Graphical Abstract
Abstract
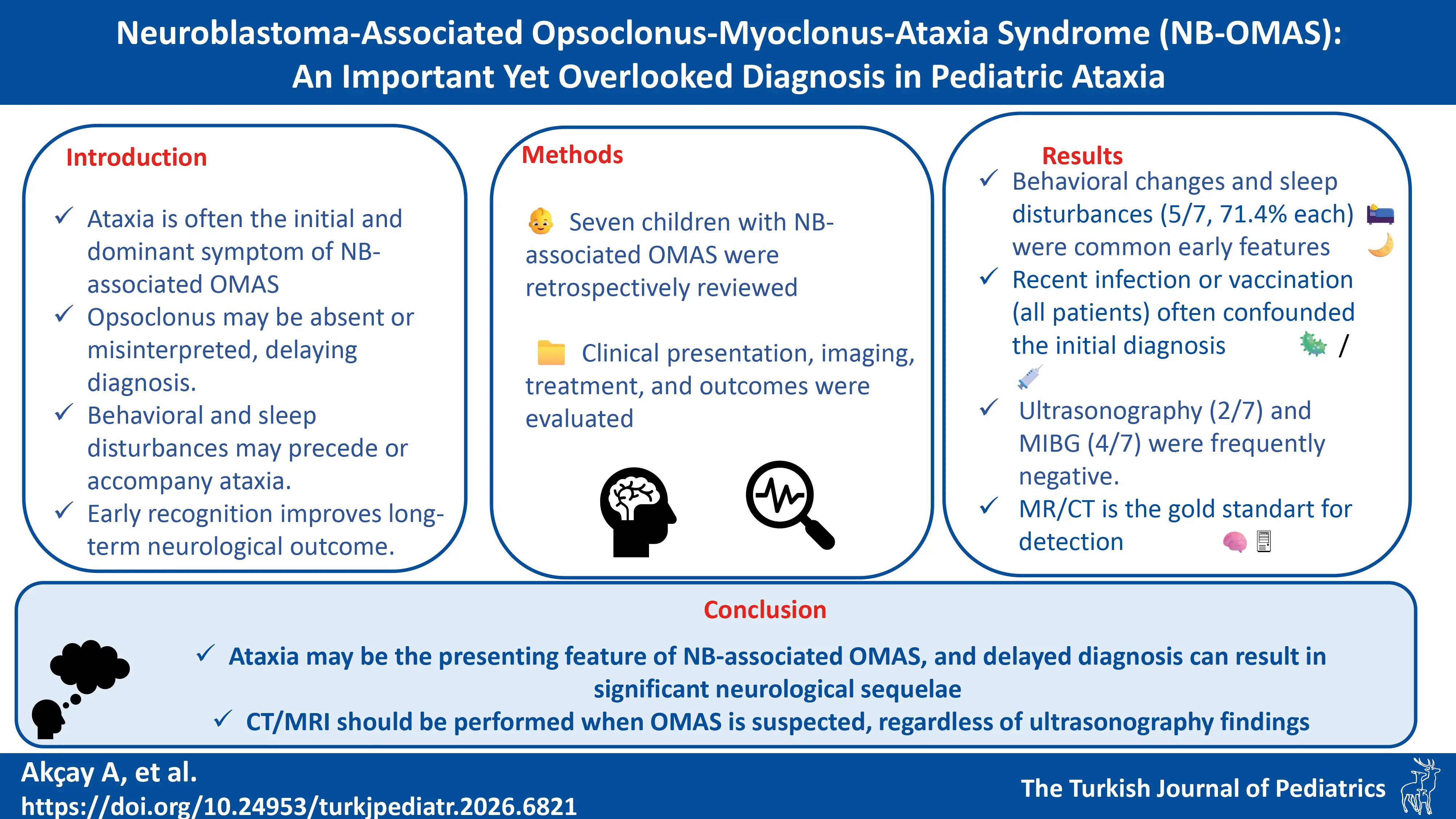
Background. Opsoclonus-myoclonus-ataxia syndrome (OMAS) is a rare, immune-mediated neurological disorder, often associated with neuroblastoma (NB) in children. This study aimed to describe the clinical features, diagnostic challenges, treatment, and outcomes of pediatric patients with NB-associated OMAS.
Methods. We retrospectively reviewed medical records of seven children diagnosed with NB-associated OMAS between November 1, 2015 and January 31, 2025. Data on demographics, clinical presentation, tumor characteristics, treatment protocols, relapses, and outcomes were collected. The Mitchell-Pike OMS Rating Scale was used to assess severity.
