| Levofloxacin |
|
|
|
|
| Colistin sulfate |
|
|
|
|
| PIP+TAZ |
|
|
|
|
| Meropenem |
|
|
|
|
Detection of antibiotic-tolerant and hypermutator phenotypes in P. aeruginosa isolates
According to the tolerance test results; it was determined that there was intra-zone growth in 30 isolates out of 151 when the tobramycin antibiotic disk was removed and a disk containing 2µg glucose was placed. It was determined that 30 (19.9%) P. aeruginosa isolates showing intra-zone growth had positive tolerance test. Based on the results of the phenotypic hypermutation test, 10 (6.6%) out of 151 isolates were identified as hypermutator phenotypes.
Biofilm production
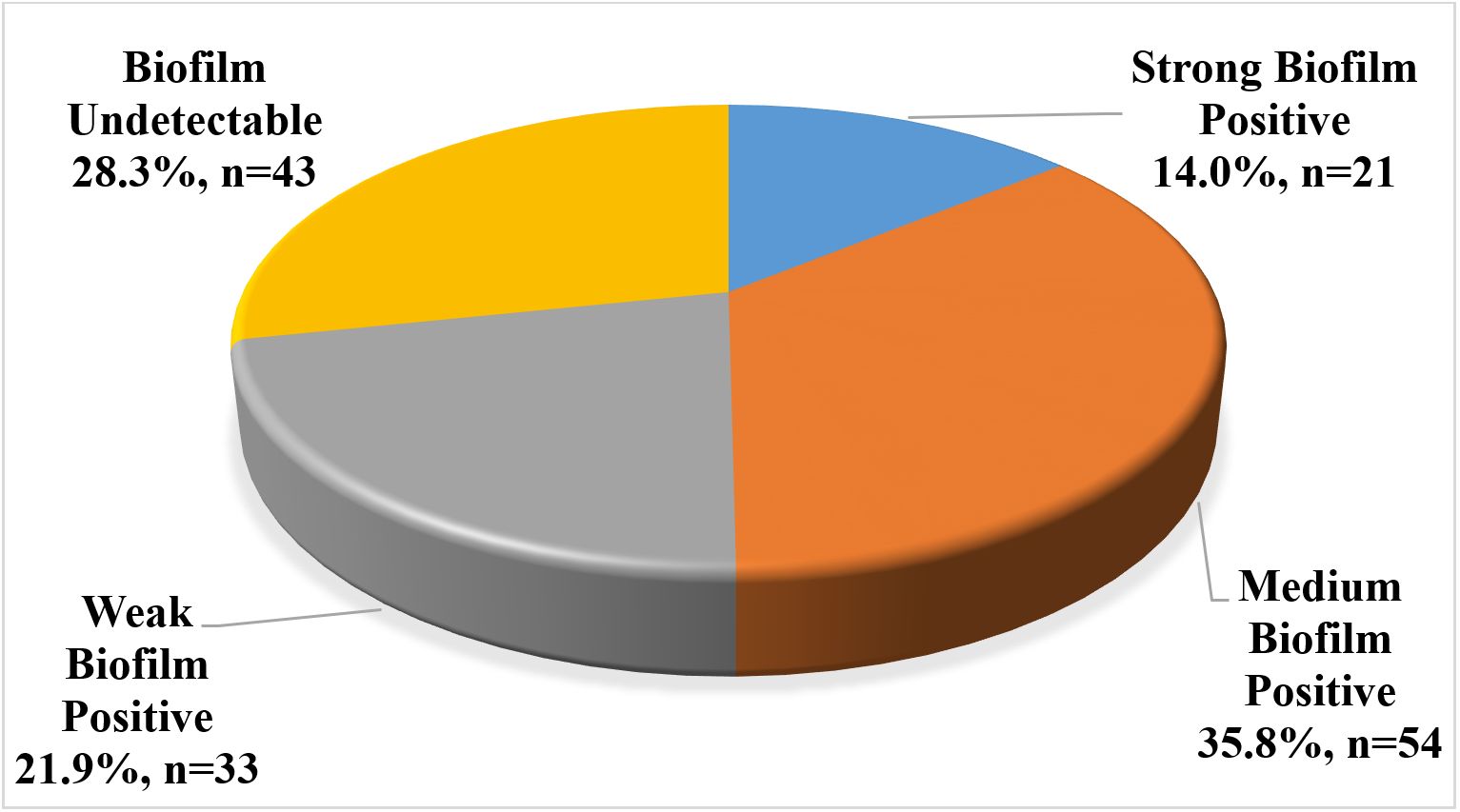
Among the 151 isolates, the biofilm formation was quantitatively detected in 108 isolates based on comparisons with the negative control. Specifically, 21 (14%) isolates exhibited strong biofilm production, 54 (35.8%) were moderate and 33 (21.9%) were weak biofilm producers. The remaining 43 (28.3%) P. aeruginosa isolates were classified as non-biofilm forming (Fig. 3).
