Graphical Abstract
Abstract
Background. Ovarian tumors are rare in children, accounting for 1-2% of all pediatric malignancies. Recent trends emphasize fertility preservation, but data on the evolution of practice and outcomes remain limited. This study examines changes in surgical management and outcomes of pediatric ovarian tumors over two decades.
Methods. All children who underwent surgery for ovarian pathologies at a tertiary pediatric surgery center between 2002 and 2021 were reviewed. Patients with histopathologically confirmed ovarian tumors were included. Clinical characteristics, surgical approaches, histopathology, and outcomes were analyzed. Patients were divided into two 10-year periods to evaluate trends over time, and age-based subgroup analysis compared prepubertal (≤12 years) and pubertal (>12 years) patients.
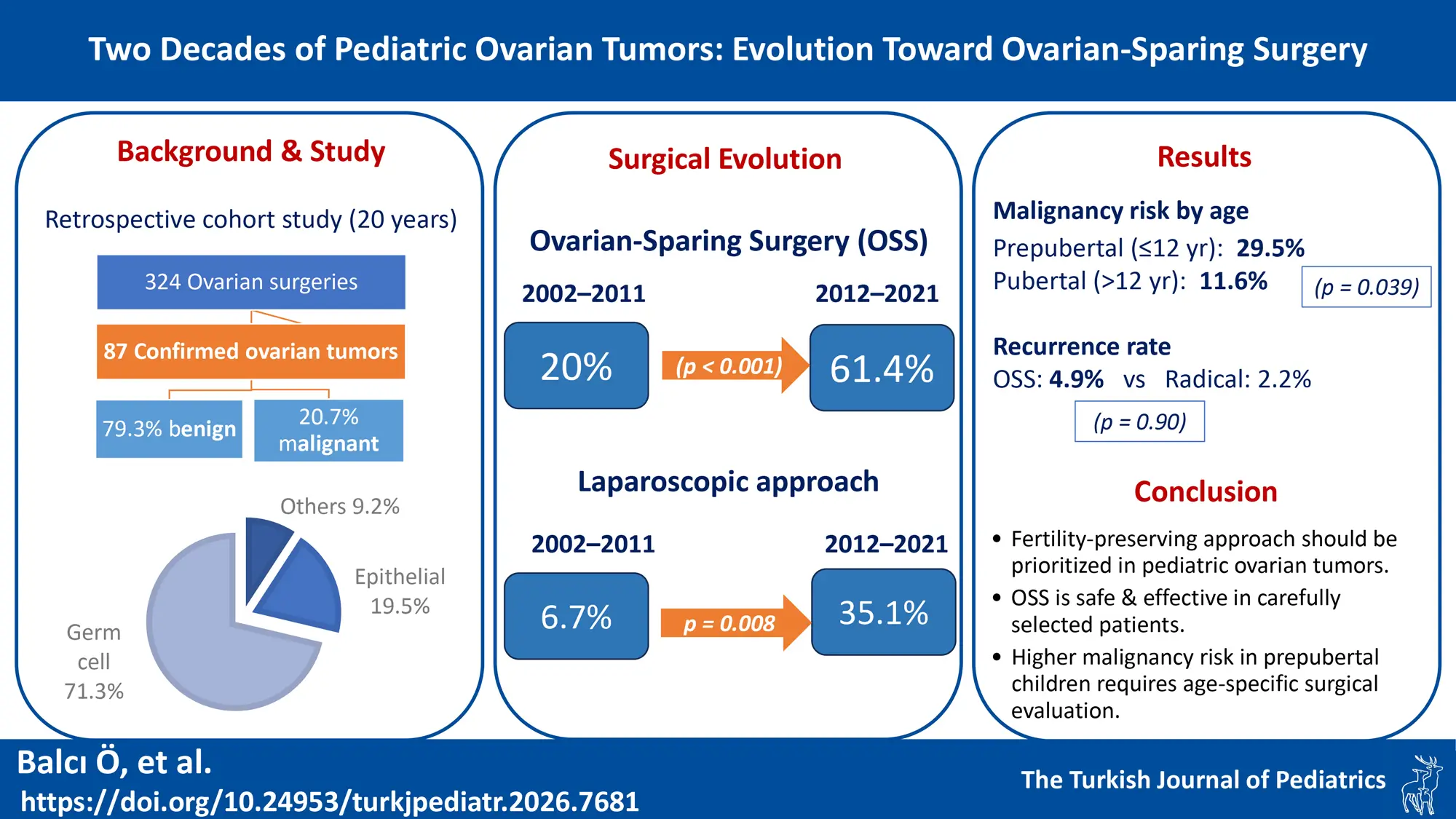
Results. Of 324 children undergoing ovarian surgery, 87 had histologically confirmed tumors. The most common histological type was mature cystic teratoma (54.0%). Eighteen tumors (20.7%) were malignant. Ovarian-sparing surgery (OSS) increased from 20.0% (6/30) to 61.4% (35/57) (p<0.001), and the laparoscopic approach increased from 6.7% to 35.1% (p=0.008). Patients undergoing OSS had significantly smaller tumors (median 7.0 cm vs 12.0 cm, p<0.001) and lower malignancy rates (2.4% vs 37.0%, p<0.001). Prepubertal patients had higher malignancy rates than pubertal patients (29.5% vs 11.6%, p=0.039). During a mean 4-year follow-up, recurrence rates were similar between OSS and oophorectomy groups (4.9% vs 2.2%, p=0.90).