Results. The cohort included four girls and three boys, aged 18–36 months (median 31). All had a history of infection and/or vaccination preceding symptom onset. Ataxia was the most common initial symptom (6/7, 85.7%), followed by behavioral disturbances and sleep disorders (5/7, 71.4% each). At presentation, opsoclonus was present in two patients, which was initially misdiagnosed as nystagmus. All but one patient had opsoclonus during the disease course. Neuroblastomas were located in abdominal (n=4), thoracic (n=1), and sacral (n=2) regions; all tumors measured <50 mm and were stage L1. First-line treatment included tumor resection, intravenous immunoglobulin, and corticosteroids. Three patients required second-line therapy (rituximab, cyclophosphamide, mycophenolate mofetil, or azathioprine). After a median follow-up of 40 months (range 26-64), four patients had no neurological sequelae; three had mild cognitive impairment.
Conclusion. NB-associated OMAS may present with non-specific symptoms, leading to diagnostic delays. Early recognition, thorough tumor screening, and prompt immunotherapy may improve neurological outcomes. Clinicians should suspect NB in children presenting with ataxia and behavioral/sleep disturbances, even in the absence of opsoclonus or abnormal findings on initial imaging.
Keywords: Opsoclonus-myoclonus syndrome, ataxia, neuroblastoma, behavioral symptoms, sleep disorders, immunotherapy
Introduction
Opsoclonus-myoclonus-ataxia syndrome (OMAS), also called “Kinsbourne syndrome” or “dancing eye syndrome” is a rare, serious and often chronic neurological disorder. It consists of three main symptoms, including opsoclonus (conjugate, multidirectional, chaotic eye movements), myoclonus (nonepileptic limb jerking that may also involve the head and face), and truncal ataxia, causing gait imbalance. Sleep disturbances, cognitive dysfunction, and behavioral changes are often present.1-3 Children with OMAS typically present with an acute or subacute onset of ataxia between 6 and 36 months of age, often accompanied by an inability to walk or sit independently.4,5
Due to its rarity, data on its epidemiology, clinical features, and outcomes are scarce.6 Three out of the following four criteria should be met for the definitive diagnosis: (1) opsoclonus, (2) ataxia, (3) myoclonus, and (4) behavioral changes and/or sleep disturbances.7 Up to one third of cases have an atypical presentation, causing delays in diagnosis by weeks or months.8
In the pediatric population, OMAS may be associated with neuroblastoma (NB), ganglioneuroblastoma, or ganglioneuroma. An underlying NB may be present in about half of OMAS cases. However, the development of OMAS as a paraneoplastic syndrome is very rare (2-3%) in children with NB. In a study performed in North America, at least 43% of OMAS cases were found to have NB.8,9 The pathophysiology of NB-associated OMAS is thought to involve an immune-mediated encephalopathy triggered by cross-reactive autoimmune responses between tumor-associated antigens and central nervous system structures. Several antibodies have been described, including immunoglobulin (Ig) G and IgM targeting neural tissues and components such as Purkinje cells. In childhood OMAS, the presence of autoantibodies that react with neuronal surface antigens supports the hypothesis of an antibody-mediated pathogenesis. The observed neurological symptoms suggest that the immune response primarily targets the cerebellum and brainstem. However, the specificity of the identified antibodies remains limited, and the precise mechanisms and epidemiology of the disease have yet to be fully elucidated. Therefore, NB should be sought in every patient with OMAS. Occasionally, OMAS has been described in association with other entities such as ovarian teratoma or hepatoblastoma.10-12
In this paper, we aimed to evaluate the clinical characteristics, treatments, and outcomes of seven children who presented to the pediatric neurology clinic and were diagnosed with NB-associated OMAS.
Patients and Methods
Medical records of children who had been treated with a diagnosis of NB-associated OMAS between November 1, 2015 and January 31, 2025 were retrieved. Data included sociodemographic characteristics, clinical symptoms, patient histories, age at onset of symptoms, findings of the clinical examination, laboratory and radiological findings, time from onset of symptoms to diagnosis, treatment protocols, number and time of relapses, and treatment outcomes. The severity of ataxia, opsoclonus, myoclonus, and sleep or mood disturbances was also evaluated at the time of diagnosis and during treatment. The Mitchell-Pike OMS Rating Scale was utilized to assess clinical status, incorporating six categories (stance, gait, arm/hand function, opsoclonus, mood/behavior, and speech), with higher scores indicating more severe clinical presentations (Table I). Patients with OMAS not associated with NB were excluded.