No statistically significant difference was observed in biofilm formation according to FEV1 values in either the initial infection or chronic colonization groups, (p>0.05).
Similarly, no significant association was found between FEV1 category (<80% vs. ≥80%) and antibiotic resistance patterns for any of the tested agents. In the chronic colonization group, strong biofilm formation was detected in 31.8% of isolates from patients with FEV1 < 80% and in 43.7% of those with FEV1 ≥ 80%. Overall, resistance rates were low, with the highest frequencies observed for tobramycin and colistin; however, these differences did not reach statistical significance. No statistically significant difference was found in antibiotic resistance rates between strong and weak biofilm-producing P. aeruginosa isolates (p>0.05).
Antibiofilm activity
A total of 75 P. aeruginosa isolates were selected for antibiofilm evaluation using the MBEC assay. These included 21 strong, 40 moderate, and four weak biofilm-producing isolates as determined by the crystal violet staining method, along with 10 non-biofilm-forming isolates that were resistant to tobramycin. The biofilm-forming capacity and susceptibility to diallyl disulfide alone and in combination with antibiotics were assessed.
Diallyl disulfide alone did not demonstrate significant antibiofilm activity against any of the tested P. aeruginosa isolates.
In combination with ciprofloxacin: No change in MBIC values was observed in 42 isolates (56%); an increase in MBIC values was detected in 21 isolates (28%), indicating potential antagonistic interaction; and a decrease in MBIC values, suggestive of synergistic or additive effects, was observed in 12 isolates (16%). Thus, the diallyl disulfide–ciprofloxacin combination was effective in reducing MBIC values in 16% of the tested isolates.
In combination with tobramycin: No change in MBIC values was observed in 36 isolates (48%), an increase in MBIC values was found in 17 isolates (22.6%), and a decrease in MBIC values was observed in 23 isolates (29.3%). Accordingly, the diallyl disulfide–tobramycin combination exhibited an enhanced antibiofilm effect in 22.3% of the total isolates.
Discussion
P. aeruginosa undergoes a characteristic evolutionary adaptation during chronic CF lung infection. Infection of patients with CF occurs via initial colonization of the airway, with accumulation of adaptive mutations in the bacterial genome associated with increased fitness in the lung environment, leading to chronicity. Adaptation of P. aeruginosa to the airway is an important aspect in the progression of CF lung disease.18
P. aeruginosa infection is quite common in patients with CF and this bacteria frequently causes acute pulmonary exacerbations. The rate of acute exacerbations caused by P. aeruginosa may vary depending on the age of the patients, the chronicity of the infection, and the geographical region. It is known that the risk of acute exacerbations is increased especially in patients with chronic P. aeruginosa infection.19
In our study, all patients were followed for a period exceeding one year, during which acute pulmonary exacerbations were identified in 50 individuals (32.5%). Among these patients, 33 experienced mild and 17 exhibited severe pulmonary exacerbations. To date, there are limited studies categorizing acute pulmonary exacerbations. In conclusion, P. aeruginosa infections are highly prevalent among patients with CF and significantly contribute to the risk of acute pulmonary exacerbations, particularly in the case of chronic colonization. These results underscore the necessity of vigilant monitoring and the implementation of targeted therapeutic strategies to effectively manage P. aeruginosa infections. Timely and appropriate interventions are essential to improving both the quality of life and clinical outcomes in patients with CF.
