Abstract
Chronic nonbacterial osteomyelitis (CNO) is a rare, autoinflammatory bone disorder that predominantly affects children and adolescents. The disease covers a broad clinical spectrum ranging from isolated bone lesions to its severe multifocal form, chronic recurrent multifocal osteomyelitis (CRMO). Although its exact pathogenesis remains elusive, recent advances highlight a pivotal role of innate immune dysregulation, particularly involving imbalanced cytokine signaling. These abnormalities drive sterile bone inflammation and osteoclast activation, leading to bone pain and lytic or sclerotic lesions. CNO remains a diagnosis of exclusion owing to the lack of specific biomarkers or standardized diagnostic criteria; however, the recently developed European Alliance of Associations for Rheumatology/American College of Rheumatology (EULAR/ACR) classification framework represents an important step toward uniformity in research and clinical trials. Whole-body magnetic resonance imaging has emerged as the imaging modality of choice for diagnosis and monitoring, while biopsy is reserved for atypical or unifocal cases. Management is empirical and guided by disease severity, with nonsteroidal anti-inflammatory drugs as first-line therapy, followed by corticosteroids, disease-modifying antirheumatic drugs, bisphosphonates, and biologic agents such as tumor necrosis factor (TNF) inhibitors in refractory cases. New insights into the interleukin (IL)-1, IL-6, and IL-17/23 pathways have opened avenues for targeted therapies, including Janus kinase (JAK) inhibitors, in difficult-to-treat patients. Despite earlier perceptions of a benign course, long-term follow-up indicates a risk of relapses and structural complications, underscoring the need for early recognition and multidisciplinary management. Ongoing international collaborations are expected to refine diagnostic precision and optimize treatment strategies to improve outcomes in pediatric CNO.
Keywords: autoinflammatory bone disease, child, chronic nonbacterial osteomyelitis, recurrent osteomyelitis, pediatric rheumatology
Introduction
Chronic non-bacterial osteomyelitis (CNO) or chronic recurrent multifocal osteomyelitis (CRMO) is a noninfectious auto-inflammatory bone disease mainly affecting children.1,2 It was first described by Gieldon et al. in 1972, as subacute and chronic ‘symmetrical’ osteomyelitis.3,4 While CNO predominantly occurs in children and adolescents, cases have been reported in all age groups. The disease expresses a wide clinical spectrum that extends from isolated asymptomatic bone lesions to its most severe multifocal presentation, CRMO, a designation introduced by Björkstén et al. in 1978.3,5 Although it has been classified in various ways over time, the term CNO is now widely used according to the most recent nomenclature.6-8
To date, CNO remains still a poorly defined and heterogeneous condition. It is regarded as a diagnosis of exclusion, given the absence of standardized accepted diagnostic criteria and validated biomarkers.9 Although the precise etiology remains unknown, accumulating evidence supports the central role of innate immune dysregulation and cytokine imbalance in disease pathogenesis.1 The complex and unpredictable behavior of CNO poses ongoing challenges in diagnosis, management, and long-term care for both patients and physicians. Delayed or missed diagnoses are common and are often related to limited awareness of the disease and the fluctuating pain patterns it produces. Many patients are evaluated by multiple clinicians before reaching a definitive diagnosis, and these delays frequently lead to unnecessary or prolonged antibiotic treatments, excessive radiation exposure due to repeated imaging studies, and even multiple bone biopsies prior to establishing the correct diagnosis.10-12
In this review, we aim to summarize the current knowledge on CNO, to provide a practical approach for diagnosis and differential diagnosis in children, and to highlight recent advances in disease management.
Epidemiology
Although the true epidemiological data may remain uncertain due to the insidious nature of the disease and limited awareness, the incidence has been estimated in several reports to range between 0.4 and 2.3 per million.7,13-15 A large cohort study from Germany comparing CNO and bacterial osteomyelitis (BOM) demonstrated that the incidence of CNO is likely underestimated and may be close to that of BOM.16
The average age at onset of pediatric CNO is between 7-12 years, with a modest predominance among females (female/male 1.5:1 to 4:1 in different series). While CNO appears to be reported more frequently in Western populations, especially in Europe and North America, cases have been documented in individuals of all racial and ethnic backgrounds.14,17 The female predominance observed in CNO contrasts with infectious osteomyelitis, which is more frequent in males.15,17-21 In contrast to the female predominance generally reported in the literature, a national Turkish cohort of 334 pediatric patients demonstrated a male predominance (56.3%) (in press; unpublished data). Consistently, three independent studies from Türkiye reported comparable male proportions (~53–56%).22-24
CNO can occur at any age, however disease onset before the age of two years should raise suspicion for a monogenic autoinflammatory condition such as Majeed syndrome or deficiency of the interleukin-1 receptor antagonist (DIRA).8
Pathophysiology and Genetics
The exact molecular pathogenic mechanism of CNO is poorly understood. However, recent advances in both genetic and molecular research have provided new insights into its underlying pathways. While a detailed discussion of the pathophysiology is beyond the scope of this review, a brief overview of the main mechanisms will be provided.
CNO is now recognized as an autoinflammatory bone disorders in which dysregulation of the innate immune system plays a central role. Studies have demonstrated dysregulated activation of the NLR family pyrin domain containing 3 (NLRP3) inflammasome, leading to an imbalance between pro- and anti-inflammatory cytokines; characterized by elevated levels of interleukin (IL)-1β, IL-6, and Tumor necrosis factor alpha (TNF-α), accompanied by a relative deficiency of IL-10.25,26 This imbalance promotes recruitment of monocytes and neutrophils to the bone marrow, resulting in sterile inflammation and increased osteoclast activity.8