| OMS: Opsoclonus-myoclonus syndrome. | ||
| Table I. Mitchell and Pike OMS Rating Scale | ||
| Stance | 0 | Standing and sitting balance normal for age |
| 1 | Mildly unstable standing for age, slightly wide based | |
| 2 | Unable to stand without support but can sit without support | |
| 3 | Unable to sit without using hands to prop or other support | |
| Gait | 0 | Walking normal for age |
| 1 | Mildly wide-based gait for age, but able to walk indoors and outdoors independently | |
| 2 | Walks only or predominantly with support from person or equipment | |
| 3 | Unable to walk even with support from person or equipment | |
| Arm/hand function | 0 | Normal for age |
| 1 | Mild, infrequent tremor or jerkiness without functional impairment | |
| 2 | Fine motor function persistently impaired for age, | |
| Opsoclonus | 0 | None |
| 1 | Rare or only when elicited by change in fixation or | |
| 2 | Frequent, interferes intermittently with fixation or tracking | |
| 3 | Persistent, interfering continuously with function and tracking | |
| Mood/behavior | 0 | Normal |
| 1 | Mild increase in irritability but consolable and/or mild sleep disturbances | |
| 2 | Irritability and sleep disturbances interfering with | |
| 3 | Persistent severe distress | |
| Speech | 0 | Normal for age, no loss |
| 1 | Mildly unclear, plateaued in development | |
| 2 | Loss of some words or some grammatical constructs (i.e., from sentences to phrases) but still communicates verbally | |
| 3 | Severe loss of verbal communication and speech. | |
The diagnosis of NB-associated OMAS was based on the presence of an NB or ganglioneuroblastoma accompanied by opsoclonus/ocular flutter and/or myoclonus and/or ataxia and/or behavioral changes/sleep disturbance, with or without irritability. Motor impairment was assessed depending on the degree of involvement, i.e., mild (the presence of abnormal neurological signs despite motor skills appropriate for age), moderate (mild or moderate decline in motor skills), and severe (severe decline in motor skills).
The patients were screened for a primary NB tumor by one or more of the following imaging or laboratory modalities: abdominal sonography, neck, chest, and abdominopelvic computed tomography (CT), magnetic resonance imaging (MRI), I-metaiodo-benzyl-guanidine (MIBG) scintigraphy, serum neuron specific enolase (NSE), urinary catecholamine metabolites, and MYCN-amplification in the tumor sample.
Initial medical treatment included intravenous immunoglobulin (IVIG) at 1-2 g/kg over 2–5 days and intravenous methylprednisolone (MP) at 20–30 mg/kg/day (max 1 g/day) for 3–5 days. Treatment outcome was classified by clinical assessment as follows: 1) complete recovery (as defined by the restoration of normal developmental stages), 2) partial recovery with mild sequelae (persistence of main symptoms of OMAS or cognitive changes that do not affect daily activities or mobility), or 3) moderate or severe sequelae (persistence of main symptoms of OMAS or cognitive changes that require specific intervention).
The study was approved by the local ethics committee and conducted in accordance with the Declaration of Helsinki.
Data were processed using the Statistical Package for Social Sciences (SPSS) version 18. Quantitative data were expressed as medians with ranges and qualitative data as frequencies and percentages.
Results
The study included seven pediatric patients (4 girls, 3 boys) who were diagnosed with and treated for NB-associated OMAS. The median age of the patients was 31 months (range 18–36). The time from symptom onset to the diagnosis of NB ranged from 3 weeks to 21 months (median 2 months). The median follow-up period was 40 months (range 26-64). Table II shows the clinical characteristics, treatments, and outcomes of the patients.
| CTX: Cyclophosphamide; GNB: Ganglioneuroblastoma; IVIG: Intravenous immunoglobulin; MMF: Mycophenolate mofetil; NA: Not available; NSE: Neuron specific enolase; OMS: Opsoclonus-myoclonus syndrome. | ||||||||||||||
| Table II. Clinical features of 7 children with opsoclonus-myoclonus-ataxia syndrome and neuroblastoma. | ||||||||||||||
| Patient no/Sex | Age (months) | Age at onset (months) | Previous history | Symptoms at presentation | Neurological symptoms | Mitchell and Pike OMS Rating Scale score |
Time from symptom onset to diagnosis |
Tumoral pathology | Location and size of tumor | NSE level | MYCN gene amplification | Treatment | Relapse | Recovery |
| 1/Female | 18 | 16 | Infection | Ataxia, tremor | Ataxia, tremor, behavioral disturbances, ocular flutter, sleep disturbance, irritability | 9 | 2 months | GNB |
Left adnexal, posterior to the ovary 18x17x26 mm |
Normal | Negative | Steroid + IVIG | No | Complete |
| 2/Male | 36 | 34 | Infection | Ataxia, hand tremor | Ataxia, loss of head control, opsoclonus, tremor, behavioral disturbances, sleep disturbance | 11 | 1.5 months | Ganglioneuroma |
Right adrenal 12x10x10 mm |
Mildly elevated | NA | Steroid + IVIG | No | Partial |
| 3/Female | 33 | 30 | Infection | Ataxia, opsoclonus | Ataxia, opsoclonus, behavioral disturbances, myoclonus, sleep disturbance | 8 | 3 months | GNB |
Right adrenal 35x20x24 mm |
Mildly elevated | Negative | None | Yes | Partial |
| 4/Male | 26 months | 25 | Infection and vaccination | Ataxia | Paraparesis, ataxia, myoclonus, behavioral disturbances | 11 | 1 month | Neuroblastoma |
Left posterior paravertebral area (T9-10) 32x18x40 mm |
Mildly elevated | Negative | Steroid + IVIG + CTX + MMF | No | Complete |
| 5/Female | 34 | 31 | Vaccination | Ataxia, tremor | Ataxia, tremor, opsoclonus | 10 | 3 months | GNB |
Sacral region 40x25x20 mm |
Normal | NA | Steroid + IVIG | No | Complete |
| 6/Male | 29 | 8 | Vaccination | Ocular flutter, tremor | Ataxia, opsoclonus, behavioral disturbances, sleep disturbance | 10 | 21 months | GNB |
Right posterior paravertebral area (L2-3) 31x14x7 mm |
Mildly elevated | NA | Steroid + IVIG + Azathioprine | Yes | Partial |
| 7/Female | 31 months | 30 months | Infection | Ataxia | Ataxia, opsoclonus, sleep disturbance, | 9 | 3 weeks | Ganglioneuroma |
Sacral region 20x31 mm |
Normal | Negative | Steroid + IVIG + Rituximab | Yes | Complete |
In all patients, a history of infection and/or vaccination was reported within 1–4 weeks prior to the onset of OMAS-related symptoms. Four patients (57%) had a history of infection, two patients (28.5%) had received vaccinations, and one patient (14%) had a history of both infection and vaccination.