Antibiotic strategies for eradication of P. aeruginosa in patients with CF have been demonstrated in different studies revealing that early eradication therapy led to significantly higher rates of microbiological clearance compared to no anti-pseudomonal treatment after two years of follow-up.19 According to a review published in 2023, early treatment with inhaled tobramycin in patients with CF with initial P. aeruginosa infection was associated with successful microbiological eradication of the microorganism from the respiratory tract samples of the patients. The same review compared oral ciprofloxacin and inhaled colistin with standard treatment revealing that early eradication therapy led to significantly higher rates of microbiological clearance of the microorganism.20
The eradication guidelines of the CF Foundation also recommend inhaled tobramycin as the first line agent for P. aeruginosa eradication. Inhaled colistin can serve as an alternative regimen while oral ciprofloxacin is recommended in addition to inhaled therapies. In cases where the patient exhibits severe symptoms or fails to respond to inhaled and oral treatments, IV antibiotic therapy should be considered. In such cases, a combination of a beta-lactam antibiotic (e.g., ceftazidime) and an aminoglycoside (e.g., tobramycin) is recommended.21
According to the hospital patient record system data, among 59 patients in whom P. aeruginosa was isolated for the first time, the preferred eradication treatments were included inhaled tobramycin alone in 27 patients, inhaled tobramycin and oral ciprofloxacin in 11 patients, inhaled colistin in six patients, ceftazidime and amikacin in four patients, meropenem and amikacin in two patients, piperacillin and amikacin in two patients, cefixime and amikacin in one patient, inhaled colistin and oral ciprofloxacin in one patient, and other treatments in one patient. Four patients did not receive treatment due to a lack of follow-up. Given the complexity and variability in clinical presentation and treatment response, the treatment strategies must be evaluated individually for each patient. Therefore, the management of pediatric patients with CF should be managed by an experienced multidisciplinary team to ensure optimal therapeutic outcomes.
The system manifestations accompanying the disease generally depend on the progression of the disease and the underlying genetic variants. EPI, a gastrointestinal system disorder, is particularly common among patients with CF. A review published in 2023 reported that 85% of children with CF have pancreatic insufficiency, while emphasizing that this rate is similarly high in adults (85-90%).22 In our study, EPI was identified in 142 of the 151 patients, confirming its high prevalence. EPI in CF is typically managed with pancreatic enzyme replacement therapy.
Bronchiectasis is another significant clinical feature observed in patients with CF. Studies have reported a high prevalence of bronchiectasis in patients with CF. For example, a study conducted in Taiwan found radiological evidence of bronchiectasis in 80-90% of patients with CF.23 Consistent with this, bronchiectasis was detected in 124 out of 151 patients in our study.
Malnutrition is also a common concern in pediatric patients with CF and its prevalence in pediatric patients with CF varies depending on age, severity of the disease, and the evaluation criteria used. According to the results of a study conducted in 2014, 22.1% of patients with CF were found to have malnutrition and 13.2% were found to have overweight or obesity.24 In our study, malnutrition was identified in a total of 85 (56.3%) of our patients, highlighting the importance of nutritional monitoring and intervention in CF care.
According to a study conducted in 2014, malnutrition was observed in 22.1% of patients with cystic fibrosis, whereas 13.2% were overweight or obese.24 In our study, malnutrition was detected in 85 patients (56.3%), highlighting the need for careful nutritional assessment and intervention in CF management.
Jarzynka et al. reported that in adult patients with CF, a moderate correlation was found between β-lactam resistance of P. aeruginosa isolates and lung function decline, while biofilm formation per se did not correlate significantly with decreased FEV1. This heterogeneity in bacterial phenotypes, as documented by Jarzynka et al., reinforces our observation that reduced FEV1 may not uniformly associate with strong biofilm or elevated resistance patterns in P. aeruginosa isolates.25 Our findings suggest that reduced lung function is not directly associated with biofilm strength or antibiotic resistance among P. aeruginosa isolates from patients with CF.