Conclusions. Surgical management of pediatric ovarian tumors has evolved significantly toward fertility-preserving approaches. OSS is safe and effective in appropriately selected patients. The higher malignancy rate in prepubertal patients highlights the importance of age-specific evaluation. Careful preoperative assessment and case selection remain essential for optimal outcomes.
Keywords: ovarian tumor, pediatric, ovarian-sparing surgery, fertility preservation, oophorectomy, mature cystic teratoma
Introduction
Ovarian tumors are uncommon in the pediatric population, representing 1-2% of all childhood malignancies and approximately 1% of pediatric tumors overall.1-3 Despite their rarity, pediatric ovarian tumors pose distinct challenges in diagnosis and management. The clinical presentation varies widely; most patients present with abdominal pain, often due to complications such as torsion, rupture, or rapid tumor growth.1,4,5 Hormone-secreting tumors may cause precocious puberty or virilization in rare cases.2,6 The tumor profile in children differs markedly from adults; germ cell tumors account for the majority (~60-70%) of pediatric ovarian neoplasms, whereas epithelial tumors predominate in adults.3,7 Mature teratomas are the single most common pediatric ovarian tumor, accounting for 30-50% of all pediatric ovarian masses.2,5 Although most ovarian tumors in children are benign, about 10-20% prove to be malignant, especially in younger patients or when tumors exceed 10 cm in size.1,7,8
Historically, the surgical management of pediatric ovarian tumors predominantly involved oophorectomy or salpingo-oophorectomy.8,9 This approach resulted in permanent loss of ovarian function on the affected side with potential implications for fertility and hormonal function. In the last two decades, surgical practice has shifted markedly toward fertility-sparing approaches.9-11 Multiple studies demonstrate that in carefully selected cases, ovarian-sparing surgery (OSS) can be performed safely without compromising oncologic outcomes.12,13 Modern high-resolution imaging and tumor markers now allow more accurate preoperative assessment of malignancy risk, and a growing body of evidence confirms that OSS in children is both safe and effective, with low recurrence rates.12-22 Reported recurrence rates are low, and most recurrences are themselves benign and manageable with repeat conservative surgery.18,20,21 Minimally invasive techniques have also become more common, as pediatric surgeons recognize that laparoscopy can achieve oncologic goals while significantly reducing postoperative pain and recovery time.19,20 In particular, questions remain about how best to select patients for OSS, the utility of intraoperative frozen-section analysis, and the long-term fertility outcomes after OSS.23,24
Despite these advances, longitudinal data on the temporal evolution of surgical practice patterns and long-term outcomes of OSS in pediatric populations remain limited. Additionally, differences in tumor characteristics and surgical approach between prepubertal and pubertal patients have not been well characterized. This study aimed to describe the epidemiological distribution of pediatric ovarian tumors at a tertiary pediatric surgery center, analyze the temporal evolution in surgical management over 20 years, compare tumor characteristics and outcomes between prepubertal and pubertal patients, and evaluate the safety and oncologic outcomes of OSS.
Materials and Methods
This retrospective cohort study was conducted at a tertiary referral center. After ethics approval, medical records of all patients aged 0-18 years who underwent surgical intervention for ovarian pathologies between January 2002 and December 2021 were reviewed. Only patients with histopathologically confirmed neoplastic disease were included. Isolated ovarian torsion without underlying tumor and non-neoplastic ovarian lesions (functional cysts, simple cysts, hemorrhagic cysts) were excluded. A total of 87 patients met the inclusion criteria and formed the study cohort. Demographic characteristics, clinical presentation, preoperative imaging findings, laboratory data, surgical details, histopathological diagnosis, postoperative course, follow-up data, and outcomes were collected for each patient.
The 20-year study period was divided into two equal decades (2002-2011 and 2012-2021) to analyze temporal trends in surgical management. This division allowed comparison of practice changes, particularly the increasing adoption of laparoscopic surgery and ovarian-sparing approaches, consistent with evolving trends reported in the literature.
For age-based subgroup analysis, patients were divided into prepubertal (≤12 years) and pubertal (>12 years) groups based on the average age of menarche in the Turkish population.
Ovarian-sparing surgery (OSS) was defined as any surgical procedure that preserved ovarian tissue; oophorectomy was defined as complete removal of the affected ovary, and salpingo-oophorectomy (S-O) as removal of both the ovary and fallopian tube on the affected side. Tumors were classified as benign or malignant based on the final histopathological diagnosis. Histological types were categorized into germ cell tumors, epithelial tumors, sex cord-stromal tumors, lymphoma, and metastatic tumors according to standard classification systems.