Findings from both human transcriptomic analyses and experimental models with PSTPIP1 mutations suggest a persistent pro-osteoclastogenic environment that interferes with normal bone homeostasis and resolution of inflammation.27 Among the key mechanisms implicated, the receptor activator of nuclear factor-κB (RANK), its ligand (RANKL), and osteoprotegerin (OPG) axis has gained attention for its role in osteoclast differentiation and survival. Overexpression of RANKL or insufficient production of OPG enhances osteoclastogenesis and bone resorption, which may explain the characteristic osteolytic lesions observed in CNO.25 This was demonstrated in an adult CNO study, supporting the role of osteoclast dysregulation in disease pathogenesis.1,28
In parallel, recent genomic studies have identified P2RX7 variants that amplify inflammasome signaling and Filamin-binding LIM Protein 1 (FBLIM1) mutations that weaken IL-10-mediated regulatory feedback.1,29,30 Together, these alterations link genetic susceptibility with innate immune hyperreactivity, creating recurrent cycles of inflammation, bone resorption, and repair that mimic bacterial osteomyelitis but occur in the absence of infection.8
Monogenic forms of CNO
A small subset of patients, particularly those presenting in infancy or with systemic features, may have monogenic autoinflammatory bone diseases. The best-known examples are Majeed syndrome (LPIN2 variants) and DIRA (IL1RN variants) and pyogenic arthritis, pyoderma gangrenosum and acne syndrome, PAPA (PSTPIP1 variants).1,29 These disorders involve uncontrolled IL-1–mediated inflammation and typically present as severe, early-onset, and recurrent forms of osteomyelitis. Recognition of these entities is essential, as they may require targeted therapy aimed at IL-1 and TNF-α blockade rather than conventional anti-inflammatory regimens.29
Clinical Presentations
The cardinal feature of CNO is recurrent bone pain of insidious onset. CNO usually begins with a slow and subtle onset of recurring bone pain, tenderness, or localized swelling that may involve one or several sites of sterile inflammation. The clinical picture is highly variable; some patients experience isolated, short-lived disease, while others develop multiple or relapsing foci that can persist for years.15 The pain often intensifies during nighttime and may cause limping or reduced mobility when weight-bearing bones are affected and mostly improves with nonsteroidal anti-inflammatory drug (NSAID) therapy.31,32 The absence of infection or malignancy, combined with the waxing and waning pattern of symptoms, frequently contributes to diagnostic delay. Tenderness, swelling, and increased warmth may be observed over the affected bone; on the other hand, these findings may be absent depending on the thickness of the surrounding soft tissue.18 Delays in diagnosis are common, as early, non-specific symptoms are frequently mistaken for benign conditions such as growing pains. Without appropriate treatment, CNO typically follows a prolonged, fluctuating course characterized by alternating phases of remission and relapse.29,33
Systemic features such as fever, malaise, fatigue, and weight loss are observed in about 15–20% of patients, yet most children with CNO remain clinically well except for symptoms related to bone pain14,34 Inflammation can occur at any site of the skeleton; however, the metaphyseal regions of the long bones, clavicle, vertebral bodies, and pelvis are the most commonly involved sites.35 Long bones of the lower extremities are affected about three times more often than those of the upper limbs, with the distal femur and proximal tibia being the most common sites. Other characteristic localizations include the clavicle, vertebrae, pelvis, and mandible. Involvement of the medial third of the clavicle is considered highly typical for CNO.27,32 Lesions of the epiphysis or diaphysis may accompany metaphyseal lesions, but isolated epiphyseal or diaphyseal involvement without metaphyseal changes is uncommon. Spontaneous improvement can be observed in some cases.36 Nearly 85% of cases present with multifocal lesions, and the involvement is symmetrical in 25–40% of patients.37 Although vertebral lesions occur less frequently, they are clinically significant because of potential complications such as fracture, spinal cord compression, kyphosis, or scoliosis. CNO tends to involve the thoracic vertebrae, and the preservation of intervertebral discs helps distinguish it from other conditions associated with vertebral destruction.11,35,38 Pelvic lesions may occasionally present as unilateral sacroiliitis, while the small bones of the hands and feet are affected less commonly.32,39
Localized swelling of the soft tissue adjacent to the affected bone may sometimes mimic arthritis. However, in some series, arthritis has been reported in up to 40% of patients with CNO. It may occur adjacent to the bone lesion (60%) or at distant sites (40%), which can make it difficult to distinguish CNO from juvenile idiopathic arthritis (JIA).6,16,34
Extraosseous manifestations and associated inflammatory conditions
CNO can occur in association with other inflammatory conditions, and a positive family history of inflammatory or autoinflammatory diseases is common. Studies have shown that approximately 20% of patients have concomitant chronic inflammatory disorders such as JIA, spondyloarthropathies, inflammatory bowel disease (IBD), pyoderma gangrenosum, familial Mediterranean fever (FMF), and SAPHO syndrome (synovitis, acne, pustulosis, hyperostosis, osteitis).1,16 Reported frequencies include inflammatory arthritis (10–41%), palmoplantar pustulosis (7–14%), psoriasis (4–22%), inflammatory bowel disease (4–22%), acne (4–22%), and FMF (approximately 10%) (Fig. 1).1,16,40-42 The overlap between CNO and SAPHO syndrome has prompted some authors to suggest that CNO may represent a pediatric counterpart of SAPHO.30 However, the typical anatomical sites of involvement differ between the two conditions. Patients with CNO who have concomitant inflammatory disorders may exhibit a more severe clinical phenotype. Therefore, in cases with an aggressive or refractory disease course, clinicians should carefully consider and screen for concomitant inflammatory or autoinflammatory diseases.17,41,43
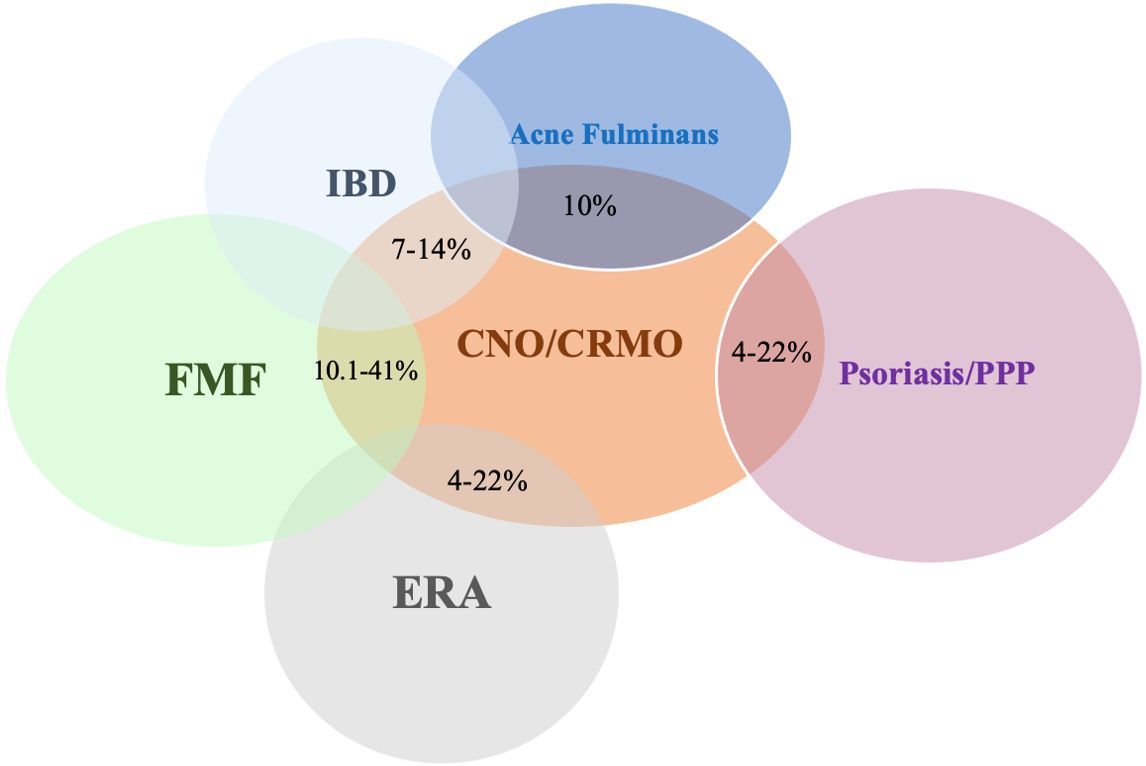
CNO/CRMO: Chronic nonbacterial osteomyelitis / Chronic recurrent multifocal osteomyelitis, ERA: Enthesitis-related arthritis, FMF: Familial Mediterranean fever, IBD: Inflammatory bowel disease, PPP: Palmoplantar pustulosis.
Laboratory and Histopathological Findings
Routine laboratory tests usually provide non-specific findings, yet they remain essential for excluding alternative or secondary causes.29 The initial diagnostic work-up in patients with suspected CNO typically includes basic laboratory tests — complete blood count (CBC), C-reactive protein (CRP), and erythrocyte sedimentation rate (ESR) — as well as imaging studies and, when necessary, bone biopsy. Although these investigations are rarely diagnostic, they are useful in ruling out infection, malignancy, or metabolic bone disease35 (Table I and Fig. 2).