P. aeruginosa complicates treatment due to its adaptability and biofilm formation ability. A review published in 2023 revealed that P. aeruginosa isolates obtained from patients with CF were particularly resistant to beta-lactams, and resistance rates to antibiotics such as meropenem, aztreonam, and ceftazidime ranged between 30–40%.26 In another study resistance to ceftazidime was observed in 30–50% of P. aeruginosa isolates.27 In contrast, in our study, ceftazidime resistance was identified in only ten isolates. According to another study, resistance rates to tobramycin among P. aeruginosa isolates in patients with CF ranged from 20% to 40%. This resistance is often attributed to chronic infections and prolonged antibiotic use, making treatment strategies difficult. Therefore, this may necessitate alternative treatment or combination therapies.28 Notably, our findings revealed a low rate of tobramycin resistance, with only 17 isolates exhibiting resistance. In our cohort, ceftazidime resistance was low, suggesting that empirical regimens incorporating ceftazidime or other beta-lactam agents may remain effective in most cases. These findings support the feasibility of carbapenem-sparing strategies, which could help preserve broader-spectrum agents and align with local antimicrobial stewardship goals. However, these results are derived from a single-center cohort, and caution should be exercised according to the patients antimicrobial susceptible testing.
A meta-analysis published in October 2024 evaluating P. aeruginosa colistin resistance rates for 32 countries found the highest resistance rates in Egypt (15%) and Pakistan (13%). Subgroup meta-analyses based on the year of publication indicated that resistance colistin increased modestly from 2% to 3% in 2009.29 In our study, colistin resistance was found to be low, with a MIC rate of 3.3%. In conclusion, this persistent resistance in patients with chronic infections complicates the treatment process and highlights the critical need for early detection, appropriate antimicrobial stewardship and aggressive treatment strategies to improve clinical outcomes in patients with CF.
Researchers investigating antibiotic tolerance have used the TDtest method, which we also applied in our study, to detect antibiotic tolerance in clinical isolates. In a study, TDtest was applied to 20 clinical Escherichia coli isolates specifically and the antibiotic tolerance profiles of these isolates were determined. The results showed that the isolates had varying levels of tolerance to different antibiotics. This study demonstrates that TDtest is a reliable method for detecting antibiotic tolerance in clinical isolates15. In our study, TDtest was performed on 151 P. aeruginosa isolates. Following removal of the tobramycin antibiotic disk and placement of a disk containing 2 µg glucose, intra-zone bacterial regrowth was observed in 30 isolates. These findings indicate that 30 (19.9%) of P. aeruginosa isolates were positive for antibiotic tolerance. The TDtest may serve as a valuable tool for the routine detection of antibiotic tolerance in clinical microbiology laboratories and may contribute to the establishment of more effective and individualized antibiotic treatment regimens for pathogenic bacteria.
Detection of hypermutator phenotype using a rifampicin containing medium is a widely applied phenotypic method, particularly in the evaluation of hypermutator phenotype in P. aeruginosa. A study published in 2016 addresses the detection of hypermutator phenotypes in P. aeruginosa in a selective medium containing rifampicin. In this study, it was observed that the mutant colony formation of hypermutator strains was 100 times more frequent than that of normal strains.30 As a result of the study, examining the hypermutator phenotypes among P. aeruginosa strains obtained from patients with CF in Australia in 2019, a total of 59 P. aeruginosa isolates were examined and 22% of them were detected as hypermutators. In our study, phenotypic hypermutation testing identified 10 (6.6%) out of the 151 isolates as phenotypic hypermutators. The findings emphasize the importance of detecting and monitoring hypermutator phenotypes, as such strains have an increased tendency to develop antibiotic resistance. Early identification of hypermutators in patients with CF may contribute to more effective treatment strategies and the prevention of multidrug resistance.