Surgical decision-making was based on an individualized assessment integrating preoperative evaluation and intraoperative findings. Factors considered included patient age, tumor size and characteristics on imaging, serum tumor marker levels (alpha-fetoprotein [AFP], beta-human chorionic gonadotropin [β-hCG], lactate dehydrogenase [LDH]), intraoperative appearance of the tumor, frozen section examination when available, and fertility preservation considerations. In general, OSS was preferred for benign-appearing tumors with normal tumor markers. Oophorectomy or salpingo-oophorectomy was performed for tumors with malignant features, large tumors precluding safe ovarian preservation, and cases with complete ovarian replacement by tumor.
In cases with ovarian torsion, detorsion was performed initially to assess ovarian viability. During the earlier study period, surgical decision-making was primarily based on ovarian circulation after detorsion, with oophorectomy performed for non-viable ovaries or when malignancy was confirmed or suspected. As our practice evolved in line with current evidence, ovarian circulation status was no longer considered a determining factor. In our current protocol, OSS is performed in all torsion cases regardless of circulation status after detorsion, unless the ovary is frankly necrotic or malignancy is confirmed or suspected.
Surgery was performed via either open laparotomy or laparoscopic approach. The surgical approach was determined based on tumor size, patient age, and body habitus, surgeon preference and experience, and institutional resources. Frozen section examination was utilized selectively at the surgeon’s discretion, particularly when malignancy was suspected. In cases of confirmed or suspected malignancy, staging procedures were performed, including inspection of all peritoneal surfaces, peritoneal washings, omentectomy, peritoneal biopsies, and contralateral ovarian inspection or biopsy as indicated. Malignant germ cell tumors were staged according to the Children’s Oncology Group (COG) staging system.25
Statistical analyses were performed using Jamovi software (version 2.5). Descriptive statistics were calculated for all variables. Continuous variables were expressed as means ± standard deviations (SD) for normally distributed data, or as median (Q1–Q3) for non-normally distributed data. Categorical variables were expressed as frequencies and percentages. The normality of continuous variables was assessed using the Shapiro-Wilk test. For normally distributed data, independent samples t-tests were used; for non-normally distributed data, Mann-Whitney U tests were employed. Categorical variables were compared using Pearson’s chi-square test or Fisher’s exact test. A p-value of <0.05 was considered statistically significant.
Results
Patient demographics and clinical characteristics are summarized in Table I. During the 20-year study period (2002-2021), a total of 324 patients underwent surgery for ovarian pathologies at our institution. Of these, 237 patients were excluded due to non-neoplastic conditions. The final analysis included 87 patients (26.9%) with histopathologically confirmed ovarian tumors. The median age at presentation was 11.9 years (Q1–Q3: 9.2–15.1 years). The most common presenting symptom was abdominal pain, occurring in 41 patients (47.1%), followed by abdominal distension (12.6%), menstrual irregularity (6.9%), and precocious puberty (5.7%). Five patients (5.7%) had incidentally discovered tumors. Eighteen patients (20.7%) presented with ovarian torsion and underwent emergency surgery at the time of admission, while the remaining 69 patients (79.3%) underwent elective surgery for ovarian masses.
| Table I. Patient demographics and clinical characteristics (N=87). | |
| Characteristic |
|
| Age, years, median (Q1–Q3) |
|
| Presenting symptom, n (%) | |
| Abdominal pain |
|
| Abdominal distension |
|
| Menstrual irregularity |
|
| Precocious puberty |
|
| Incidental finding |
|
| Other |
|
| Tumor laterality, n (%) | |
| Right |
|
| Left |
|
| Bilateral |
|
| Tumor size, cm, median (Q1–Q3) |
|
| Ovarian torsion, n (%) |
|
| Mean follow-up, years (mean ± SD) |
|
Tumor laterality showed a right-sided predominance, with 46 tumors (52.9%) occurring on the right ovary, 37 (42.5%) on the left, and 4 patients (4.6%) presenting with synchronous bilateral involvement. The median tumor size at surgery was 10.0 cm (Q1–Q3: 6.0–15.0 cm).
The temporal evolution of surgical approaches is summarized in Table II. Overall, ovarian-sparing surgery (OSS) was performed in 41 patients (47.1%), while oophorectomy or salpingo-oophorectomy was performed in 46 patients (52.9%). The surgical approach was predominantly open surgery in 65 cases (74.7%), with laparoscopic surgery performed in 22 cases (25.3%). When analyzing temporal trends, a dramatic shift in surgical practice was observed. The rate of ovarian-sparing surgery increased significantly from 20% (6/30) in the first period to 61.4% (35/57) in the second period (p<0.001). Concurrently, the use of the laparoscopic approach increased from 6.7% to 35.1% between the two periods (p=0.008). Laparoscopy was used significantly more often in OSS than in oophorectomy (39.0% vs 13.0%, p=0.011). Patients in the more recent period were older (median age 12.8 vs 11.1 years, p=0.025), had smaller tumors (median size 8.0 cm vs 13.5 cm, p<0.001) and a lower rate of malignancy (14.0% vs 33.3%, p=0.019).