|
*May be incomplete. AIDs: autoinflammatory diseases, CRP: C-reactive protein, ESR: erythrocyte sedimentation rate, HSM: hepatosplenomegaly, LAP: lymphadenopathy, LDH: Lactate dehydrogenase, NSAIDs: nonsteroidal anti-inflammatory drugs |
||
| Table I. Clues for differential diagnosis in chronic nonbacterial osteomyelitis* | ||
| Category | Red flag findings | Possible alternative diagnoses |
| Age onset | Onset <2 years | Monogenic AIDs |
| Symptoms and physical examination | High-grade fever, marked malaise, weight loss, night sweats, severe pain (can’t bear weight, unable to walk), HSM, LAP | Bacterial osteomyelitis, malignancy, other inflammatory disorders and infections |
| Laboratory | Very high CRP or ESR, cytopenia, markedly elevated LDH or uric acid, low alkaline phosphatase, low phosphate, vitamin C deficiency | Infection, leukemia, lymphoma, hypophosphatasia, scurvy |
| Imaging |
Solitary lesion, cortical destruction with periosteal reaction, soft tissue mass, or lack of multifocality, localization at skull and diaphysis periosteal elevation with subperiosteal hemorrhage |
Malignancy, bacterial osteomyelitis, scurvy |
| Histopathology | Presence of atypical cells, granulomatous inflammation, or necrosis | Malignancy, infection, histiocytosis |
| Response to therapy | Absence of response to NSAIDs/anti-inflammatory treatments | Infection, neoplasm, metabolic bone disease |
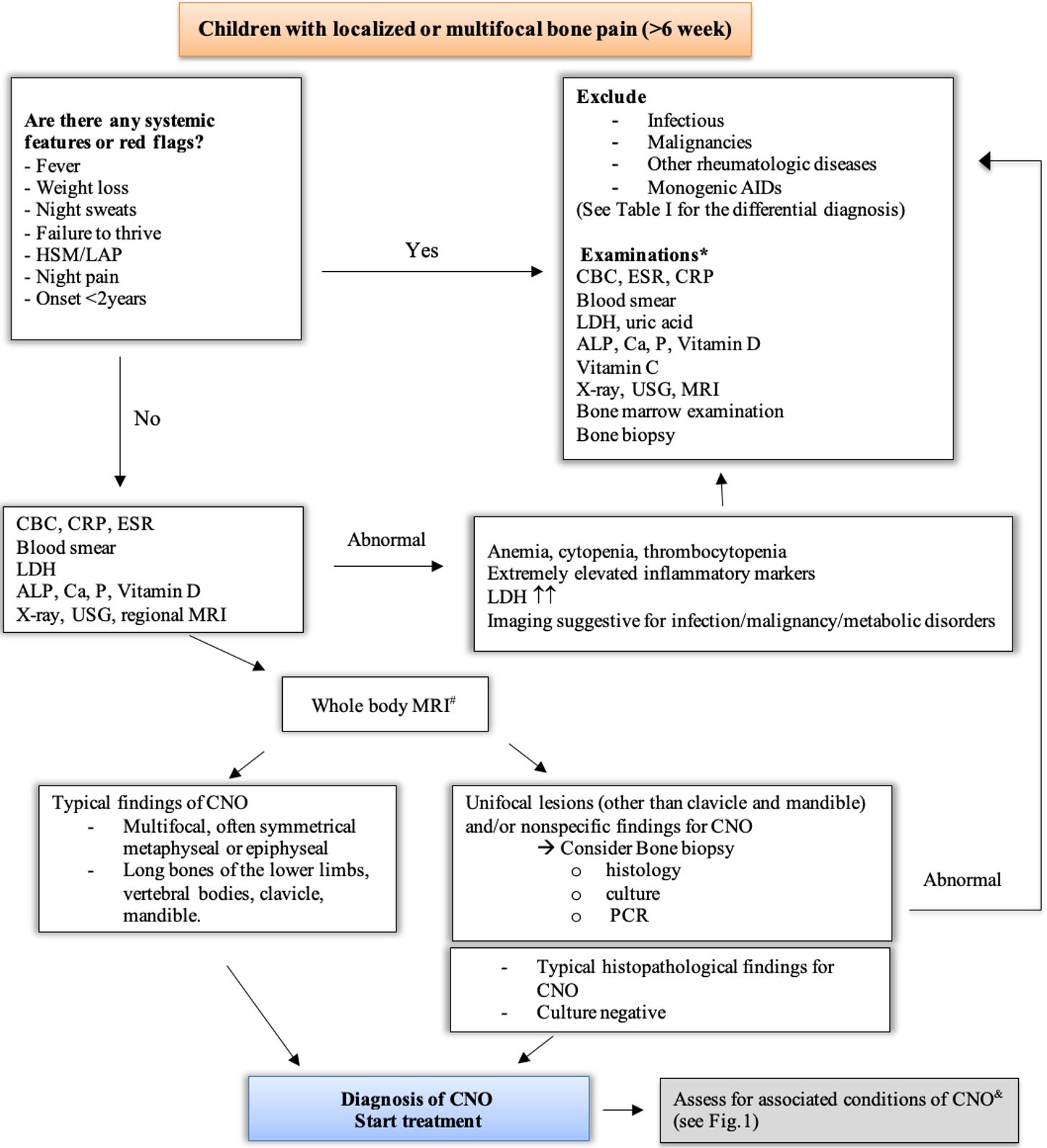
*Diagnostic tests should be performed and interpreted stepwise, based on clinical indication
#Gold standard for diagnostic imaging in CNO
&CNO is associated with psoriasis, PPP, acne, IBD, spondyloarthropathy/ERA and FMF
AID, auto-inflammatory disease; ALP, alkaline phosphatase; CBC, complete blood count; CNO, chronic nonbacterial osteomyelitis; CRP, C-reactive protein; ERA, enthesitis related arthritis; ESR, erythrocyte sedimentation rate; FMF, familial Mediterranean fever; GIS, gastrointestinal system; HSM, hepatosplenomegaly; IBD, inflammatory bowel disease; LAP, lymphadenopathy; LDH, lactate dehydrogenase; MRI, magnetic resonance imaging; PPP, palmoplantar pustulosis; P, phosphate; PCR, polymerase chain reaction; USG, ultrasonography.
CBC findings are usually within normal limits. A mild elevation in white blood cell count may occasionally be observed, whereas cytopenia is uncommon and should prompt evaluation for possible hematologic malignancy.32 Mild elevations in inflammatory markers such as CRP and ESR are reported in nearly half of the patients, with frequencies ranging between 49% and 80%. Markedly increased values (for example, threefold above the upper limit of normal) should raise suspicion for other underlying conditions.29,44 Autoantibodies, including antinuclear antibodies (ANA) and HLA-B27, may rarely be detected in some individuals, but these lack diagnostic specificity.16-18,29,37,45-47 Other useful laboratory tests are primarily aimed at differential diagnosis: lactate dehydrogenase (LDH) and uric acid for malignancies, alkaline phosphatase for hypophosphatasia, and vitamin C levels for scurvy. In CNO, these parameters are usually within normal limits. Since no serologic marker is disease-specific or reliable for monitoring, laboratory results should always be interpreted alongside the clinical and imaging context.43
Recent investigations have identified elevated levels of several cytokines and chemokines in CNO, such as TNF-α, IL-6, IL-12, monocyte chemoattractant protein-1 (MCP-1), C-C motif ligand 11 (CCL11), CCL5, collagen 1α, and the soluble IL-2 receptor (sIL-2R).37,48,49 These mediators may eventually aid in distinguishing CNO from infectious or malignant mimickers and in assessing inflammatory activity. However, current evidence is preliminary and requires confirmation in larger, ethnically diverse cohorts using standardized analytic methods.43 In addition, urinary N-telopeptide has been proposed as a possible biomarker of disease flares in patients receiving bisphosphonate therapy, but this observation also awaits independent validation.50
A bone biopsy may be warranted to exclude infection or malignancy, particularly in unifocal disease, atypical lesion sites, very early onset (< 2 years of age), or unexpected progression despite treatment.1,35,51 When indicated, sampling should target the most active or symptomatic bone lesion. Histopathological findings in CNO/CRMO are not pathognomonic but rather demonstrate features of chronic inflammation.27 Typical observations include bone marrow edema, vascular congestion, lymphoplasmacytic infiltration, and varying degrees of fibrosis or sclerosis. In long-standing lesions, osteonecrosis may occur, whereas biopsies from inactive sites may appear normal. Although non-specific, these features help confirm an inflammatory process and exclude other etiologies.10,13,35,52,53
Radiologic Imaging in CNO
Imaging approach
CNO generally presents with non-specific clinical signs and/or laboratory tests, which underscores the importance of imaging techniques. Radiographs may reveal diagnostic or suspicious findings for CNO or may be unremarkable. The subsequent approach should involve either targeted or whole-body magnetic resonance imaging (WB-MRI). Presently, MRI has become the primary imaging modality for diagnosing CNO, monitoring treatment effectiveness and follow-up. In addition, WB-MRI may be crucial in treatment decisions with its capability to show disease burden including asymptomatic foci of osteitis.11,54 Computed tomography (CT) has limited use due to radiation exposure and its inability to effectively demonstrate bone marrow lesions. However, in certain anatomical regions, including the pelvis, scapula, and spine, CT is superior to both radiographs and MRI in demonstrating CNO-related osteosclerosis.33