In the study conducted by Winstanley et al., it was reported that 80% of P. aeruginosa isolates in patients with CF had the ability to form biofilm.31 Similarly in a study conducted in Tehran in 2021, it was found that 76% of P. aeruginosa isolates in CF patients had the ability to form biofilm.32 In our study, using the CV method, biofilm formation was quantitatively demonstrated in 108 (71.5%) of 151 isolates, while 43 (28.3%) P. aeruginosa isolates did not form biofilm. Studies show that bacterial resistance is enhanced in biofilm-forming bacteria, especially in the lungs of patients with CF. Biofilms facilitate prolonged bacterial persistence and increased resistance to antimicrobial treatments.
The Calgary Biofilm Device (CBD) is widely used for the evaluation of antibiofilm susceptibility in patients with CF. This device helps determine antibiotic resistance profiles in biofilm-producing bacterial populations. According to a study conducted at the Biofilm Center in Canada, elevated MIC values were detected for tobramycin and its antibiofilm activity was found to be quite high, indicating that tobramycin alone can prevent biofilm development at certain concentrations.33 In another study published by Chen et al. in 2020, CBD was used to evaluate the effect of tobramycin on 24-hour P. aeruginosa biofilms. It was observed that the bactericidal activity of tobramycin was concentration-dependent rather than time-independent with increased efficacy observed after 72 hours.34
Garlic, recognized for its natural antimicrobial properties, has been shown to be effective against bacteria. In a 2005 study investigating the antibiofilm activity of 1% garlic extract in combination with tobramycin, the combination was effective against most biofilm-forming isolates, while tobramycin alone had minimal effect. Moreover, garlic extract alone showed no significant effect on biofilm viability.35 In another study examining the effect of garlic extract on both biofilm activity, the active compounds in garlic, such as allicin, were found to inhibit P. aeruginosa by disrupting quorum sensing mechanisms. In another study, the active compounds in garlic, particularly allicin, were found to inhibit P. aeruginosa by disrupting quorum sensing mechanisms. Garlic extract reduced bacterial infectivity, impaired biofilm structure formed by P. aeruginosa in the lungs and contributed to biofilm dispersal.36
In our study, the antibiofilm activity of ciprofloxacin alone was found to be effective in 16% of the total P. aeruginosa isolates. Similarly, the combination of garlic extract (diallyl disulfide) with ciprofloxacin was also effective indicating no difference between the combination treatment and the ciprofloxacin alone. However, tobramycin alone showed antibiofilm activity in 29.3% of the isolates, whereas the combination of garlic extract and tobramycin was effective in 22.3% of our total isolates. Furthermore, garlic extract alone demonstrated no detectable effect on biofilm viability but tobramycin alone was more effective. These results highlight the limited synergistic potential of garlic extract in combination therapies and underscore the importance of optimizing antibiotic use for the treatment of chronic biofilm-associated infections. The antibiofilm activity of diallyl disulfide- garlic extract was assessed using in vitro MBEC assays. These results reflect laboratory conditions and do not represent clinical outcomes. Currently, our center does not use any garlic-derived products clinically. Interestingly, 28% of isolates showed increased MBIC values when treated with a combination of ciprofloxacin and diallyl disulfide, suggesting a potential antagonistic interaction. These findings should not be directly extrapolated to clinical practice and are instead considered hypothesis-generating, providing a basis for future preclinical and clinical studies.
Among P. aeruginosa isolates from all patients with CF, 17 (81%) of the strong biofilm producers and 44 (81.5%) of the moderate biofilm producers were found to be susceptible to all antibiotics. These findings suggest that biofilm strength alone may not be an independent predictor of resistance. Eleven (52.4%) of the strong biofilm producers and 24 of the moderate biofilm producers were from patients exhibiting colonization. Examination of the eradication regimens administered to these patients revealed that 25 (71.4%) received inhaled tobramycin therapy. Based on these findings, it was concluded that inhaled tobramycin alone may be an appropriate option for the eradication of P. aeruginosa colonization in patients with CF.