Patients’ OMAS scores ranged between 8 and 11 points at the time of admission. By history, ataxia was the most common initial symptom (6/7, 85.7%), and all patients exhibited ataxia at the time of admission. Three patients (43%) experienced severe ataxia that impaired their ability to stand. Behavioral disturbances (5/7, 71.4%) and sleep disorders (5/7, 71.4%) were the second most common initial symptoms. In patient 1, sleep disturbance preceded the onset of ataxia by two months. Of the five patients with behavioral symptoms, four (Patients 1, 2, 3, and 6) presented with significant behavioral abnormalities, including aggression and inconsolability. All patients but one (Patient 4) developed abnormal eye movements (opsoclonus/ocular flutter) during the clinical course; however, two patients (Patients 3 and 6) who initially presented with abnormal eye movements were misinterpreted as having horizontal nystagmus. Myoclonus developed later in two patients (28.5%).
Five patients (71%) received IVIG treatment before the diagnosis of NB-associated OMAS because they had initially been diagnosed with post-infectious cerebellar ataxia (Patients 1, 2, 3, 5, and 7). Patient 3 had received two diagnoses, first Guillain-Barre syndrome followed by post-infectious cerebellar ataxia.
In two patients (Patients 1 and 2), abdominal ultrasonography failed to detect intra-abdominal NB. These patients received IVIG with the diagnosis of post-infectious cerebellar ataxia, but symptoms recurred one month after IVIG, at which time MRI was performed, helping detect the tumors.
One patient (Patient 4) initially presented with ataxic gait, progressive limb weakness, inability to walk or sit, and loss of head control. Examination revealed paraparetic signs in the lower extremities. Spinal MRI showed spinal root compression at the T9–10 level. Another patient (Patient 6) presented at 8 months of age with abnormal eye movements interpreted as nystagmus. Cranial imaging at another center was normal, and IVIG and steroid therapy were administered. At 12 months of age, the patient developed ataxia and tremor, and spinal MRI performed at 29 months confirmed NB.
Routine biochemical tests were within normal limits in all patients. Serum NSE levels were measured in all patients, being normal in four and mildly elevated in three patients. None of the patients had elevated urinary catecholamine levels.
All brain MRIs were unremarkable. The localizations of NB were in the thoracic region (Patient 4), abdominal region (Patients 1, 2, 3, and 6), and sacral region (Patients 5 and 7). Among abdominal tumors, one was located behind the ovary, two in the adrenal region, and one at the lumbar vertebral level. The tumor size was less than 50 mm in all patients.
All patients had stage L1 tumors according to the International Neuroblastoma Risk Group Staging System (INRGSS). All were also in the very low-risk group according to the Turkish Pediatric Oncology Group-Neuroblastoma-2020 protocol. MIBG scans performed in four patients were negative.
All patients underwent surgical excision, with complete removal of tumors except for one patient (Patient 6). In one patient (Patient 4), the diagnosis of NB was established via a tru-cut biopsy, and dexamethasone was initiated due to spinal root compression before total resection. Pathological evaluation after tumor excision revealed ganglioneuroblastoma in four patients (Patients 1, 3, 5, and 6), ganglioneuroma in two (Patients 2 and 7), and NB in one (Patient 4). N-Myc amplification performed in four patients was negative.
Following surgery, six patients (86%) were followed with monthly IVIG and steroid therapy for at least one year. Complete resolution was achieved with IVIG and steroids in patients 1, 2, and 5. One patient (Patient 3) who discontinued treatment after surgery developed recurrence of symptoms within the first year despite initial remission. Three patients (43%) received second-line therapy following inadequate response to first-line treatment, due to relapses in two (Patient 6 and 7) and due to partial improvement in one (Patient 4). Patient 7 experienced a relapse following an infection during the sixth month of IVIG and steroid treatment and was successfully treated with rituximab. Patient 6 received azathioprine and Patient 4 received cyclophosphamide and mycophenolate mofetil, both becoming symptom-free.