Lesion characteristics in imaging
Radiographs and CT
Radiographs and CT reveal round or ovoid osteolytic bone lesions centered at physeal lines which extend to both epiphysis and metaphysis of long bones (Fig. 3a). Sclerotic changes develop subsequently, progressing to hyperostosis and bony expansion, accompanied by a smooth or lamellated periosteal reaction in persistent chronic lesions (Fig. 3b). However, radiographic findings may be occult or normal at the early or healing phases of the disease.11,33
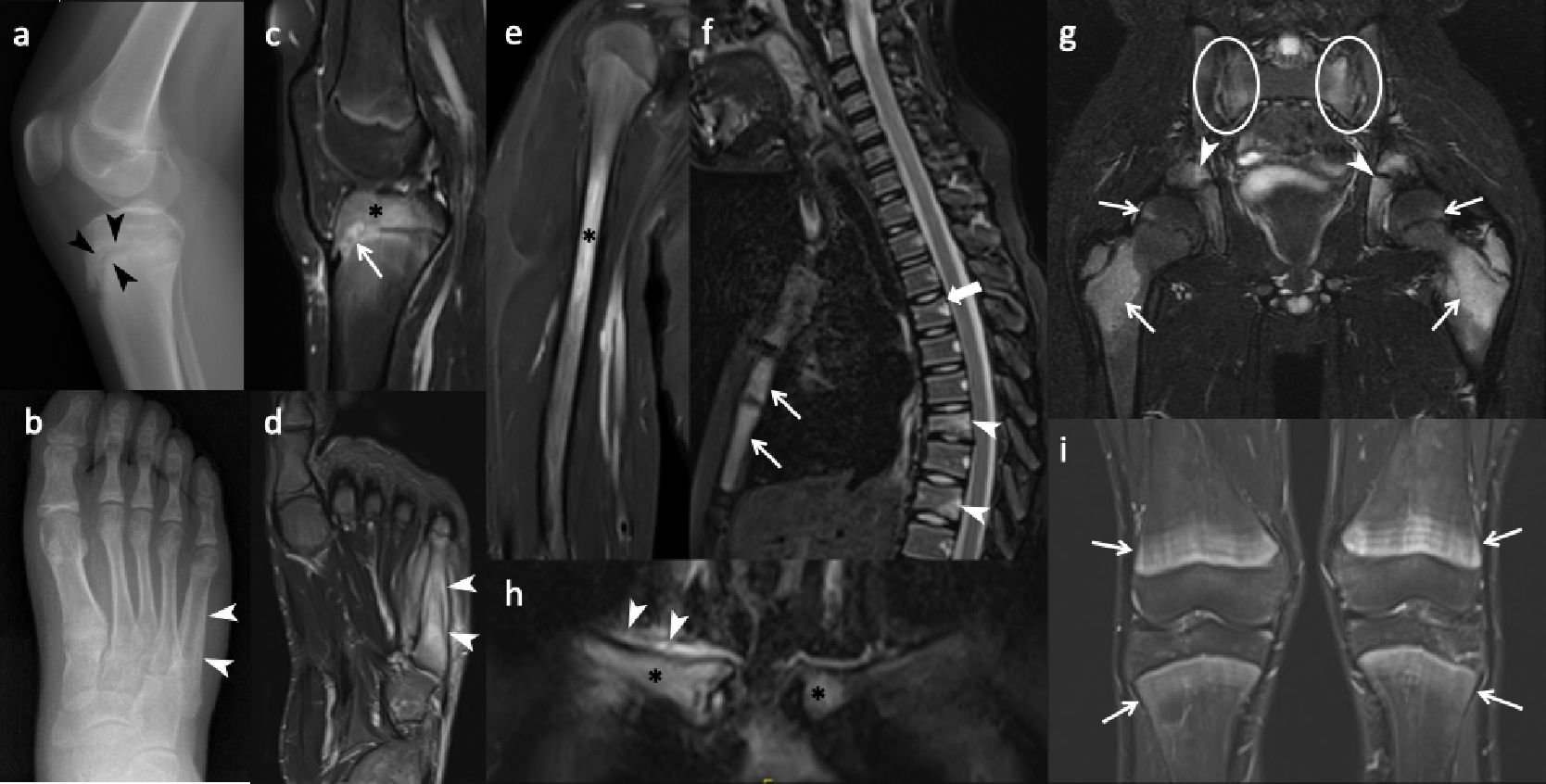
In addition to the initial work-up, radiographs are essential for follow-up of complications. Due to physitis, early physeal closure may occur through the formation of a physeal bridge or bar. Radiographs can demonstrate the characteristic “physeal tongue,” which appears as a lytic or sclerotic tubular structure extending toward the metaphysis. Another point in follow-up imaging is treatment associated findings in patients who were treated with cyclic bisphosphonates (e.g., pamidronate), including fine sclerotic lines in the metaphysis known as zebra stripes, resulting from inhibited osteoclast activity.39
Magnetic resonance imaging
Whether focused or encompassing the whole body, CNO presents several unique characteristics on MRI. The hallmark of CNO on MRI is physitis related periphyseal osteitis that appears as hyperintense foci on fluid sensitive sequences (STIR or T2-weighted fat-saturated) and hypointense foci on T1-weighted images (Fig. 3c). Periosteal reaction, surrounding soft tissue edema and reactive synovitis may accompany the osteitis (Fig. 3d).54 Lesions more commonly locate at the metaphysis or epi-/apophysis of a long bone (Fig. 3c). Diaphyseal involvement is less common (Fig. 3e). In flat and round bones, lesions affect the epi- or metaphyseal-equivalent regions of each bone that are in close proximity to the growth plates (i.e., apophyses of the iliac bones, endplates of the vertebrae, bones surrounding sacroiliac joints, Y-cartilage, ischiopubic synchondrosis) (Fig. 3f, 3g).11,54 Pelvic bones including the proximal femurs and sacroiliac joints are the most commonly involved regions across the body. Long bones of the lower extremity (lower legs and feet > bones around the knee), upper extremities, and vertebral bodies are other sites that are usually involved in descending order. The clavicle, sternum, scapula, and mandible are other involved body parts (Fig. 3h).39 In the spine, the thoracic and sacral vertebrae are the most commonly involved sites and osteitis is usually located focally at the superior endplate of the vertebra.39,55 More importantly, the spine is the commonest site of pathological fracture in CNO and as high as half of cases with spinal disease resulted in vertebral deformities such as vertebra plana or scoliosis (Fig. 3f). 33
After treatment, osteitis foci may fully resolve or heal with sclerosis. Vertebral height loss may persist without osteitis as a marker of previous disease. A diagnostic pitfall after bisphosphonate treatment is the presence of bilateral symmetric bright metaphyseal bands on STIR. These high-signal bands can mimic disease relapse, and their abrupt demarcation in relation to the adjacent metadiaphysis can be a key feature for differentiating them from true osteitis (Fig. 3i).54
CNO mimickers
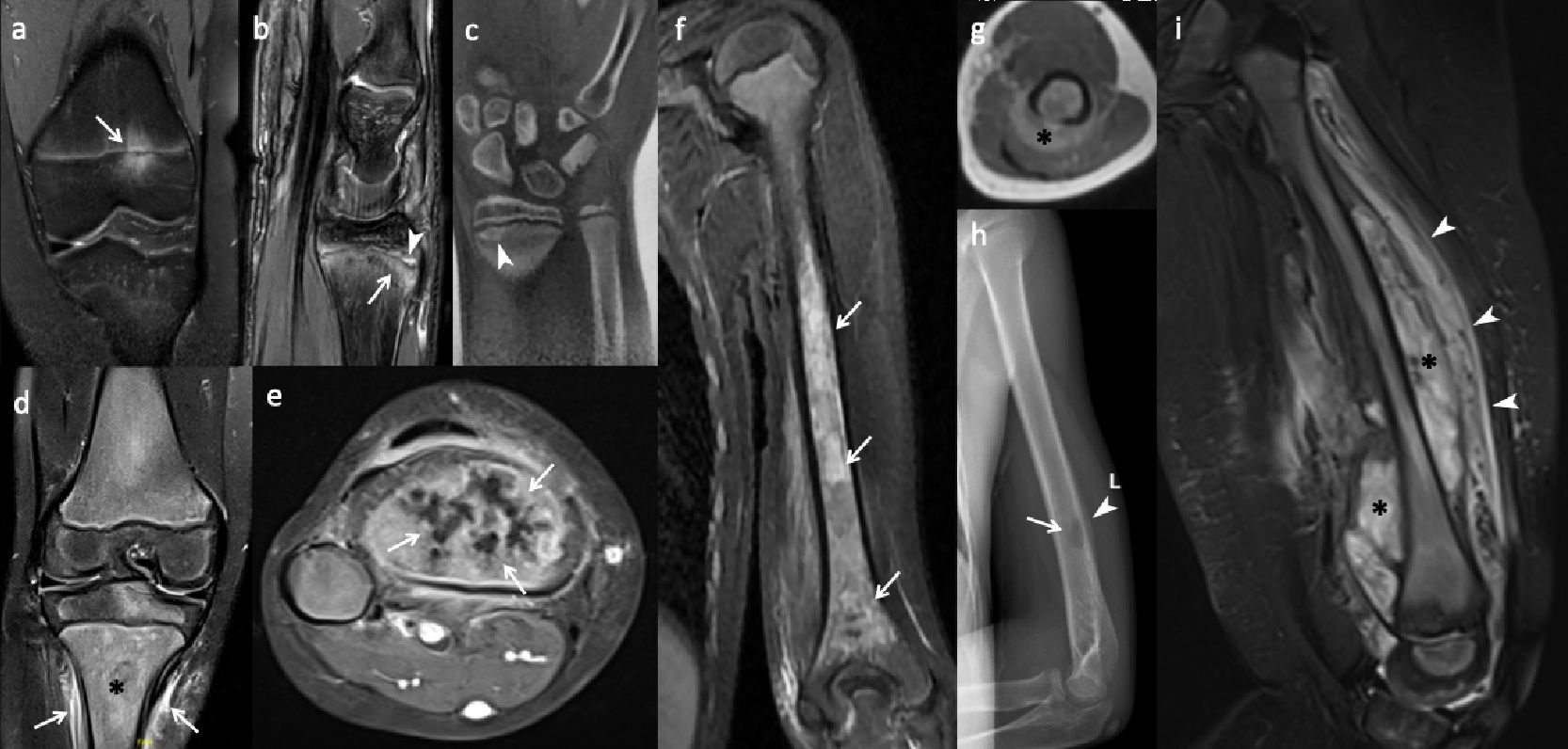
Normal variants of bone marrow, such as red marrow in the metaphysis during childhood and residual red bone marrow foci in the feet, should not be misinterpreted as osteitis (Fig. 4d). Focal periphyseal edema (FOPE) is another incidental finding located around the physis during the physeal closure period (Fig. 4a). Bone contusions, physeal stress injuries, stress fractures, or reactions may result in or associated with osteitis, but history of trauma—either acute or chronic—and the location of these lesions can help differentiate them from CNO (Fig. 4b, 4c). Hematogenous osteomyelitis can also mimic osteitis in CNO, but in addition to the patient’s history, physical examination, and laboratory findings, the presence of an abscess inside the bone and surrounding soft tissue, heterogeneous enhancement of bone marrow, and localized disease (as opposed to multifocal lesions) can aid in differentiation between these two entities (Fig. 4d, 4e). Neoplastic, infiltrative, or metastatic bone lesions, such as Ewing sarcoma, Langerhans cell histiocytosis, and leukemia, can pose diagnostic challenges; however, factors such as the patient’s age, the presence of an extraosseous solid soft tissue component of the tumor, abrupt lesion contour, and lesion location and shape are key features to consider when evaluating these pathologies (Fig. 4f‒h). Scurvy, as a metabolic condition, is another mimicker that presents with metaphyseal osteitis. However, subperiosteal hemorrhages are not seen in CNO (Fig. 4i).33,54
Diagnosis and Classification Criteria
Diagnosis is challenging and typically relies on excluding other potential conditions. It is traditionally established through a combination of clinical evaluation, radiologic findings, histologic examination, and characteristic signs and symptoms.3 Differential diagnoses of CNO are summarized in Table I.