Although no statistically significant correlations were identified between FEV1 values, biofilm formation, and antibiotic resistance, these findings emphasize the complexity of host–pathogen interactions in CF. In line with this, no significant difference was found in antibiotic resistance rates between strong and weak biofilm-producing P. aeruginosa isolates (p > 0.05). Although biofilm formation is recognized as a key virulence factor contributing to antimicrobial tolerance, our results suggest that biofilm strength alone may not be a decisive predictor of antibiotic resistance or pulmonary function decline in CF isolates. These observations highlight that bacterial phenotypic adaptation, including biofilm production and resistance development, likely depends on local microenvironmental and immunological factors rather than solely on lung function status, underscoring the need for future multi-omic studies to clarify how biofilm phenotypes interact with resistance gene expression and clinical outcomes.
Conclusion
In conclusion, this study provides a comprehensive phenotypic characterization of P. aeruginosa isolates obtained from pediatric patients with CF, demonstrating a high prevalence of biofilm formation alongside generally low antimicrobial resistance rates. Although antibiotic resistance, including resistance to colistin and ceftazidime, remained limited in this cohort, the frequent detection of antibiotic-tolerant and hypermutator phenotypes highlights the complexity of managing chronic P. aeruginosa infections in CF. Importantly, no significant associations were identified between biofilm strength, lung function parameters, or antimicrobial resistance, suggesting that biofilm formation alone may not be a reliable predictor of clinical severity or resistance patterns. The observed antibiofilm effects of selected agents under in vitro conditions warrant further investigation but should be interpreted cautiously given the single-center design and limited sample size. Future multicenter and longitudinal studies integrating clinical, microbiological, and molecular data are needed to better define the clinical relevance of these phenotypic traits and to optimize therapeutic strategies for CF–associated P. aeruginosa infections.
Ethical approval
The study was approved by Non-Interventional Ethics Committee of Hacettepe University (date: March 30, 2021, number: 2021/07-11).
Source of funding
This study was supported by the Scientific Research Projects Coordination Unit of Hacettepe University (Project No: TSA-2021-19346).
Conflict of interest
The authors declare that there is no conflict of interest.
References
- Balfour-Lynn IM, Lees B, Hall P, et al. Multicenter randomized controlled trial of withdrawal of inhaled corticosteroids in cystic fibrosis. Am J Respir Crit Care Med 2006; 173: 1356-1362. https://doi.org/10.1164/rccm.200511-1808OC
- Riordan JR, Rommens JM, Kerem B, et al. Identification of the cystic fibrosis gene: cloning and characterization of complementary DNA. Science 1989; 245: 1066-1073. https://doi.org/10.1126/science.2475911
- Boucher RC. New concepts of the pathogenesis of cystic fibrosis lung disease. Eur Respir J 2004; 23: 146-158. https://doi.org/10.1183/09031936.03.00057003
- Cohen TS, Prince A. Cystic fibrosis: a mucosal immunodeficiency syndrome. Nat Med 2012; 18: 509-519. https://doi.org/10.1038/nm.2715
- Ryan C, Ross S, Davey P, et al. Prevalence and causes of prescribing errors: the PRescribing Outcomes for Trainee Doctors Engaged in Clinical Training (PROTECT) study. PLoS One 2014; 9: e79802. https://doi.org/10.1371/journal.pone.0079802