After a minimum follow-up of two years, four patients (57%) had complete recovery and three (43%) had partial recovery (Table II). No motor deficits were observed.
Discussion
NB-associated OMAS is a rare condition with many questions still unanswered. This study reports data on the presenting symptoms, diagnostic steps, clinical follow-up, and treatment of seven patients diagnosed with NB-associated OMAS. The female-to-male ratio was 4:3. Some studies reported a female predominance,13-15 while others reported no sex difference.16,17 OMAS has a lower incidence in infancy, with some studies reporting it as a rare diagnosis before age 1, which is thought to be related to the limited capacity of younger infants to produce specific antibodies to the nervous system in the first year of life.18 The age range of the patients in our study was 18-36 months.
The symptoms of OMAS have been shown to coincide with the period during which routine pediatric vaccination schedules are carried out. Singhi et al. reported that all patients with NB-associated OMAS had a history of fever or vaccination 1–10 days previous to disease onset.19 In an epidemiological study of OMAS patients, 85% of patients reportedly received vaccinations before the onset of neurological symptoms and NB-associated OMAS accounted for 41% of the patient population.20 In a study conducted in Türkiye, prodromal infection was detected in 26% of all OMAS cases with or without NB.16 All of our patients had a history of infection/vaccination before symptom onset, suggesting the role of infections or vaccines as triggering factors for OMAS.
Opsoclonus, the best-known symptom that gives its name to the syndrome, may appear as a late finding or may be absent in some atypical cases. Its intensity and frequency can vary within the same individual. In older individuals, it may also manifest as ocular flutter. It can be confused with nystagmus or epileptic seizures.5,7,13,19,21 Myoclonus occurs less frequently. In a study by Galstyan et al., all patients developed ataxia and opsoclonus, with only one patient developing myoclonus.13,17 In another study, opsoclonus, myoclonus, and ataxia were observed concomitantly in 67% of patients with OMAS at presentation, while a combination of two of these symptoms was observed in 33% of patients.18 In the present series, two patients had myoclonus as a late symptom. One patient had no opsoclonus. Abnormal eye movements of two patients were misinterpreted as nystagmus at admission.
The occurrence of ataxia prior to opsoclonus or myoclonus in OMAS may lead to a misdiagnosis of acute cerebellar ataxia.13 Four of our patients were misdiagnosed as having postinfectious cerebellar ataxia, while one was misdiagnosed as Guillain-Barré syndrome and postinfectious cerebellar ataxia. Therefore, a diagnostic work-up for OMAS to differentiate it from acute cerebellar ataxia is critical to tumor diagnosis.13 Diagnostic work-up should also include assessment of behavioral changes and sleep disturbances. Irritability and sleep disorders are reported in 60-83% of patients,16,17,21 while some studies found behavioral changes in all patients.3,19,22 Since acute cerebellar ataxia is almost universally the initial misdiagnosis in OMAS patients, a high index of suspicion is required in the presence of ataxia and irritability in toddlers, even if opsoclonus or myoclonus has not yet developed.13
Sleep disturbance or behavioral symptoms may be observed a few weeks before the onset of typical OMAS symptoms.10 In one of our patients, sleep disturbance started two months before the onset of ataxia. Behavioral changes and sleep disturbances should be carefully questioned in pediatric patients presenting with ataxia, and an underlying tumor (namely NB) should be sought. Even if a tumor is not detected by ultrasonography, further imaging methods (CT/MRI) should be performed.
NB can occur anywhere in the sympathetic nervous system23, making detection difficult and necessitating a comprehensive investigation. The first-line tests can be misleading, with high false-negative rates. Since dancing eye syndrome/OMAS-related tumors are usually low-grade, MIBG screening and tests dependent on metabolic activity such as urinary catecholamine metabolites may not be sensitive enough to detect the tumor.21,22 In a study by Brunklaus et al., only 24% of NB-associated OMAS cases were positive for urinary catecholamines.8 None of the patients in our study had elevated urinary catecholamine levels, while four patients showed a mild increase in NSE. Regarding tumor localization, retroperitoneal region (65%), adrenal glands (40%), mediastinum (15%), cervical region (11%), and pelvic region (3%) were most commonly reported.24 In a study of 462 NB cases from Türkiye, the abdomen, thorax, pelvis, and neck were found at frequencies of 72.2%, 14.9%, 3.8%, and 3.2%, respectively.25 In two studies on NB-associated OMAS, the abdominal region had significant predominance at 43% and 93%, each for the primary tumor localization.9,29 One study reported a markedly higher rate for mediastinal tumor localization (49%) in patients with NB-associated OMAS, compared with NB patients without OMAS.5 In our patients, mediastinal localization was seen in one patient (14.2%). Three patients had pelvic NB, one of which was located behind the left ovary and could not be visualized in two ultrasound examinations.