| OSS = ovarian-sparing surgery; S-O = salpingo-oophorectomy. | |||
| Table II. Evolution of surgical approaches by time period. | |||
| Variable |
|
|
|
| Surgery type, n (%) |
|
||
| OSS |
|
|
|
| Oophorectomy/S-O |
|
|
|
| Surgical approach, n (%) |
|
||
| Open |
|
|
|
| Laparoscopic |
|
|
|
| Tumor size, cm, median (Q1–Q3) |
|
|
|
| Malignancy, n (%) |
|
|
|
| Age, years, median (Q1–Q3) |
|
|
|
Comparison of surgical groups is presented in Table III. Patients who underwent ovarian-sparing surgery had significantly smaller tumors compared to those who underwent oophorectomy (median size 7.0 cm, Q1–Q3: 5.0–10.0 vs median 12.0 cm, Q1–Q3: 9.0–20.0, p<0.001; mean size 8.1 ± 5.0 cm vs 14.5 ± 7.7 cm). Only 1 of 41 patients (2.4%) who underwent OSS had a malignant tumor, compared to 17 of 46 patients (37.0%) in the oophorectomy group (p<0.001).
| OSS = ovarian-sparing surgery; O/SO = oophorectomy/salpingo-oophorectomy. | |||
| Table III. Comparison between ovarian-sparing surgery and oophorectomy. | |||
| Variable |
|
|
|
| Age, years (mean ± SD) |
|
|
|
| Tumor size, cm, median (Q1–Q3) |
|
|
|
| Malignancy, n (%) |
|
|
|
| Torsion, n (%) |
|
|
|
| Histological type, n (%) |
|
||
| Germ cell tumors |
|
|
|
| Epithelial tumors |
|
|
|
| Sex cord-stromal |
|
|
|
| Other |
|
|
|
| Recurrence, n (%) |
|
|
|
Torsion was present in 18 of 87 patients (20.7%). Among torsion cases, the malignancy rate was notably low at 5.6% (1/18), compared to 24.6% (17/69) in patients without torsion (p=0.075) (Table III). There was no significant difference in tumor size between patients with and without torsion (median 10.0 cm vs 9.5 cm, p=0.773). Among those with torsion, 6 of 18 (33.3%) underwent ovarian-sparing surgery, compared with 35 of 69 (50.7%) in patients without torsion (p=0.188).
Histopathological findings are detailed in Table IV. Histopathological examination revealed that 69 tumors (79.3%) were benign and 18 tumors (20.7%) were malignant. Germ cell tumors comprised the majority of cases (62 tumors, 71.3%), followed by epithelial tumors (17 tumors, 19.5%), sex cord-stromal tumors (5 tumors, 5.7%), lymphoma (2 cases, 2.3%), and metastatic rhabdomyosarcoma (1 case, 1.2%). Mature cystic teratoma was the most common diagnosis, accounting for 47 cases (54.0%). All epithelial tumors were benign, consisting of serous cystadenoma or cystadenofibroma (12 cases, 13.8%) and mucinous cystadenoma (5 cases, 5.7%). The sex cord-stromal tumors included sclerosing stromal tumor (4 cases) and one unspecified sex cord-stromal tumor.
| Table IV. Histopathological distribution of ovarian tumors (N=87). | |
| Histological Type |
|
| Benign tumors |
|
| Malignant tumors |
|
| Germ cell tumors |
|
| Mature cystic teratoma |
|
| Immature teratoma |
|
| Dysgerminoma/gonadoblastoma |
|
| Yolk sac tumor |
|
| Mixed germ cell tumor |
|
| Epithelial tumors |
|
| Serous cystadenoma/cystadenofibroma |
|
| Mucinous cystadenoma |
|
| Sex cord-stromal tumors |
|
| Lymphoma |
|
| Rhabdomyosarcoma (metastatic) |
|
The 18 malignant tumors consisted of 15 malignant germ cell tumors (83.3% of malignant cases), 2 lymphomas (11.1%), and 1 rhabdomyosarcoma (5.6%). No malignant epithelial or sex cord-stromal tumors were identified. Among the 15 malignant germ cell tumors, staging according to COG criteria revealed 8 (53.3%) Stage I, 6 (40.0%) Stage III with malignant cytology or peritoneal/omental implants, and 1 (6.7%) Stage IV with liver and diaphragmatic metastasis (Table IV). Patients with malignant tumors had significantly larger masses compared to those with benign tumors (median size 17.5 cm vs 8.5 cm, p=0.005).
The distribution of tumor histology differed significantly between surgical groups (p<0.001). In the ovarian-sparing surgery group, germ cell tumors (21 cases, 51.2%) predominated, followed by epithelial tumors (16 cases, 39.0%) and sex cord-stromal tumors (4 cases, 9.8%). The oophorectomy/salpingo-oophorectomy group also consisted primarily of germ cell tumors (41 cases, 89.1%), with only 1 epithelial tumor (2.2%), 1 sex cord-stromal tumor (2.2%), and 3 cases of lymphoma or rhabdomyosarcoma (6.5%).