Until recently, the absence of validated classification criteria for CNO posed a major limitation in both research and clinical practice. Earlier attempts to define such criteria were derived from small, single-center cohorts and lacked external validation, which limited their applicability and use in multicenter studies. Classification frameworks, in contrast to diagnostic criteria, are essential to delineate homogeneous patient populations, support epidemiologic investigations, and ensure consistency in evaluating disease features and treatment responses.13 Two independent sets of classification/diagnosis criteria for CNO/CRMO were previously proposed both based on retrospective analyses encompassing patients with diverse clinical presentations—ranging from isolated inflammatory bone lesions to multifocal and relapsing disease.10,37 However, the small sample sizes and absence of systematic comparison with alternative diagnoses, except for bacterial osteomyelitis, restricted their reliability. Moreover, neither set underwent prospective validation in independent cohorts. Consequently, universally accepted and externally validated classification criteria for CNO remained unavailable.11,13 To address this gap, an international collaborative effort between the European Alliance of Associations for Rheumatology (EULAR) and the American College of Rheumatology (ACR) recently resulted in the development and validation of new pediatric CNO classification criteria (Table II).56 These criteria incorporate a weighted scoring system that integrates clinical, imaging, and laboratory domains. They represent an important advance and are expected to facilitate consistency in research and clinical trial settings. However, it should be emphasized that classification frameworks are primarily designed for research standardization rather than for routine diagnosis. Consequently, some patients with clinically evident CNO may not fulfill the required threshold for classification.11,57 Recently, we evaluated the validation of this newly proposed classification criteria in our own patient cohort. The EULAR/ACR classification criteria for pediatric CNO demonstrated high specificity (97.9%) and sensitivity (92.7%), confirming their robustness and clinical applicability.57 Multifocal and symmetric bone lesions, especially those involving the clavicle and mandible, were strongly associated with fulfillment of these criteria. Validation within a pediatric cohort further underscores their value for standardized disease classification and consistency across research studies.57
|
CNO, chronic nonbacterial osteomyelitis; CT, computed tomography; LDH, lactate dehydrogenase; MRI, magnetic resonance imaging; PET/CT: positron emission tomography/computed tomography; WBMRI: whole body MRI. *Typical imaging findings include but are not limited to lytic, sclerotic lesions with or without periosteal reaction, hyperostosis on radiograph or CT, bone marrow edema or hyperintensity in a fluid-sensitive sequence of MRI. Non-arthritic bone sites refer to bones whose joint-forming ends do not demonstrate imaging signs of arthritis including synovial thickening, enhancement and/or moderate to large joint effusions. **Includes but not limited to primary or metastatic malignancy in bone, leukemia, lymphoma, neuroblastoma, infectious osteomyelitis, metabolic bone disease, vitamin C deficiency, hypophosphatasia, and monogenic bone diseases including Majeed or deficiency of interleukin-1 receptor antagonist (DIRA). ***Due to difference in laboratory assays across institutions, no absolute threshold is defined. Levels ≥ 700 IU or > 2 times of the upper normal limit may be considered pathological. #Defined as the presence of immune cells including neutrophils, monocytes, lymphocytes and/or plasma cells. &Axial arthritis is defined as imaging confirmation of inflammation within the sacroiliac joint or intervertebral joint. $Cutaneous conditions include psoriasis, palmoplantar pustulosis, pyoderma gangrenosum, acne fulminans, and hidradenitis suppurativa. |
|
| Table II. EULAR/ACR classification criteria for pediatric CNO.56 | |
|
Step 1, Verify entry criteria (all should be present): 1. Bone pain and/or musculoskeletal functional limitation ≥6 weeks; 2. Age of onset <18 years old; 3. Abnormal findings from radiograph and/or advanced imaging including MRI, CT, bone scintigraphy at non-arthritic bone sites* |
|
|
Step 2, Verify exclusion criteria (none should be present): 1. Confirmatory evidence of mutually exclusive mimicker diseases**; 2. Platelet <100,000/mm3; 3. Pathological LDH concerning malignancy***; 4. Complete and sustained clinical and laboratory response to antimicrobial treatment alone. |
|
|
Step 3, Add the score: Add the highest value in each of the 9 domains below. A score ≥ 55 is required to classify a patient as CNO. |
|
| Criteria domains / levels |
|
| Bone biopsy | |
| Signs of inflammation# AND fibrosis |
|
| Signs of fibrosis only |
|
| Signs of inflammation only |
|
| No signs of inflammation or fibrosis in bone biopsy |
|
| Age at the onset of symptoms | |
| ≥ 3 years old |
|
| < 3 years old |
|
| Sites of bone lesions based on imaging | |
| Clavicle and/or mandible |
|
| Sites other than clavicle, mandible, skull, or hand |
|
| Skull or hand without clavicle or mandible |
|
| Distribution pattern of bone lesions based on imaging | |
| Multifocal lesions (in ≥ 2 bones) with symmetrical pattern (bilateral involvement of at least one bone) |
|
| Multifocal without any symmetrical pattern bone involvement |
|
| Unifocal without whole body imaging such as WBMRI, PET/CT performed |
|
| Unifocal with whole body imaging performed |
|
| Coexisting conditions prior to the diagnosis of CNO | |
| Inflammatory bowel disease (IBD) |
|
| Cutaneous condition$ without IBD |
|
| Axial arthritis& without cutaneous condition or IBD |
|
| None |
|
| Hemoglobin (normal range varies by age) | |
| ≥ 10 g/dL |
|
| < 10 g/dL |
|
| Fever (oral/temporal temperature above 38 °C and not related to common infections) | |
| Absence of fever |
|
| Presence of fever |
|
| Erythrocyte sedimentation rate (Normal range: 0-20 mm/hr) | |
| <60 mm/hr |
|
| ≥ 60 mm/hr |
|
| C-reactive protein (Normal range: 0-10 mg/L) | |
| < 30 mg/L |
|
| ≥ 30 mg/L |
|
Fig. 2. provides an overview of the diagnostic algorithm for patients with CNO.