- Konstan MW, Morgan WJ, Butler SM, et al. Risk factors for rate of decline in forced expiratory volume in one second in children and adolescents with cystic fibrosis. J Pediatr 2007; 151: 134-139.e1. https://doi.org/10.1016/j.jpeds.2007.03.006
- Balasubramanian D, Schneper L, Kumari H, Mathee K. A dynamic and intricate regulatory network determines Pseudomonas aeruginosa virulence. Nucleic Acids Res 2013; 41: 1-20. https://doi.org/10.1093/nar/gks1039
- Høiby N, Frederiksen B, Pressler T. Eradication of early Pseudomonas aeruginosa infection. J Cyst Fibros 2005; 4(Suppl 2): 49-54. https://doi.org/10.1016/j.jcf.2005.05.018
- Taccetti G, Campana S, Festini F, Mascherini M, Döring G. Early eradication therapy against Pseudomonas aeruginosa in cystic fibrosis patients. Eur Respir J 2005; 26: 458-461. https://doi.org/10.1183/09031936.05.00009605
- Campodónico VL, Gadjeva M, Paradis-Bleau C, Uluer A, Pier GB. Airway epithelial control of Pseudomonas aeruginosa infection in cystic fibrosis. Trends Mol Med 2008; 14: 120-133. https://doi.org/10.1016/j.molmed.2008.01.002
- Matsui H, Grubb BR, Tarran R, et al. Evidence for periciliary liquid layer depletion, not abnormal ion composition, in the pathogenesis of cystic fibrosis airways disease. Cell 1998; 95: 1005-1015. https://doi.org/10.1016/s0092-8674(00)81724-9
- Worlitzsch D, Tarran R, Ulrich M, et al. Effects of reduced mucus oxygen concentration in airway Pseudomonas infections of cystic fibrosis patients. J Clin Invest 2002; 109: 317-325. https://doi.org/10.1172/JCI13870
- Zabner J, Smith JJ, Karp PH, Widdicombe JH, Welsh MJ. Loss of CFTR chloride channels alters salt absorption by cystic fibrosis airway epithelia in vitro. Mol Cell 1998; 2: 397-403. https://doi.org/10.1016/s1097-2765(00)80284-1
- Anstead M, Saiman L, Mayer-Hamblett N, et al. Pulmonary exacerbations in CF patients with early lung disease. J Cyst Fibros 2014; 13: 74-79. https://doi.org/10.1016/j.jcf.2013.07.006
- Gefen O, Chekol B, Strahilevitz J, Balaban NQ. TDtest: easy detection of bacterial tolerance and persistence in clinical isolates by a modified disk-diffusion assay. Sci Rep 2017; 7: 41284. https://doi.org/10.1038/srep41284
- Oliver A, Cantón R, Campo P, Baquero F, Blázquez J. High frequency of hypermutable Pseudomonas aeruginosa in cystic fibrosis lung infection. Science 2000; 288: 1251-1254. https://doi.org/10.1126/science.288.5469.1251
- O’Toole GA. Microtiter dish biofilm formation assay. J Vis Exp 2011; (47): e2437. https://doi.org/10.3791/2437
- Li Z, Kosorok MR, Farrell PM, et al. Longitudinal development of mucoid Pseudomonas aeruginosa infection and lung disease progression in children with cystic fibrosis. JAMA 2005; 293: 581-588. https://doi.org/10.1001/jama.293.5.581
- Blanchard AC, Waters VJ. Opportunistic pathogens in cystic fibrosis: epidemiology and pathogenesis of lung infection. J Pediatric Infect Dis Soc 2022; 11: S3-S12. https://doi.org/10.1093/jpids/piac052
- Langton Hewer SC, Smith S, Rowbotham NJ, Yule A, Smyth AR. Antibiotic strategies for eradicating Pseudomonas aeruginosa in people with cystic fibrosis. Cochrane Database Syst Rev 2023; 6: CD004197. https://doi.org/10.1002/14651858.CD004197.pub6
- Mogayzel PJ, Naureckas ET, Robinson KA, et al. Cystic Fibrosis Foundation pulmonary guideline. pharmacologic approaches to prevention and eradication of initial Pseudomonas aeruginosa infection. Ann Am Thorac Soc 2014; 11: 1640-1650. https://doi.org/10.1513/AnnalsATS.201404-166OC
- Ong T, Ramsey BW. Cystic fibrosis: a review. JAMA 2023; 329: 1859-1871. https://doi.org/10.1001/jama.2023.8120