Age-based subgroup comparisons are presented in Table V. Age-based subgroup analysis revealed significant differences between prepubertal (≤12 years, n=44) and pubertal (>12 years, n=43) patients. Prepubertal patients had a significantly higher malignancy rate (29.5% vs 11.6%, p=0.039). The distribution of tumor types differed between groups: germ cell tumors predominated in prepubertal patients (84.1%) while epithelial tumors were more common in pubertal patients (30.2% vs 9.1%). Notably, all sex cord-stromal tumors (n=5) occurred in pubertal patients. Tumor size did not differ significantly between age groups (p=0.495). The rate of ovarian-sparing surgery was higher in pubertal patients, though not statistically significant (55.8% vs 38.6%, p=0.109). Torsion was more common in prepubertal patients (27.3% vs 14.0%), but this difference was not statistically significant (p=0.125).
| OSS = ovarian-sparing surgery. | |||
| Table V. Comparison between prepubertal and pubertal patients. | |||
| Variable |
|
|
|
| Malignancy, n (%) |
|
|
|
| OSS, n (%) |
|
|
|
| Torsion, n (%) |
|
|
|
| Tumor size, cm, median (Q1–Q3) |
|
|
|
| Tumor type, n (%) | |||
| Germ cell |
|
|
|
| Epithelial |
|
|
|
| Sex cord-stromal |
|
|
|
| Other |
|
|
|
Frozen section examination was documented in 9 cases (10.3%). Additional staging procedures were performed in cases of confirmed or suspected malignancy, including omentectomy, peritoneal biopsies, and contralateral ovarian biopsies when clinically indicated.
The mean hospital stay was 4.4 ± 3.8 days. Postoperative complications were documented in 6 patients and included wound infections, prolonged ileus, and other minor complications, all of which resolved with conservative management. No major surgical complications or perioperative mortality occurred in this series.
The mean follow-up duration was 4.0 ± 3.6 years. During the follow-up period, 3 patients (3.4%) developed local recurrence: one serous cystadenoma and one mucinous cystadenoma, both following ovarian-sparing surgery, and one yolk sac tumor following salpingo-oophorectomy. Both benign recurrences were successfully managed with repeat ovarian-sparing surgery. The patient with recurrent yolk sac tumor underwent re-resection and chemotherapy. One additional patient (1.2%) with an immature teratoma developed metastatic disease following salpingo-oophorectomy. All four patients with recurrence or metastasis had tumor rupture during the initial surgery (p<0.001). The overall recurrence rate was similar between the ovarian-sparing surgery group (2 of 41, 4.9%) and the oophorectomy group (1 of 46, 2.2%) (p=0.90).
One patient died during the follow-up period due to progressive lymphoma. No patient developed contralateral ovarian tumors requiring surgical intervention during the follow-up period, although 2 patients had metachronous bilateral disease at initial presentation.
Discussion
From a comprehensive review of 324 patients who underwent surgery for ovarian pathologies over the 20-year period, this study focused on the 87 patients (26.9%) with histopathologically confirmed ovarian tumors. This proportion is consistent with reported rates in the literature, where true neoplasms account for 20-30% of pediatric ovarian masses requiring surgical intervention.1-3
The demographic and clinical characteristics of our cohort align with previously published pediatric series.1-8,12,13,17,18 The median age of 11.9 years is consistent with the peak incidence of ovarian tumors in late childhood and adolescence. The predominance of abdominal pain as the presenting symptom reflects the high frequency of complications such as torsion, rupture, or rapid tumor growth in this age group.1,4,5
Our age-based subgroup analysis revealed important differences between prepubertal and pubertal patients. The significantly higher malignancy rate in prepubertal patients (29.5% vs 11.6%, p=0.039) is consistent with the literature suggesting that malignant germ cell tumors are more common in younger children.7,8 This finding has important implications for surgical planning, as prepubertal patients may warrant more thorough preoperative evaluation for malignancy. The predominance of germ cell tumors in prepubertal patients (84.1%) versus the higher proportion of epithelial tumors in pubertal patients (30.2%) reflects the different tumor biology across age groups. Notably, all sex cord-stromal tumors occurred in pubertal patients, which may be related to hormonal influences on these tumor types. These age-related differences support the recommendation for age-specific risk assessment algorithms in pediatric ovarian tumors.
The presence of ovarian torsion in 20.7% of cases deserves special mention, as torsion is one of the most common complications of pediatric ovarian masses and often necessitates emergency surgery.9,15 The American Pediatric Surgical Association systematic review of 96 studies found overwhelming evidence supporting ovarian detorsion rather than oophorectomy, with no reported thromboembolic events after detorsion and minimal risk of occult malignancy.26 Our analysis revealed that torsion was associated with a remarkably low malignancy rate of only 5.6%, consistent with the 0.4–5% malignancy rate reported in torsed ovaries.26 However, despite this low malignancy rate, ovarian-sparing surgery was performed in only 33.3% of torsion cases compared to 50.7% in non-torsion cases, reflecting earlier institutional practice where surgical decision-making was primarily driven by ovarian viability and circulation status after detorsion. Current evidence demonstrates that gross ovarian appearance does not correlate with long-term viability, as follicular development has been documented even in necrotic-appearing ovaries.9,15,26 Accordingly, our protocol has evolved: ovarian-sparing surgery is now performed in all torsion cases regardless of circulation status, unless the ovary is frankly necrotic or malignancy is confirmed or suspected.