Treatment
Despite significant progress in understanding its clinical and immunopathologic spectrum, a standardized therapeutic protocol for CNO has not yet been established. Current management strategies are largely empirical and guided by disease severity, lesion distribution, and response to previous therapies.1,30 Recently, the Childhood Arthritis and Rheumatology Research Alliance (CARRA) reviewed consensus treatment protocols aimed at standardizing therapeutic approaches and prospectively capturing real-world data on treatment outcomes.29,51
NSAIDs represent the initial treatment of choice in patients without vertebral lesions and may lead to remission in some individuals. Nevertheless, disease relapses occur in over 50% of patients within two years of starting therapy.11,16,29,46 For refractory or multifocal cases, glucocorticoids, disease-modifying antirheumatic drugs (DMARDs) such as sulfasalazine and methotrexate (MTX), biologic agents targeting TNF-α, and bisphosphonates have also been reported to be effective in the treatment of CNO.2
In patients with vertebral involvement or insufficient response to NSAID monotherapy, CARRA recommends three main categories of systemic treatment: (1) conventional synthetic DMARDs, (2) bisphosphonates, and (3) TNF inhibitors, which can be administered alone or in combination with MTX to reduce anti-drug antibody formation.51
Despite the lack of head-to-head comparative trials and universally accepted treatment algorithms, most rheumatologists favor bisphosphonate therapy—either as monotherapy or in combination with other DMARDs—particularly in children with spinal involvement.25,29,50,58,59 In addition, CARRA suggests that a short course of systemic glucocorticoids (up to six weeks) may be used to manage acute disease flares alongside these systemic agents.51
Emerging biologic and targeted therapies, including IL-1 and IL-6 inhibitors, are currently under investigation for refractory cases and for monogenic autoinflammatory bone diseases. However, contrary to expectations, variable treatment responses have been reported in the literature with these drugs.60,61 The limited efficacy observed may be attributed to pathophysiological heterogeneity and tissue-level differences in therapeutic response1,30
IL-17/IL-23 also are potential therapeutic targets for CNO. The Janus kinase (JAK) signaling pathway has emerged as a key regulator of innate and adaptive immune responses, orchestrating cytokine-mediated inflammation. Pharmacologic inhibition of JAKs has been shown to modulate the production of proinflammatory cytokines such as IL-6, IL-17, and IL-23.30 Biologic agents targeting the IL-17 pathway, such as secukinumab, have been administered to patients with SAPHO syndrome, largely owing to their proven efficacy in psoriasis.11,62,63 Nevertheless, further studies are required to establish their safety profile and clinical efficacy in this population.11
Prognosis and Outcome
CNO typically follows a relapsing–remitting course. Although outcomes vary among studies, the overall consensus is that CNO is not as benign as previously believed. With longer follow-up durations, patients initially thought to be in remission may experience disease flares.17,18,43
In a cohort of 178 patients followed for an average of four years, 57% remained non-remissive, and 26% developed permanent sequelae.17 While in many cases the disease progresses initially and later subsides without lasting disability, several studies have documented persistent or severe disease courses in a subset of patients. Reported long-term complications include vertebral collapse or fracture leading to kyphosis, scoliosis, or neurological impairment; limb-length discrepancies secondary to growth plate involvement; extremity deformities (varus/valgus); recurrent clavicular lesions resulting in thoracic outlet syndrome; and mandibular involvement causing malocclusion or masticatory dysfunction.7,18,43,51,53
Long-term follow-up studies suggest that disease activity can persist for several years in a subset of patients. A longitudinal study in adults with CNO reported disease recurrence as late as 15 years after onset, emphasizing the importance of sustained follow-up and a coordinated transition to adult rheumatology care.35,52
Conclusion
CNO is an autoinflammatory bone disorder characterized by dysregulated cytokine activity. Clinical manifestations range from mild, localized pain to multifocal skeletal involvement. WB-MRI remains the gold standard for diagnosis and follow-up, yet CNO continues to be regarded as a diagnosis of exclusion owing to the absence of specific biomarkers. Early recognition is crucial to prevent complications such as vertebral fractures and chronic pain. Recent consensus treatment plans and translational research have improved understanding of disease pathophysiology, but further studies are needed to validate biomarkers for early diagnosis and differentiation from mimicking conditions, as well as long-term prognosis, and to refine long-term management strategies.
Source of funding
The authors declare the study received no funding.
Conflict of interest
The authors declare that there is no conflict of interest.
References
- Hofmann SR, Kapplusch F, Girschick HJ, et al. Chronic Recurrent Multifocal Osteomyelitis (CRMO): presentation, pathogenesis, and treatment. Curr Osteoporos Rep 2017; 15: 542-554. https://doi.org/10.1007/s11914-017-0405-9
- Hedrich CM, Hahn G, Girschick HJ, Morbach H. A clinical and pathomechanistic profile of chronic nonbacterial osteomyelitis/chronic recurrent multifocal osteomyelitis and challenges facing the field. Expert Rev Clin Immunol 2013; 9: 845-854. https://doi.org/10.1586/1744666X.2013.824670
- Buch K, Thuesen ACB, Brøns C, Schwarz P. Chronic non-bacterial osteomyelitis: a review. Calcif Tissue Int 2019; 104: 544-553. https://doi.org/10.1007/s00223-018-0495-0
- Giedion A. [Subacute and chronic “symmetrical” osteomyelitis]. Ann Radiol 1972; 15: 329-342.