The diagnostic approach for pediatric patients with acute ataxia includes a thoracic X-ray, ultrasonography of the neck, abdomen, and pelvis, and urine tests for adrenal metabolites. If the results are normal, a detailed neck, chest, and abdominal MRI scan is performed.7 Although primary tests such as thoracic X-ray and abdominal ultrasound may be useful,8 thoracic/abdominal CT and MRI are the most sensitive imaging modalities to detect occult NB in OMAS.26 In a study of OMAS patients conducted in the UK, MIBG could identify NB in none of four patients.22 In another study, sensitivity rates of 86% and 99%, and specificity rates of 85% and 95% were found for full-body MRI and combined MRI and scintigraphy, respectively.27 Therefore, MIBG should not replace MRI/CT in the diagnosis of NB-associated OMAS, but can be used to monitor disease recurrences.
Manifestations of symptoms depend on the tumor localization. In cases of spinal cord compression, symptoms are seen in 5-7% of patients.24 One of our patients developed paraparesis due to spinal cord compression. Brain imaging is usually normal,26 but abnormalities such as cerebellar or cerebral cortex atrophy may also be seen.16,26,28 No cerebellar or cerebral cortex abnormalities were seen in our patients.
In recent years, NB has been increasingly diagnosed in patients with OMAS thanks to advances in imaging modalities,6 though time to diagnosis of OMAS still remains highly variable.13 In previous studies, the mean time from the onset of OMAS symptoms to diagnosis ranged from 2 days to 14 months.16,20,29,30 The duration of symptoms before the detection of a tumor also varies from 6 weeks to as long as 17 months.31 Early detection of the tumor may lead to early treatment of symptoms and improved quality of life.13
Compared to rates of 30–34% in the general NB population, the two-year survival rate is significantly higher (89.3%) in NB patients with OMAS, attributed to the autoimmune pathophysiology of OMAS and its limiting effect on tumor growth and spread.5 Furthermore, NBs in OMAS patients are characterized by their smaller size possibly due to early diagnosis, with tumor diameters being <50 mm in 62–80% of cases.26,32 In all our patients, tumor diameters were <50 mm.
Long-term neurological sequelae are seen in 70–80% of pediatric patients with OMAS.18 Neurological sequelae developed in three of our cases (43%): one was diagnosed with a 29-month developmental delay, and another did not receive appropriate treatment despite relapse. A 5-year follow-up study from Turkey reported neuropsychiatric sequelae in 40% of patients.33 Recent data suggest that use of multiple agents leading to increased immunosuppression improves developmental outcomes of OMAS.13 On the other hand, reports have been discrepant regarding the relationship between delayed treatment of NB-associated OMAS and the development of neurological sequelae. In a case series from Japan in which the mean time to tumor diagnosis was 7 months after the onset of OMAS, neurological sequelae were more common in patients with a longer interval between tumor diagnosis and initial treatment.34 In a long-term prognosis study, it was observed that the time between the onset of OMAS and the diagnosis of NB-associated OMAS was longer in the group with cognitive impairment.9 Similar results were found in a study from Italy.29 However, there have been studies that did not find this association.21,35
Bearing in mind the rarity of the condition, reported cases in the literature may provide new insights into pediatric cases with ataxia and OMAS that could be caused by an unrecognized tumor (i.e., NB).
The limitations of this study include its retrospective design, small sample size, differences in treatment protocols, and relatively short follow-up periods. Despite these limitations, with detailed clinical description, it addresses a significant diagnostic challenge in identifying NB-associated OMAS in pediatric patients presenting with acute ataxia.
Conclusion
NB-associated OMAS is a rare condition usually co-presenting with ataxia in the pediatric population. Delayed diagnosis is common in patients presenting with ataxia, which can lead to significant neurological sequelae. A detailed investigation for the underlying NB should be made in patients with ataxia, before establishing the diagnosis of acute cerebellar ataxia. Sleep dysregulation and behavioral changes usually suggest OMAS, and if present, the patient should undergo CT/MRI for NB, even if no tumor is detected on ultrasonography.
Ethical approval
The study was approved by Ethics Committee of the Koc University Faculty of Medicine (date: 25.07.2025, number: 2025.341.IRB2.163).
Source of funding
The authors declare the study received no funding.
Conflict of interest
The authors declare that there is no conflict of interest.