Consistent with pediatric tumor patterns, over half of our cases were mature teratomas, whereas epithelial tumors predominate in adults.3,7 Our overall malignancy rate (~20%) lies within the expected 10-20% range for pediatric ovarian tumors.1,7,8 The decrease in malignancy rate from 33.3% in the earlier decade to 14.0% in the later decade likely reflects enhanced preoperative diagnostics, including better imaging and malignancy risk algorithms, which allow identification and removal of benign tumors earlier before they become so large that they appear malignant.10,14 We observed significantly smaller tumors in the later period (median 8.0 cm vs 13.5 cm, p<0.001), consistent with earlier detection through improved imaging.
The most notable finding in our study is the dramatic three-fold increase in ovarian-sparing surgery over time, from 20.0% in the first decade to 61.4% in the second decade. This evolution parallels global trends reported in the literature.16-24 Abbas et al. and Oue et al. found that OSS can be performed safely not only for benign tumors but even for select borderline or low-grade malignant cases, without compromising outcomes.12,13 In Poland, Szymon et al. observed OSS usage climb from 31% to 75% over 20 years, reflecting a similar change in practice.21 A recent systematic review and meta-analysis by Pio et al., encompassing 1,734 cases, demonstrated that ovarian-sparing surgery achieves excellent oncologic outcomes with no recurrence disadvantage compared to oophorectomy.18 Similarly, Łuczak et al. reported over two decades of success with ovarian preservation, emphasizing the importance of thorough preoperative evaluation.23,24
Importantly, the favorable outcomes observed in our OSS group must be interpreted in the context of careful patient selection. Patients selected for ovarian-sparing surgery had significantly smaller tumors (median 7 cm vs 12 cm) and markedly lower malignancy rates (2.4% vs 37.0%) compared to those undergoing oophorectomy. This selection bias is inherent to the surgical decision-making process: surgeons appropriately reserve OSS for tumors that appear benign based on preoperative and intraoperative assessment. Therefore, while our data confirm that OSS is safe in appropriately selected patients, they cannot demonstrate equivalence between OSS and oophorectomy for all tumor types. The excellent outcomes in the OSS group reflect successful identification of suitable candidates rather than proof that OSS would be equally safe for tumors currently treated with oophorectomy.
Tumor size emerged as an important discriminator between the OSS and oophorectomy groups (median 7 cm vs 12 cm, p<0.001). In general, larger tumors are more often malignant and can completely obliterate the normal ovary, making preservation unfeasible.8,12,14,23 However, size alone is not an automatic contraindication for OSS; we successfully performed OSS on some very large masses (up to 25 cm) when other factors indicated benign disease. This experience, similar to Łuczak et al.’s report, underscores that the surgeon’s intraoperative evaluation of tumor characteristics is more important than any strict size cutoff for deciding on ovarian preservation.23 This approach is also supported by McCauley et al., who demonstrated that OSS is feasible even for giant cystic masses ≥15 cm, with 62.5% of preoperative candidates successfully undergoing ovarian preservation and 100% showing morphologically normal ovarian tissue on postoperative imaging.27 Each case should be assessed individually, integrating imaging, tumor marker results, intraoperative findings, and frozen section pathology when available.16,18,21,23
Frozen section analysis has limited utility in pediatric ovarian tumors, largely because most are germ cell in origin and exhibit marked histologic diversity.25 In our study, frozen section was used in only 9 patients (10.3%). A recent Children’s Oncology Group study demonstrated only 61.7% diagnostic accuracy in pediatric germ cell tumors.25 The poor performance reflects the heterogeneous and often mixed composition of these tumors, predisposing them to sampling error.
Our outcomes affirm that OSS did not increase recurrence risk. Recurrence occurred in 4.9% of OSS patients versus 2.2% of those who had an oophorectomy, a non-significant difference (p=0.90). This aligns with published recurrence rates of 2-5% after fertility-sparing ovarian surgery.18,20,21,23 The two benign recurrences in the OSS group (serous cystadenoma and mucinous cystadenoma) were effectively treated with repeat conservative surgery, preserving ovarian function. The single malignant recurrence (yolk sac tumor) occurred in a patient who had undergone salpingo-oophorectomy, demonstrating that recurrence risk is not unique to OSS. Notably, all patients with recurrence or metastasis had experienced intraoperative tumor rupture during initial surgery, highlighting tumor rupture as a critical modifiable risk factor.8,23,24 McCauley et al. further confirmed the safety of OSS, reporting that all patients who underwent ovarian-sparing surgery for giant masses showed morphologically normal ovarian tissue on postoperative imaging with only one recurrence.27
The use of laparoscopy increased markedly in our cohort, rising from 6.7% to 35.1% (p=0.008), paralleling the global shift toward minimally invasive surgery in pediatric practice. Piotrowska-Gall et al. and Guillén et al. have demonstrated that laparoscopic ovarian-sparing surgery is both safe and feasible.19,20 In our series, laparoscopy was used significantly more often in ovarian-sparing operations than in oophorectomy (39.0% vs 13.0%, p=0.011), likely reflecting its suitability for smaller, benign-appearing tumors.