- Björkstén B, Gustavson KH, Eriksson B, Lindholm A, Nordström S. Chronic recurrent multifocal osteomyelitis and pustulosis palmoplantaris. J Pediatr 1978; 93: 227-231. https://doi.org/10.1016/s0022-3476(78)80501-0
- Girschick HJ, Raab P, Surbaum S, et al. Chronic non-bacterial osteomyelitis in children. Ann Rheum Dis 2005; 64: 279-285. https://doi.org/10.1136/ard.2004.023838
- Ramachandran S, Zhao Y, Ferguson PJ. Update on treatment responses and outcome measure development in chronic nonbacterial osteomyelitis. Curr Opin Rheumatol 2023; 35: 255-264. https://doi.org/10.1097/BOR.0000000000000954
- Stern SM, Ferguson PJ. Autoinflammatory bone diseases. Rheum Dis Clin North Am 2013; 39: 735-749. https://doi.org/10.1016/j.rdc.2013.05.002
- Chang E, Vickery J, Zaiat N, et al. Chronic Recurrent Multifocal Osteomyelitis (CRMO): a study of 12 cases from one institution and literature review. Fetal Pediatr Pathol 2022; 41: 759-770. https://doi.org/10.1080/15513815.2021.1978602
- Roderick MR, Shah R, Rogers V, Finn A, Ramanan AV. Chronic Recurrent Multifocal Osteomyelitis (CRMO) - advancing the diagnosis. Pediatr Rheumatol Online J 2016; 14: 47. https://doi.org/10.1186/s12969-016-0109-1
- Lim L, Laxer RM. Chronic nonbacterial osteomyelitis: a noninfectious autoinflammatory disorder of bone. J Pediatr 2025; 283: 114636. https://doi.org/10.1016/j.jpeds.2025.114636
- Price-Kuehne F, Armon K. Chronic Non-bacterial Osteomyelitis (CNO) In Childhood: a review. Curr Pediatr Rev 2024; 20: 405-411. https://doi.org/10.2174/1573396319666221027123723
- Singhal S, Landes C, Shukla R, McCann LJ, Hedrich CM. Classification and management strategies for paediatric chronic nonbacterial osteomyelitis and chronic recurrent multifocal osteomyelitis. Expert Rev Clin Immunol 2023; 19: 1101-1116. https://doi.org/10.1080/1744666X.2023.2218088
- Jansson AF, Grote V, ESPED Study Group. Nonbacterial osteitis in children: data of a German incidence surveillance study. Acta Paediatr 2011; 100: 1150-1157. https://doi.org/10.1111/j.1651-2227.2011.02205.x
- Walsh P, Manners PJ, Vercoe J, Burgner D, Murray KJ. Chronic recurrent multifocal osteomyelitis in children: nine years’ experience at a statewide tertiary paediatric rheumatology referral centre. Rheumatology (Oxford) 2015; 54: 1688-1691. https://doi.org/10.1093/rheumatology/kev013
- Schnabel A, Range U, Hahn G, Siepmann T, Berner R, Hedrich CM. Unexpectedly high incidences of chronic non-bacterial as compared to bacterial osteomyelitis in children. Rheumatol Int 2016; 36: 1737-1745. https://doi.org/10.1007/s00296-016-3572-6
- Wipff J, Costantino F, Lemelle I, et al. A large national cohort of French patients with chronic recurrent multifocal osteitis. Arthritis Rheumatol 2015; 67: 1128-1137. https://doi.org/10.1002/art.39013
- Girschick H, Finetti M, Orlando F, et al. The multifaceted presentation of chronic recurrent multifocal osteomyelitis: a series of 486 cases from the Eurofever international registry. Rheumatology (Oxford) 2018; 57: 1504. https://doi.org/10.1093/rheumatology/key143
- Andronikou S, Mendes da Costa T, Hussien M, Ramanan AV. Radiological diagnosis of chronic recurrent multifocal osteomyelitis using whole-body MRI-based lesion distribution patterns. Clin Radiol 2019; 74: 737.e3-737.e15. https://doi.org/10.1016/j.crad.2019.02.021
- Beck C, Morbach H, Beer M, et al. Chronic nonbacterial osteomyelitis in childhood: prospective follow-up during the first year of anti-inflammatory treatment. Arthritis Res Ther 2010; 12: R74. https://doi.org/10.1186/ar2992
- Aden S, Wong S, Yang C, et al. Increasing cases of chronic nonbacterial osteomyelitis in children: a series of 215 cases from a single tertiary referral center. J Rheumatol 2022; 49: 929-934. https://doi.org/10.3899/jrheum.210991
- Sağ E, Sönmez HE, Demir S, et al. Chronic recurrent multifocal osteomyelitis in children: a single center experience over five years. Turk J Pediatr 2019; 61: 386-391. https://doi.org/10.24953/turkjped.2019.03.010
- Sözeri B, Ayaz NA, Atıkan BY, et al. Clinical experiences in Turkish pediatric patients with chronic recurrent multifocal osteomyelitis. Turk J Pediatr 2019; 61: 879-884. https://doi.org/10.24953/turkjped.2019.06.009
- Ulu K, Işgüder R, Gül Karadağ Ş, et al. Clinical characteristics and predictors for recurrence in chronic nonbacterial osteomyelitis: a retrospective multicenter analysis. Turk J Med Sci 2023; 53: 1105-1111. https://doi.org/10.55730/1300-0144.5675
- Hofmann SR, Schnabel A, Rösen-Wolff A, Morbach H, Girschick HJ, Hedrich CM. Chronic nonbacterial osteomyelitis: pathophysiological concepts and current treatment strategies. J Rheumatol 2016; 43: 1956-1964. https://doi.org/10.3899/jrheum.160256
- Kaya Akca U, Sag E, Aydın B, et al. Chronic non-bacterial osteomyelitis and immune checkpoint molecules. Clin Rheumatol 2024; 43: 553-560. https://doi.org/10.1007/s10067-023-06761-y
- Wipff J, Adamsbaum C, Kahan A, Job-Deslandre C. Chronic recurrent multifocal osteomyelitis. Joint Bone Spine 2011; 78: 555-560. https://doi.org/10.1016/j.jbspin.2011.02.010
- Leerling AT, Andeweg EH, Faber J, et al. immunological characterization of Chronic Nonbacterial Osteomyelitis (CNO) in adults: a cross-sectional exploratory study. JBMR Plus 2023; 7: e10818. https://doi.org/10.1002/jbm4.10818
- Zhao DY, McCann L, Hahn G, Hedrich CM. Chronic Nonbacterial Osteomyelitis (CNO) and Chronic Recurrent Multifocal Osteomyelitis (CRMO). J Transl Autoimmun 2021; 4: 100095. https://doi.org/10.1016/j.jtauto.2021.100095
- Roberts E, Charras A, Hahn G, Hedrich CM. An improved understanding of pediatric chronic nonbacterial osteomyelitis pathophysiology informs current and future treatment. J Bone Miner Res 2024; 39: 1523-1538. https://doi.org/10.1093/jbmr/zjae141
- Reiser C, Klotsche J, Hospach A, et al. First-year follow-up of children with chronic nonbacterial osteomyelitis-an analysis of the German National Pediatric Rheumatologic Database from 2009 to 2018. Arthritis Res Ther 2021; 23: 281. https://doi.org/10.1186/s13075-021-02658-w
- Triaille C, De Bruycker JJ, Miron MC, Lecouvet F, Girschick H, Wouters C. Update on the diagnosis and treatment of CNO in children: a clinician’s perspective. Eur J Pediatr 2024; 184: 48. https://doi.org/10.1007/s00431-024-05823-w
- Sheikh Z, Bhatt D, Chowdhury M, Johnson K. Challenges in the imaging and diagnosis of Chronic Non-bacterial Osteomyelitis (CNO). Clin Radiol 2025; 85: 106905. https://doi.org/10.1016/j.crad.2025.106905
- Borzutzky A, Stern S, Reiff A, et al. Pediatric chronic nonbacterial osteomyelitis. Pediatrics 2012; 130: e1190-e1197. https://doi.org/10.1542/peds.2011-3788
- Koryllou A, Mejbri M, Theodoropoulou K, Hofer M, Carlomagno R. Chronic nonbacterial osteomyelitis in children. children (Basel) 2021; 8: 551. https://doi.org/10.3390/children8070551