- Cheng WC, Chang CL, Sheu CC, et al. Correlating Reiff scores with clinical, functional, and prognostic factors: characterizing noncystic fibrosis bronchiectasis severity: validation from a nationwide multicenter study in Taiwan. Eur J Med Res 2024; 29: 286. https://doi.org/10.1186/s40001-024-01870-z
- Panagopoulou P, Fotoulaki M, Nikolaou A, Nousia-Arvanitakis S. Prevalence of malnutrition and obesity among cystic fibrosis patients. Pediatr Int 2014; 56: 89-94. https://doi.org/10.1111/ped.12214
- Jarzynka S, Makarewicz O, Weiss D, et al. The impact of pseudomonas aeruginosainfection in adult cystic fibrosis patients-a single polish centre study. Pathogens 2023; 12: 1440. https://doi.org/10.3390/pathogens12121440
- Perikleous EP, Gkentzi D, Bertzouanis A, Paraskakis E, Sovtic A, Fouzas S. Antibiotic resistance in patients with cystic fibrosis: past, present, and future. Antibiotics (Basel) 2023; 12: 217. https://doi.org/10.3390/antibiotics12020217
- Ramsay KA, Rehman A, Wardell ST, et al. Ceftazidime resistance in Pseudomonas aeruginosa is multigenic and complex. PLoS One 2023; 18: e0285856. https://doi.org/10.1371/journal.pone.0285856
- Barclay ML, Begg EJ, Chambers ST, Thornley PE, Pattemore PK, Grimwood K. Adaptive resistance to tobramycin in Pseudomonas aeruginosa lung infection in cystic fibrosis. J Antimicrob Chemother 1996; 37: 1155-1164. https://doi.org/10.1093/jac/37.6.1155
- Narimisa N, Keshtkar A, Dadgar-Zankbar L, et al. Prevalence of colistin resistance in clinical isolates of Pseudomonas aeruginosa: a systematic review and meta-analysis. Front Microbiol 2024; 15: 1477836. https://doi.org/10.3389/fmicb.2024.1477836
- Vanderwoude J, Azimi S, Read TD, Diggle SP. The role of hypermutation and collateral sensitivity in antimicrobial resistance diversity of Pseudomonas aeruginosa populations in cystic fibrosis lung infection. mBio 2024; 15: e0310923. https://doi.org/10.1128/mbio.03109-23
- Clark ST, Guttman DS, Hwang DM. Diversification of Pseudomonas aeruginosa within the cystic fibrosis lung and its effects on antibiotic resistance. FEMS Microbiol Lett 2018; 365: fny026. https://doi.org/10.1093/femsle/fny026
- Kodori M, Nikmanesh B, Hakimi H, Ghalavand Z. Antibiotic susceptibility and biofilm formation of bacterial isolates derived from pediatric patients with cystic fibrosis from Tehran, Iran. Arch Razi Inst 2021; 76: 397-406. https://doi.org/10.22092/ari.2020.128554.1416
- Díez-Aguilar M, Morosini MI, Köksal E, Oliver A, Ekkelenkamp M, Cantón R. Use of calgary and microfluidic bioflux systems to test the activity of fosfomycin and tobramycin alone and in combination against cystic fibrosis Pseudomonas aeruginosa biofilms. Antimicrob Agents Chemother 2017; 62: e01650-17. https://doi.org/10.1128/AAC.01650-17
- Chen X, Thomsen TR, Winkler H, Xu Y. Influence of biofilm growth age, media, antibiotic concentration and exposure time on Staphylococcus aureus and Pseudomonas aeruginosa biofilm removal in vitro. BMC Microbiol 2020; 20: 264. https://doi.org/10.1186/s12866-020-01947-9
- Rasmussen TB, Bjarnsholt T, Skindersoe ME, et al. Screening for quorum-sensing inhibitors (QSI) by use of a novel genetic system, the QSI selector. J Bacteriol 2005; 187: 1799-1814. https://doi.org/10.1128/JB.187.5.1799-1814.2005
- Bjarnsholt T, Jensen PØ, Rasmussen TB, et al. Garlic blocks quorum sensing and promotes rapid clearing of pulmonary Pseudomonas aeruginosa infections. Microbiology (Reading) 2005; 151: 3873-3880. https://doi.org/10.1099/mic.0.27955-0
License
Copyright (c) 2026 The Author(s). This is an open access article distributed under the Creative Commons Attribution License (CC BY), which permits unrestricted use, distribution, and reproduction in any medium or format, provided the original work is properly cited.