The importance of fertility preservation cannot be overstated in this young population. Even the loss of one ovary can potentially diminish future ovarian reserve and hormonal function, a concern that becomes critical if a problem later arises in the remaining ovary.11,13,21 The sharp rise in our use of OSS reflects growing awareness of these fertility issues among both surgeons and families. However, fertility preservation should never compromise oncologic safety, and our data demonstrate that these goals are compatible when patients are carefully selected. Zhao et al. recently demonstrated excellent fertility-sparing outcomes even in borderline ovarian tumors in children and adolescents, further supporting the safety of this approach.22
Several limitations of our study merit consideration. The retrospective design carries inherent limitations, including potential selection bias. As a single-center study, our findings may not be fully generalizable. The mean follow-up of 4 years may be insufficient to capture late complications or assess long-term fertility outcomes. Importantly, we did not systematically assess long-term ovarian function; anti-Müllerian hormone levels and pubertal hormone profiles were not routinely measured during follow-up, precluding direct evaluation of fertility preservation outcomes. Our sample size, particularly for malignant tumors, limits the power to detect differences in rare outcomes. Despite these limitations, our data provide valuable epidemiological information on pediatric ovarian tumor distribution, and the 20-year timeframe documents a significant paradigm shift toward fertility preservation.
In conclusion, our 20-year experience demonstrates a significant evolution in the surgical management of pediatric ovarian tumors, with ovarian-sparing surgery rates increasing from 20% to 61.4%. This shift toward fertility preservation has been achieved without compromising oncologic safety as evidenced by comparable recurrence rates between surgical approaches. The higher malignancy rate in prepubertal patients highlights the importance of age-specific evaluation. Torsion was associated with low malignancy risk, supporting a conservative approach. Our findings support individualized surgical decision-making that prioritizes fertility preservation in appropriately selected patients based on careful preoperative evaluation and intraoperative assessment, while acknowledging that the favorable outcomes in OSS reflect careful patient selection. Future research should focus on developing validated risk stratification tools, standardized management protocols for torsion cases, and prospective assessment of long-term fertility outcomes.
Ethical approval
The study was approved by Dr Sami Ulus Maternity and Children’s Health and Diseases Training and Research Hospital Ethics Committee (date: September 22, 2020, number: 2020-KAEK-141/432-E-22/09-427). Due to the retrospective nature of this study and the use of anonymized data from medical records, the requirement for informed consent was waived by the ethics committee.
Source of funding
The authors declare the study received no funding.
Conflict of interest
The authors declare that there is no conflict of interest.
References
- de Silva KS, Kanumakala S, Grover SR, Chow CW, Warne GL. Ovarian lesions in children and adolescents--an 11-year review. J Pediatr Endocrinol Metab 2004; 17: 951-957. https://doi.org/10.1515/jpem.2004.17.7.951
- Amies Oelschlager AM, Sawin R. Teratomas and ovarian lesions in children. Surg Clin North Am 2012; 92: 599-613, viii. https://doi.org/10.1016/j.suc.2012.03.005
- Hermans AJ, Kluivers KB, Wijnen MH, Bulten J, Massuger LF, Coppus SF. Diagnosis and treatment of adnexal masses in children and adolescents. Obstet Gynecol 2015; 125: 611-615. https://doi.org/10.1097/AOG.0000000000000665
- Taskinen S, Fagerholm R, Lohi J, Taskinen M. Pediatric ovarian neoplastic tumors: incidence, age at presentation, tumor markers and outcome. Acta Obstet Gynecol Scand 2015; 94: 425-429. https://doi.org/10.1111/aogs.12598
- Chen VW, Ruiz B, Killeen JL, Coté TR, Wu XC, Correa CN. Pathology and classification of ovarian tumors. Cancer 2003; 97: 2631-2642. https://doi.org/10.1002/cncr.11345
- Schultz KA, Ness KK, Nagarajan R, Steiner ME. Adnexal masses in infancy and childhood. Clin Obstet Gynecol 2006; 49: 464-479. https://doi.org/10.1097/00003081-200609000-00007
- Brookfield KF, Cheung MC, Koniaris LG, Sola JE, Fischer AC. A population-based analysis of 1037 malignant ovarian tumors in the pediatric population. J Surg Res 2009; 156: 45-49. https://doi.org/10.1016/j.jss.2009.03.069