References
- Dale RC. Childhood opsoclonus myoclonus. Lancet Neurol 2003; 2: 270. https://doi.org/10.1016/s1474-4422(03)00374-0
- Singer HS, Mink JW, Gilbert DL, Jankovic J. Movement disorders in autoimmune diseases. In: Fernandez HB, Obeso JA, Ondo WG, editors. Movement disorders in childhood. Amsterdam: Elsevier; 2016: 409-426. https://doi.org/10.1016/B978-0-12-411573-6.00018-8
- Meena JP, Seth R, Chakrabarty B, Gulati S, Agrawala S, Naranje P. Neuroblastoma presenting as opsoclonus-myoclonus: a series of six cases and review of literature. J Pediatr Neurosci 2016; 11: 373-377. https://doi.org/10.4103/1817-1745.199462
- Mitchell WG, Davalos-Gonzalez Y, Brumm VL, et al. Opsoclonus-ataxia caused by childhood neuroblastoma: developmental and neurologic sequelae. Pediatrics 2002; 109: 86-98.
- Xie S, Bai C, Li K, Dong K, Yao W. Comparison of mediastinal and non-mediastinal neuroblastoma and ganglioneuroblastoma associated with opsoclonus-myoclonus syndrome: a systematic review and meta-analysis. Transl Cancer Res 2022; 11: 3741-3753. https://doi.org/10.21037/tcr-22-1120
- Caffarelli M, Kimia AA, Torres AR. Acute ataxia in children: a review of the differential diagnosis and evaluation in the emergency department. Pediatr Neurol 2016; 65: 14-30. https://doi.org/10.1016/j.pediatrneurol.2016.08.025
- Poretti A, Benson JE, Huisman TAGM, Boltshauser E. Acute ataxia in children: approach to clinical presentation and role of additional investigations. Neuropediatrics 2013; 44: 127-141. https://doi.org/10.1055/s-0032-1329909
- Brunklaus A, Pohl K, Zuberi SM, de Sousa C. Investigating neuroblastoma in childhood opsoclonus-myoclonus syndrome. Arch Dis Child 2012; 97: 461-463. https://doi.org/10.1136/adc.2010.204792
- Sun Q, Wang Y, Xie Y, Wu P, Li S, Zhao W. Long-term neurological outcomes of children with neuroblastoma with opsoclonus-myoclonus syndrome. Transl Pediatr 2022; 11: 368-374. https://doi.org/10.21037/tp-21-519
- Hero B, Schleiermacher G. Update on pediatric opsoclonus myoclonus syndrome. Neuropediatrics 2013; 44: 324-329. https://doi.org/10.1055/s-0033-1358604
- Storz C, Bares R, Ebinger M, Handgretinger R, Tsiflikas I, Schäfer JF. Diagnostic value of whole-body MRI in Opsoclonus-myoclonus syndrome: a clinical case series (3 case reports). BMC Med Imaging 2019; 19: 70. https://doi.org/10.1186/s12880-019-0372-y
- Blaes F, Fühlhuber V, Korfei M, et al. Surface-binding autoantibodies to cerebellar neurons in opsoclonus syndrome. Ann Neurol 2005; 58: 313-317. https://doi.org/10.1002/ana.20539
- Pranzatelli MR, Tate ED, McGee NR. Demographic, clinical, and immunologic features of 389 children with opsoclonus-myoclonus syndrome: a cross-sectional study. Front Neurol 2017; 8: 468. https://doi.org/10.3389/fneur.2017.00468
- Zhu H, Wu W, Chen L, et al. Clinical analysis of pediatric opsoclonus-myoclonus syndrome in one of the national children’s medical center in China. Front Neurol 2021; 12: 744041. https://doi.org/10.3389/fneur.2021.744041
- Hasegawa S, Matsushige T, Kajimoto M, et al. A nationwide survey of opsoclonus-myoclonus syndrome in Japanese children. Brain Dev 2015; 37: 656-660. https://doi.org/10.1016/j.braindev.2014.10.010
- Yıldırım M, Öncel İ, Bektaş Ö, et al. Clinical features and outcomes of opsoclonus myoclonus ataxia syndrome. Eur J Paediatr Neurol 2022; 41: 19-26. https://doi.org/10.1016/j.ejpn.2022.09.002
- Galstyan A, Wilbur C, Selby K, Hukin J. Opsoclonus-myoclonus syndrome: a new era of improved prognosis? Pediatr Neurol 2017; 72: 65-69. https://doi.org/10.1016/j.pediatrneurol.2017.03.011
- Elzomor H, El Menawi S, Elawady H, et al. Neuroblastoma-associated opsoclonous myoclonous ataxia syndrome: profile and outcome report on 15 Egyptian patients. J Pediatr Hematol Oncol 2023; 45: e194-e199. https://doi.org/10.1097/MPH.0000000000002466
- Singhi P, Sahu JK, Sarkar J, Bansal D. Clinical profile and outcome of children with opsoclonus-myoclonus syndrome. J Child Neurol 2014; 29: 58-61. https://doi.org/10.1177/0883073812471433