- Nico MAC, Araújo FF, Guimarães JB, et al. Chronic nonbacterial osteomyelitis: the role of whole-body MRI. Insights Imaging 2022; 13: 149. https://doi.org/10.1186/s13244-022-01288-3
- Jansson A, Renner ED, Ramser J, et al. Classification of non-bacterial osteitis: retrospective study of clinical, immunological and genetic aspects in 89 patients. Rheumatology (Oxford) 2007; 46: 154-160. https://doi.org/10.1093/rheumatology/kel190
- Kostik MM, Kopchak OL, Maletin AS, Mushkin AY. The peculiarities and treatment outcomes of the spinal form of chronic non-bacterial osteomyelitis in children: a retrospective cohort study. Rheumatol Int 2020; 40: 97-105. https://doi.org/10.1007/s00296-019-04479-2
- Aydıngöz Ü, Yıldız AE, Ayaz E, Batu ED, Özen S. Preferential involvement of the pelvis and hips along with active sacroiliitis in chronic nonbacterial osteomyelitis: MRI of 97 patients from a single tertiary referral center. Eur Radiol 2024; 34: 4979-4987. https://doi.org/10.1007/s00330-023-10558-7
- Ferguson PJ, Laxer RM. Autoinflammatory bone disorders. In: Petty RE, Wedderburn LR, editors. Textbook of pediatric rheumatology. 7th ed. Philadelphia: Elsevier; 2016: 627-641. https://doi.org/10.1016/B978-0-323-24145-8.00048-X
- Öksüz Aydın B, Aydın F, Taş Ö, Bahçeci O, Özçakar ZB. Associated diseases and their effects on disease course in patients with chronic non-bacterial osteomyelitis: retrospective experience from a single center. Clin Rheumatol 2025; 44: 855-862. https://doi.org/10.1007/s10067-025-07306-1
- Karaçayir N, Tunçez Ş, Öner N, et al. Chronic nonbacterial osteomyelitis associated with familial mediterranean fever in children. J Clin Rheumatol 2024; 30: 89-93. https://doi.org/10.1097/RHU.0000000000002061
- Zhao Y, Ferguson PJ. Chronic nonbacterial osteomyelitis and chronic recurrent multifocal osteomyelitis in children. Pediatr Clin North Am 2018; 65: 783-800. https://doi.org/10.1016/j.pcl.2018.04.003
- Oliver M, Lee TC, Halpern-Felsher B, et al. Disease burden and social impact of pediatric chronic nonbacterial osteomyelitis from the patient and family perspective. Pediatr Rheumatol Online J 2018; 16: 78. https://doi.org/10.1186/s12969-018-0294-1
- Gaal A, Basiaga ML, Zhao Y, Egbert M. Pediatric chronic nonbacterial osteomyelitis of the mandible: Seattle children’s hospital 22-patient experience. Pediatr Rheumatol Online J 2020; 18: 4. https://doi.org/10.1186/s12969-019-0384-8
- Schnabel A, Range U, Hahn G, Berner R, Hedrich CM. Treatment response and longterm outcomes in children with chronic nonbacterial osteomyelitis. J Rheumatol 2017; 44: 1058-1065. https://doi.org/10.3899/jrheum.161255
- Kaiser D, Bolt I, Hofer M, et al. Chronic nonbacterial osteomyelitis in children: a retrospective multicenter study. Pediatr Rheumatol Online J 2015; 13: 25. https://doi.org/10.1186/s12969-015-0023-y
- Hofmann SR, Kubasch AS, Range U, et al. Serum biomarkers for the diagnosis and monitoring of Chronic Recurrent Multifocal Osteomyelitis (CRMO). Rheumatol Int 2016; 36: 769-779. https://doi.org/10.1007/s00296-016-3466-7
- Hofmann SR, Böttger F, Range U, et al. Serum interleukin-6 and CCL11/Eotaxin may be suitable biomarkers for the diagnosis of chronic nonbacterial osteomyelitis. Front Pediatr 2017; 5: 256. https://doi.org/10.3389/fped.2017.00256
- Miettunen PM, Wei X, Kaura D, Reslan WA, Aguirre AN, Kellner JD. Dramatic pain relief and resolution of bone inflammation following pamidronate in 9 pediatric patients with persistent Chronic Recurrent Multifocal Osteomyelitis (CRMO). Pediatr Rheumatol Online J 2009; 7: 2. https://doi.org/10.1186/1546-0096-7-2
- Zhao Y, Wu EY, Oliver MS, et al. Consensus treatment plans for chronic nonbacterial osteomyelitis refractory to nonsteroidal antiinflammatory drugs and/or with active spinal lesions. Arthritis Care Res (Hoboken) 2018; 70: 1228-1237. https://doi.org/10.1002/acr.23462
- Concha S, Hernández-Ojeda A, Contreras O, Mendez C, Talesnik E, Borzutzky A. Chronic nonbacterial osteomyelitis in children: a multicenter case series. Rheumatol Int 2020; 40: 115-120. https://doi.org/10.1007/s00296-019-04400-x
- Skrabl-Baumgartner A, Singer P, Greimel T, Gorkiewicz G, Hermann J. Chronic non-bacterial osteomyelitis: a comparative study between children and adults. Pediatr Rheumatol Online J 2019; 17: 49. https://doi.org/10.1186/s12969-019-0353-2
- Aydıngöz Ü, Yıldız AE. MRI in the diagnosis and treatment response assessment of chronic nonbacterial osteomyelitis in children and adolescents. Curr Rheumatol Rep 2022; 24: 27-39. https://doi.org/10.1007/s11926-022-01053-x
- Guariento A, Sharma P, Andronikou S. MRI features of spinal chronic recurrent multifocal osteomyelitis/chronic non-bacterial osteomyelitis in children. Pediatr Radiol 2023; 53: 2092-2103. https://doi.org/10.1007/s00247-023-05688-5
- Zhao Y, Oliver MS, Schnabel A, et al. EULAR/ACR classification criteria for paediatric Chronic Nonbacterial Osteomyelitis (CNO). Ann Rheum Dis 2025; 84: 1458-1468. https://doi.org/10.1016/j.ard.2024.11.006
- Unal D, Yigit YD, Basaran O, et al. Validation of the EULAR/ACR classification criteria for chronic nonbacterial osteomyelitis in a pediatric cohort. Clin Rheumatol 2025; 44: 4229-4236. https://doi.org/10.1007/s10067-025-07653-z
- Ferguson PJ, El-Shanti HI. Autoinflammatory bone disorders. Curr Opin Rheumatol 2007; 19: 492-498. https://doi.org/10.1097/BOR.0b013e32825f5492
- Hospach T, Langendoerfer M, von Kalle T, Maier J, Dannecker GE. Spinal involvement in Chronic Recurrent Multifocal Osteomyelitis (CRMO) in childhood and effect of pamidronate. Eur J Pediatr 2010; 169: 1105-1111. https://doi.org/10.1007/s00431-010-1188-5
- Eleftheriou D, Gerschman T, Sebire N, Woo P, Pilkington CA, Brogan PA. Biologic therapy in refractory chronic non-bacterial osteomyelitis of childhood. Rheumatology (Oxford) 2010; 49: 1505-1512. https://doi.org/10.1093/rheumatology/keq122
- Pardeo M, Pires Marafon D, Messia V, Garganese MC, De Benedetti F, Insalaco A. Anakinra in a cohort of children with chronic nonbacterial osteomyelitis. J Rheumatol 2017; 44: 1231-1238. https://doi.org/10.3899/jrheum.160690
- Goenka A, Roderick M, Finn A, Ramanan AV. The jigsaw puzzle of chronic non-bacterial osteomyelitis: are anti-IL17 therapies the next piece? Rheumatology (Oxford) 2020; 59: 459-461. https://doi.org/10.1093/rheumatology/kez492
- Hedrich CM, Morbach H, Reiser C, Girschick HJ. New insights into adult and pediatric Chronic Non-bacterial Osteomyelitis (CNO). Curr Rheumatol Rep 2020; 22: 52. https://doi.org/10.1007/s11926-020-00928-1
License
Copyright (c) 2026 The Author(s). This is an open access article distributed under the Creative Commons Attribution License (CC BY), which permits unrestricted use, distribution, and reproduction in any medium or format, provided the original work is properly cited.