- Papic JC, Finnell SM, Slaven JE, Billmire DF, Rescorla FJ, Leys CM. Predictors of ovarian malignancy in children: overcoming clinical barriers of ovarian preservation. J Pediatr Surg 2014; 49: 144-7; discussion 147-8. https://doi.org/10.1016/j.jpedsurg.2013.09.068
- Cass DL, Hawkins E, Brandt ML, et al. Surgery for ovarian masses in infants, children, and adolescents: 102 consecutive patients treated in a 15-year period. J Pediatr Surg 2001; 36: 693-699. https://doi.org/10.1053/jpsu.2001.22939
- Templeman CL, Fallat ME. Benign ovarian masses. Semin Pediatr Surg 2005; 14: 93-99. https://doi.org/10.1053/j.sempedsurg.2005.01.004
- Canlorbe G, Chabbert-Buffet N, Uzan C. Fertility-Sparing Surgery for Ovarian Cancer. J Clin Med 2021; 10: 4235. https://doi.org/10.3390/jcm10184235
- Oue T, Uehara S, Sasaki T, et al. Treatment and ovarian preservation in children with ovarian tumors. J Pediatr Surg 2015; 50: 2116-2118. https://doi.org/10.1016/j.jpedsurg.2015.08.036
- Abbas PI, Dietrich JE, Francis JA, Brandt ML, Cass DL, Lopez ME. Ovarian-Sparing Surgery in Pediatric Benign Ovarian Tumors. J Pediatr Adolesc Gynecol 2016; 29: 506-510. https://doi.org/10.1016/j.jpag.2016.03.009
- Oltmann SC, Garcia N, Barber R, Huang R, Hicks B, Fischer A. Can we preoperatively risk stratify ovarian masses for malignancy? J Pediatr Surg 2010; 45: 130-134. https://doi.org/10.1016/j.jpedsurg.2009.10.022
- Madenci AL, Vandewalle RJ, Dieffenbach BV, et al. Multicenter pre-operative assessment of pediatric ovarian malignancy. J Pediatr Surg 2019; 54: 1921-1925. https://doi.org/10.1016/j.jpedsurg.2019.02.019
- Birbas E, Kanavos T, Gkrozou F, Skentou C, Daniilidis A, Vatopoulou A. Ovarian Masses in Children and Adolescents: A Review of the Literature with Emphasis on the Diagnostic Approach. Children (Basel) 2023; 10: 1114. https://doi.org/10.3390/children10071114
- Bayramoglu Z, Timur B, Kızmazoglu D, et al. Pediatric and young adult ovarian masses: clinical approach, diagnostic evaluation, and management. Front Pediatr 2025; 13: 1639582. https://doi.org/10.3389/fped.2025.1639582
- Pio L, Abu-Zaid A, Zaghloul T, et al. Ovarian-sparing surgery for ovarian tumors in children: A systematic review and meta-analysis. Eur J Surg Oncol 2023; 49: 106923. https://doi.org/10.1016/j.ejso.2023.04.022
- Piotrowska-Gall A, Strzelecka A, Wróbel J, et al. Laparoscopic Ovarian-Sparing Surgery for the Management of Benign Ovarian Lesions in Pediatric Patients: A Retrospective Analysis. J Pediatr Surg 2024; 59: 400-406. https://doi.org/10.1016/j.jpedsurg.2023.10.057
- Guillén G, Martín-Giménez MP, López-Fernández S, Molino JA, López M. Results of Ovarian Sparing Surgery in Pediatric Patients: Is There a Place for Laparoscopy? J Laparoendosc Adv Surg Tech A 2020; 30: 458-463. https://doi.org/10.1089/lap.2019.0515
- Szymon O, Bogusz B, Taczanowska-Niemczuk A, Maślanka M, Górecki W. Ovarian Sparing Surgery in Mature Ovarian Teratomas in Children: A 20-Year Single-Center Experience. Eur J Pediatr Surg 2021; 31: 2-7. https://doi.org/10.1055/s-0040-1716877
- Zhao J, Wang D, Wang R, et al. Fertility-sparing surgery in children and adolescents with borderline ovarian tumors: a retrospective study. J Ovarian Res 2024; 17: 96. https://doi.org/10.1186/s13048-024-01409-0
- Łuczak J, Bagłaj M. Selecting treatment method for ovarian masses in children - 24 years of experience. J Ovarian Res 2017; 10: 59. https://doi.org/10.1186/s13048-017-0353-0
- Łuczak J, Bagłaj M, Dryjański P, et al. What Should Be the Topics of a Prospective Study on Ovarian Masses in Children?-Results of a Multicenter Retrospective Study and a Scoping Literature Review. Curr Oncol 2022; 29: 1488-1500. https://doi.org/10.3390/curroncol29030125
- Dicken BJ, Billmire DF, Rich B, et al. Utility of frozen section in pediatric and adolescent malignant ovarian nonseminomatous germ cell tumors: A report from the children’s oncology group. Gynecol Oncol 2022; 166: 476-480. https://doi.org/10.1016/j.ygyno.2022.06.013
- Dasgupta R, Renaud E, Goldin AB, et al. Ovarian torsion in pediatric and adolescent patients: A systematic review. J Pediatr Surg 2018; 53: 1387-1391. https://doi.org/10.1016/j.jpedsurg.2017.10.053
- McCauley CJ, Foote M, Laronda MM, Grabowski J, Rowell EE, Lautz TB. Ovarian sparing surgery can be achieved for many pediatric patients with giant adnexal cystic masses. J Pediatr Surg 2026; 61: 162871. https://doi.org/10.1016/j.jpedsurg.2025.162871
License
Copyright (c) 2026 The Author(s). This is an open access article distributed under the Creative Commons Attribution License (CC BY), which permits unrestricted use, distribution, and reproduction in any medium or format, provided the original work is properly cited.