- Tate ED, Allison TJ, Pranzatelli MR, Verhulst SJ. Neuroepidemiologic trends in 105 US cases of pediatric opsoclonus-myoclonus syndrome. J Pediatr Oncol Nurs 2005; 22: 8-19. https://doi.org/10.1177/1043454204272560
- Krug P, Schleiermacher G, Michon J, et al. Opsoclonus-myoclonus in children associated or not with neuroblastoma. Eur J Paediatr Neurol 2010; 14: 400-409. https://doi.org/10.1016/j.ejpn.2009.12.005
- Pang KK, de Sousa C, Lang B, Pike MG. A prospective study of the presentation and management of dancing eye syndrome/opsoclonus-myoclonus syndrome in the United Kingdom. Eur J Paediatr Neurol 2010; 14: 156-161. https://doi.org/10.1016/j.ejpn.2009.03.002
- Maris JM. Recent advances in neuroblastoma. N Engl J Med 2010; 362: 2202-2211. https://doi.org/10.1056/NEJMra0804577
- Ozerov SS, Samarin AE, Andreev ES, et al. Neurosurgical aspects of the treatment of neuroblastoma patients. Zh Vopr Neirokhir Im N N Burdenko 2016; 80: 50-57. https://doi.org/10.17116/neiro201680350-57
- Aydn GB, Kutluk MT, Yalçn B, et al. Neuroblastoma in Turkish children: experience of a single center. J Pediatr Hematol Oncol 2009; 31: 471-480. https://doi.org/10.1097/MPH.0b013e3181a6dea4
- Urtiaga Valle S, Souvannanorath S, Leboucq N, et al. Monocentric retrospective clinical outcome in a group of 13 patients with opsoclonus myoclonus syndrome, proposal of diagnostic algorithm and review of the literature. Eur J Paediatr Neurol 2022; 40: 18-27. https://doi.org/10.1016/j.ejpn.2022.07.002
- Pfluger T, Schmied C, Porn U, et al. Integrated imaging using MRI and 123I metaiodobenzylguanidine scintigraphy to improve sensitivity and specificity in the diagnosis of pediatric neuroblastoma. AJR Am J Roentgenol 2003; 181: 1115-1124. https://doi.org/10.2214/ajr.181.4.1811115
- Anand G, Bridge H, Rackstraw P, et al. Cerebellar and cortical abnormalities in paediatric opsoclonus-myoclonus syndrome. Dev Med Child Neurol 2015; 57: 265-272. https://doi.org/10.1111/dmcn.12594
- De Grandis E, Parodi S, Conte M, et al. Long-term follow-up of neuroblastoma-associated opsoclonus-myoclonus-ataxia syndrome. Neuropediatrics 2009; 40: 103-111. https://doi.org/10.1055/s-0029-1237723
- Mitchell WG, Wooten AA, O’Neil SH, Rodriguez JG, Cruz RE, Wittern R. Effect of increased immunosuppression on developmental outcome of Opsoclonus Myoclonus Syndrome (OMS). J Child Neurol 2015; 30: 976-982. https://doi.org/10.1177/0883073814549581
- Russo C, Cohn SL, Petruzzi MJ, de Alarcon PA. Long-term neurologic outcome in children with opsoclonus-myoclonus associated with neuroblastoma: a report from the Pediatric Oncology Group. Med Pediatr Oncol 1997; 28: 284-288. https://doi.org/10.1002/(sici)1096-911x(199704)28:4<284::aid-mpo7>3.0.co;2-e
- Plantaz D, Michon J, Valteau-Couanet D, et al. Opsoclonus-myoclonus syndrome associated with non-metastatic neuroblastoma. Long-term survival. Study of the French Society of Pediatric Oncologists. Arch Pediatr 2000; 7: 621-628. https://doi.org/10.1016/s0929-693x(00)80129-3
- Aktekin EH, Gezer HÖ, Yazıcı N, Erol İ, Erbay A, Sarıalioğlu F. Five years follow-up of opsoclonus-myoclonus-ataxia syndrome-associated neurogenic tumors in children. Neuropediatrics 2024; 55: 57-62. https://doi.org/10.1055/s-0043-1768143
- Takama Y, Yoneda A, Nakamura T, et al. Early detection and treatment of neuroblastic tumor with opsoclonus-myoclonus syndrome improve neurological outcome: a review of five cases at a single institution in Japan. Eur J Pediatr Surg 2016; 26: 54-59. https://doi.org/10.1055/s-0035-1564714
- Rudnick E, Khakoo Y, Antunes NL, et al. Opsoclonus-myoclonus-ataxia syndrome in neuroblastoma: clinical outcome and antineuronal antibodies-a report from the Children’s Cancer Group Study. Med Pediatr Oncol 2001; 36: 612-622. https://doi.org/10.1002/mpo.1138
License
Copyright (c) 2026 The Author(s). This is an open access article distributed under the Creative Commons Attribution License (CC BY), which permits unrestricted use, distribution, and reproduction in any medium or format, provided the original work is properly cited.















